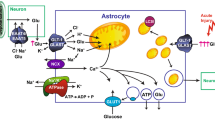
Abstract
The primary function assigned to the sodium-dependent glutamate transporters, also known as excitatory amino acid transporters (EAATs), is to maintain the extracellular glutamate concentration in the low micromolar range, allowing glutamate to be used as a signaling molecule in the brain and preventing its cytotoxic effects. However, glutamate and cyst(e)ine, that is also a substrate of EAATs, are also important metabolites used for instance in the synthesis of the main antioxidant glutathione. This review describes the evidence suggesting that EAATs, by providing glutathione precursors, are crucial to prevent oxidative death in particular cells of the nervous system while being dispensable in others. This differential importance may depend on the way antioxidant defenses are maintained in each cell type and on the metabolic fate of transported substrates, both being probably controlled by EAAT interacting proteins. As oxidative stress invariably contributes to various forms of cell death, a better understanding of how antioxidant defenses are maintained in particular brain cells will probably help to develop protective strategies in degenerative insults specifically affecting these cells.




Similar content being viewed by others
References
Agulhon C, Petravicz J, McMullen AB et al (2008) What is the role of astrocyte calcium in neurophysiology? Neuron 59:932–946
Allen JW, Shanker G, Aschner M (2001) Methylmercury inhibits the in vitro uptake of the glutathione precursor, cystine, in astrocytes, but not in neurons. Brain Res 894:131–140
Aoyama K, Suh SW, Hamby AM et al (2006) Neuronal glutathione deficiency and age-dependent neurodegeneration in the EAAC1 deficient mouse. Nat Neurosci 9:119–126
Aoyama K, Watabe M, Nakaki T (2011) Modulation of neuronal glutathione synthesis by EAAC1 and its interacting protein GTRAP3-18. Amino Acids (this issue)
Araque A, Parpura V, Sanzgiri RP et al (1999) Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 22:208–215
Araque A, Li N, Doyle RT et al (2000) SNARE protein-dependent glutamate release from astrocytes. J Neurosci 20:666–673
Arriza JL, Fairman WA, Wadiche JI et al (1994) Functional comparisons of three glutamate transporter subtypes cloned from human motor cortex. J Neurosci 14:5559–5569
Arriza JL, Eliasof S, Kavanaugh MP et al (1997) Excitatory amino acid transporter 5, a retinal glutamate transporter coupled to a chloride conductance. Proc Natl Acad Sci USA 94:4155–4160
Attwell D, Barbour B, Szatkowski M (1993) Nonvesicular release of neurotransmitter. Neuron 11:401–407
Bak LK, Schousboe A, Waagepetersen HS (2006) The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem 98:641–653
Banjac A, Perisic T, Sato H et al (2008) The cystine/cysteine cycle: a redox cycle regulating susceptibility versus resistance to cell death. Oncogene 27:1618–1628
Bannai S (1986) Exchange of cystine and glutamate across plasma membrane of human fibroblasts. J Biol Chem 261:2256–2263
Beart PM, O’Shea RD (2007) Transporters for l-glutamate: an update on their molecular pharmacology and pathological involvement. Br J Pharmacol 150:5–17
Bellocchio EE, Reimer RJ, Fremeau RT Jr et al (2000) Uptake of glutamate into synaptic vesicles by an inorganic phosphate transporter. Science 289:957–960
Bender AS, Reichelt W, Norenberg MD (2000) Characterization of cystine uptake in cultured astrocytes. Neurochem Int 37:269–276
Bockaert J, Fagni L, Dumuis A et al (2004) GPCR interacting proteins (GIP). Pharmacol Ther 103:203–221
Boudker O, Ryan RM, Yernool D et al (2007) Coupling substrate and ion binding to extracellular gate of a sodium-dependent aspartate transporter. Nature 445:387–393
Bridges RJ, Esslinger CS (2005) The excitatory amino acid transporters: pharmacological insights on substrate and inhibitor specificity of the EAAT subtypes. Pharmacol Ther 107:271–285
Bridges RJ, Stanley MS, Anderson MW et al (1991) Conformationally defined neurotransmitter analogues. Selective inhibition of glutamate uptake by one pyrrolidine-2, 4-dicarboxylate diastereomer. J Med Chem 34:717–725
Brightman MW, Reese TS (1969) Junctions between intimately apposed cell membranes in the vertebrate brain. J Cell Biol 40:648–677
Brocke L, Bendahan A, Grunewald M et al (2002) Proximity of two oppositely oriented reentrant loops in the glutamate transporter GLT-1 identified by paired cysteine mutagenesis. J Biol Chem 277:3985–3992
Bruijn LI, Becher MW, Lee MK et al (1997) ALS-linked SOD1 mutant G85R mediates damage to astrocytes and promotes rapidly progressive disease with SOD1-containing inclusions. Neuron 18:327–338
Buddhala C, Hsu CC, Wu JY (2009) A novel mechanism for GABA synthesis and packaging into synaptic vesicles. Neurochem Int 55:9–12
Charles AC, Merrill JE, Dirksen ER et al (1991) Intercellular signaling in glial cells: calcium waves and oscillations in response to mechanical stimulation and glutamate. Neuron 6:983–992
Chen W, Aoki C, Mahadomrongkul V et al (2002) Expression of a variant form of the glutamate transporter GLT1 in neuronal cultures and in neurons and astrocytes in the rat brain. J Neurosci 22:2142–2152
Chen W, Mahadomrongkul V, Berger UV et al (2004) The glutamate transporter GLT1a is expressed in excitatory axon terminals of mature hippocampal neurons. J Neurosci 24:1136–1148
Cho Y, Bannai S (1990) Uptake of glutamate and cysteine in C-6 glioma cells and in cultured astrocytes. J Neurochem 55:2091–2097
Conn PJ (2003) Physiological roles and therapeutic potential of metabotropic glutamate receptors. Ann NY Acad Sci 1003:12–21
Conrad M, Sato H (2011) The oxidative stress-inducible cystine/glutamate antiporter, system x −c : cystine supplier and beyond. Amino Acids (this issue)
Cornell-Bell AH, Finkbeiner SM, Cooper MS et al (1990) Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 247:470–473
Curtis DR, Phillis JW, Watkins JC (1959) Chemical excitation of spinal neurones. Nature 183:611–612
D’Antoni S, Berretta A, Bonaccorso CM et al (2008) Metabotropic glutamate receptors in glial cells. Neurochem Res 33:2436–2443
Damier P, Hirsch EC, Zhang P et al (1993) Glutathione peroxidase, glial cells and Parkinson’s disease. Neuroscience 52:1–6
Danbolt NC (2001) Glutamate uptake. Prog Neurobiol 65:1–105
David JC, Yamada KA, Bagwe MR et al (1996) AMPA receptor activation is rapidly toxic to cortical astrocytes when desensitization is blocked. J Neurosci 16:200–209
Dermietzel R, Traub O, Hwang TK et al (1989) Differential expression of three gap junction proteins in developing and mature brain tissues. Proc Natl Acad Sci USA 86:10148–10152
Diamond JS (2001) Neuronal glutamate transporters limit activation of NMDA receptors by neurotransmitter spillover on CA1 pyramidal cells. J Neurosci 21:8328–8338
Dringen R (2000) Metabolism and functions of glutathione in brain. Prog Neurobiol 62:649–671
Dringen R, Hamprecht B (1996) Glutathione content as an indicator for the presence of metabolic pathways of amino acids in astroglial cultures. J Neurochem 67:1375–1382
Dringen R, Kranich O, Loschmann PA et al (1997) Use of dipeptides for the synthesis of glutathione by astroglia-rich primary cultures. J Neurochem 69:868–874
Dringen R, Hamprecht B, Broer S (1998) The peptide transporter PepT2 mediates the uptake of the glutathione precursor CysGly in astroglia-rich primary cultures. J Neurochem 71:388–393
Dringen R, Pfeiffer B, Hamprecht B (1999) Synthesis of the antioxidant glutathione in neurons: supply by astrocytes of CysGly as precursor for neuronal glutathione. J Neurosci 19:562–569
Dringen R, Gutterer JM, Hirrlinger J (2000) Glutathione metabolism in brain metabolic interaction between astrocytes and neurons in the defense against reactive oxygen species. Eur J Biochem 267:4912–4916
Dringen R, Gutterer JM, Gros C et al (2001) Aminopeptidase N mediates the utilization of the GSH precursor CysGly by cultured neurons. J Neurosci Res 66:1003–1008
Dunlop J (2006) Glutamate-based therapeutic approaches: targeting the glutamate transport system. Curr Opin Pharmacol 6:103–107
Fairman WA, Vandenberg RJ, Arriza JL et al (1995) An excitatory amino-acid transporter with properties of a ligand-gated chloride channel. Nature 375:599–603
Fedoroff S, McAuley WA, Houle JD et al (1984) Astrocyte cell lineage. V. Similarity of astrocytes that form in the presence of dBcAMP in cultures to reactive astrocytes in vivo. J Neurosci Res 12:14–27
Fedoroff S, Ahmed I, Wang E (1990) The relationship of expression of statin, the nuclear protein of nonproliferating cells, to the differentiation and cell cycle of astroglia in cultures and in situ. J Neurosci Res 26:1–15
Feng W, Zhang M (2009) Organization and dynamics of PDZ-domain-related supramodules in the postsynaptic density. Nat Rev Neurosci 10:87–99
Fonnum F (1984) Glutamate: a neurotransmitter in mammalian brain. J Neurochem 42:1–11
Frigerio F, Casimir M, Carobbio S et al (2008) Tissue specificity of mitochondrial glutamate pathways and the control of metabolic homeostasis. Biochim Biophys Acta 1777:965–972
Gallo V, Ghiani CA (2000) Glutamate receptors in glia: new cells, new inputs and new functions. Trends Pharmacol Sci 21:252–258
Gegelashvili G, Schousboe A (1998) Cellular distribution and kinetic properties of high-affinity glutamate transporters. Brain Res Bull 45:233–238
Gegelashvili G, Civenni G, Racagni G et al (1996) Glutamate receptor agonists up-regulate glutamate transporter GLAST in astrocytes. Neuroreport 8:261–265
Gegelashvili G, Danbolt NC, Schousboe A (1997) Neuronal soluble factors differentially regulate the expression of the GLT1 and GLAST glutamate transporters in cultured astroglia. J Neurochem 69:2612–2615
Ghijsen WE, Leenders AG, Lopes da Silva FH (2003) Regulation of vesicle traffic and neurotransmitter release in isolated nerve terminals. Neurochem Res 28:1443–1452
Gouaux E (2009) The molecular logic of sodium-coupled neurotransmitter transporters. Philos Trans R Soc Lond B Biol Sci 364:149–154
Gouix E, Léveillé F, Nicole O et al (2009) Reverse glial glutamate uptake triggers neuronal cell death through extrasynaptic NMDA receptor activation. Mol Cell Neurosci 40:463–473
Gras G, Samah B, Hubert A et al. (2011) EEAT expression by macrophages and microglia: still more question than answers. Amino Acids (this issue)
Grewer C, Gameiro A, Zhang Z et al (2008) Glutamate forward and reverse transport: from molecular mechanism to transporter-mediated release after ischemia. IUBMB Life 60:609–619
Had L, Faivre-Sarrailh C, Legrand C et al (1993) The expression of tropomyosin genes in pure cultures of rat neurons, astrocytes and oligodendrocytes is highly cell-type specific and strongly regulated during development. Brain Res Mol Brain Res 18:77–86
Had-Aissouni L (2011) Maintenance of antioxidant defenses of brain cells: plasma membrane glutamate transporters and beyond. Amino Acids (this issue)
Had-Aissouni L, Re DB, Nieoullon A et al (2002) Importance of astrocytic inactivation of synaptically released glutamate for cell survival in the central nervous system—are astrocytes vulnerable to low intracellular glutamate concentrations? J Physiol Paris 96:317–322
Hamberger AC, Chiang GH, Nylen ES et al (1979) Glutamate as a CNS transmitter. I. Evaluation of glucose and glutamine as precursors for the synthesis of preferentially released glutamate. Brain Res 168:513–530
Hansson E, Ronnback L (2003) Glial neuronal signaling in the central nervous system. FASEB J 17:341–348
Harada T, Harada C, Nakamura K et al (2007) The potential role of glutamate transporters in the pathogenesis of normal tension glaucoma. J Clin Invest 117:1763–1770
Hardingham GE, Fukunaga Y, Bading H (2002) Extrasynaptic NMDARs oppose synaptic NMDARs by triggering CREB shut-off and cell death pathways. Nat Neurosci 5:405–414
Hassel B (2000) Carboxylation and anaplerosis in neurons and glia. Mol Neurobiol 22:21–40
Haugeto O, Ullensvang K, Levy LM et al (1996) Brain glutamate transporter proteins form homomultimers. J Biol Chem 271:27715–27722
Hayes D, Wiessner M, Rauen T et al (2005) Transport of l-[14C]cystine and l-[14C]cysteine by subtypes of high affinity glutamate transporters over-expressed in HEK cells. Neurochem Int 46:585–594
Hertz L, Dringen R, Schousboe A et al (1999) Astrocytes: glutamate producers for neurons. J Neurosci Res 57:417–428
Hertz L, Hansson E, Ronnback L (2001) Signaling and gene expression in the neuron-glia unit during brain function and dysfunction: Holger Hyden in memoriam. Neurochem Int 39:227–252
Hertz L, Peng L, Dienel GA (2007) Energy metabolism in astrocytes: high rate of oxidative metabolism and spatiotemporal dependence on glycolysis/glycogenolysis. J Cereb Blood Flow Metab 27:219–249
Himi T, Ikeda M, Yasuhara T et al (2003) Role of neuronal glutamate transporter in the cysteine uptake and intracellular glutathione levels in cultured cortical neurons. J Neural Transm 110:1337–1348
Hirrlinger J, Schulz JB, Dringen R (2002) Glutathione release from cultured brain cells: multidrug resistance protein 1 mediates the release of GSH from rat astroglial cells. J Neurosci Res 69:318–326
Hisano S (2003) Vesicular glutamate transporters in the brain. Anat Sci Int 78:191–204
Holmseth S, Scott HA, Real K et al (2009) The concentrations and distributions of three C-terminal variants of the GLT1 (EAAT2; slc1a2) glutamate transporter protein in rat brain tissue suggest differential regulation. Neuroscience 162:1055–1071
Huang YH, Bergles DE (2004) Glutamate transporters bring competition to the synapse. Curr Opin Neurobiol 14:346–352
Ivanov A, Pellegrino C, Rama S et al (2006) Opposing role of synaptic and extrasynaptic NMDA receptors in regulation of the extracellular signal-regulated kinases (ERK) activity in cultured rat hippocampal neurons. J Physiol 572:789–798
Kanai Y, Hediger MA (1992) Primary structure and functional characterization of a high-affinity glutamate transporter. Nature 360:467–471
Kavanaugh MP, Bendahan A, Zerangue N et al (1997) Mutation of an amino acid residue influencing potassium coupling in the glutamate transporter GLT-1 induces obligate exchange. J Biol Chem 272:1703–1708
Keelan J, Allen NJ, Antcliffe D et al (2001) Quantitative imaging of glutathione in hippocampal neurons and glia in culture using monochlorobimane. J Neurosci Res 66:873–884
Khanna S, Roy S, Ryu H et al (2003) Molecular basis of vitamin E action: tocotrienol modulates 12-lipoxygenase, a key mediator of glutamate-induced neurodegeneration. J Biol Chem 278:43508–43515
Kimelberg HK, Rutledge E, Goderie S et al (1995) Astrocytic swelling due to hypotonic or high K+ medium causes inhibition of glutamate and aspartate uptake and increases their release. J Cereb Blood Flow Metab 15:409–416
Koch HP, Kavanaugh MP, Esslinger CS et al (1999) Differentiation of substrate and nonsubstrate inhibitors of the high-affinity, sodium-dependent glutamate transporters. Mol Pharmacol 56:1095–1104
Kovacs AD, Cebers G, Cebere A et al (2002) Selective and AMPA receptor-dependent astrocyte death following prolonged blockade of glutamate reuptake in rat cerebellar cultures. Exp Neurol 174:58–71
Kranich O, Hamprecht B, Dringen R (1996) Different preferences in the utilization of amino acids for glutathione synthesis in cultured neurons and astroglial cells derived from rat brain. Neurosci Lett 219:211–214
Kranich O, Dringen R, Sandberg M et al (1998) Utilization of cysteine and cysteine precursors for the synthesis of glutathione in astroglial cultures: preference for cystine. Glia 22:11–18
Krebs HA (1935) Metabolism of amino-acids: the synthesis of glutamine from glutamic acid and ammonia, and the enzymic hydrolysis of glutamine in animal tissues. Biochem J 29:1951–1969
Krebs HA, Eggleston LV, Hems R (1949) Distribution of glutamine and glutamic acid in animal tissues. Biochem J 44:159–163
Kullmann DM, Erdemli G, Asztely F (1996) LTP of AMPA and NMDA receptor-mediated signals: evidence for presynaptic expression and extrasynaptic glutamate spill-over. Neuron 17:461–474
Lehre KP, Levy LM, Ottersen OP et al (1995) Differential expression of two glial glutamate transporters in the rat brain: quantitative and immunocytochemical observations. J Neurosci 15:1835–1853
Lewerenz J, Maher P, Methner A (2011) Regulation of xCT expression and system x −c function in neuronal cells. Amino Acids (this issue)
Lievens JC, Bernal F, Forni C et al (2000a) Characterization of striatal lesions produced by glutamate uptake alteration: cell death, reactive gliosis, and changes in GLT1 and GADD45 mRNA expression. Glia 29:222–232
Lievens JC, Salin P, Had-Aissouni L et al (2000b) Differential effects of corticostriatal and thalamostriatal deafferentation on expression of the glutamate transporter GLT1 in the rat striatum. J Neurochem 74:909–919
Liu D, Smith CL, Barone FC et al (1999) Astrocytic demise precedes delayed neuronal death in focal ischemic rat brain. Brain Res Mol Brain Res 68:29–41
Lodge D (2009) The history of the pharmacology and cloning of ionotropic glutamate receptors and the development of idiosyncratic nomenclature. Neuropharmacology 56:6–21
Longuemare MC, Rose CR, Farrell K et al (1999) K(+) -induced reversal of astrocyte glutamate uptake is limited by compensatory changes in intracellular Na+. Neuroscience 93:285–292
Lortet S, Samuel D, Had-Aissouni L et al (1999) Effects of PKA and PKC modulators on high affinity glutamate uptake in primary neuronal cell cultures from rat cerebral cortex. Neuropharmacology 38:395–402
Machiyama Y, Balazs R, Merei T (1970) Incorporation of (14C) glutamate into glutathione in rat brain. J Neurochem 17:449–453
Maciejewski PK, Rothman DL (2008) Proposed cycles for functional glutamate trafficking in synaptic neurotransmission. Neurochem Int 52:809–825
Mandal PK, Seiler A, Perisic T et al (2010) System x(c)- and thioredoxin reductase 1 cooperatively rescue glutathione deficiency J Biol Chem 285:22244–22253
Martin DL, Rimvall K (1993) Regulation of gamma-aminobutyric acid synthesis in the brain. J Neurochem 60:395–407
Martin LJ, Brambrink AM, Lehmann C et al (1997) Hypoxia-ischemia causes abnormalities in glutamate transporters and death of astroglia and neurons in newborn striatum. Ann Neurol 42:335–348
Martinez-Hernandez A, Bell KP, Norenberg MD (1977) Glutamine synthetase: glial localization in brain. Science 195:1356–1358
Massieu L, Morales-Villagran A, Tapia R (1995) Accumulation of extracellular glutamate by inhibition of its uptake is not sufficient for inducing neuronal damage: an in vivo microdialysis study. J Neurochem 64:2262–2272
Massieu L, Del Rio P, Montiel T (2001) Neurotoxicity of glutamate uptake inhibition in vivo: correlation with succinate dehydrogenase activity and prevention by energy substrates. Neuroscience 106:669–677
Matute C, Sanchez-Gomez MV, Martinez-Millan L et al (1997) Glutamate receptor-mediated toxicity in optic nerve oligodendrocytes. Proc Natl Acad Sci USA 94:8830–8835
Matute C, Alberdi E, Domercq M et al (2007) Excitotoxic damage to white matter. J Anat 210:693–702
McBean G (2011) The transsulfuration pathway: a source of cysteine for glutathione in astrocytes. Amino Acids (this issue)
McKenna MC (2007) The glutamate–glutamine cycle is not stoichiometric: fates of glutamate in brain. J Neurosci Res 85:3347–3358
McKenna MC, Tildon JT, Stevenson JH et al (1996) New insights into the compartmentation of glutamate and glutamine in cultured rat brain astrocytes. Dev Neurosci 18:380–390
Montana V, Ni Y, Sunjara V et al (2004) Vesicular glutamate transporter-dependent glutamate release from astrocytes. J Neurosci 24:2633–2642
Montana V, Malarkey EB, Verderio C et al (2006) Vesicular transmitter release from astrocytes. Glia 54:700–715
Montiel T, Camacho A, Estrada-Sanchez AM et al (2005) Differential effects of the substrate inhibitor l-trans-pyrrolidine-2, 4-dicarboxylate (PDC) and the non-substrate inhibitor dl-threo-beta-benzyloxyaspartate (dl-TBOA) of glutamate transporters on neuronal damage and extracellular amino acid levels in rat brain in vivo. Neuroscience 133:667–678
Murphy TH, Miyamoto M, Sastre A et al (1989) Glutamate toxicity in a neuronal cell line involves inhibition of cystine transport leading to oxidative stress. Neuron 2:1547–1558
Murphy TH, Schnaar RL, Coyle JT (1990) Immature cortical neurons are uniquely sensitive to glutamate toxicity by inhibition of cystine uptake. FASEB J 4:1624–1633
Musser DA, Oseroff AR (1994) The use of tetrazolium salts to determine sites of damage to the mitochondrial electron transport chain in intact cells following in vitro photodynamic therapy with Photofrin II. Photochem Photobiol 59:621–626
Mytilineou C, Kokotos Leonardi ET, Kramer BC et al (1999) Glial cells mediate toxicity in glutathione-depleted mesencephalic cultures. J Neurochem 73:112–119
Nafia I, Re DB, Masmejean F et al (2008) Preferential vulnerability of mesencephalic dopamine neurons to glutamate transporter dysfunction. J Neurochem 105:484–496
Naito S, Ueda T (1985) Characterization of glutamate uptake into synaptic vesicles. J Neurochem 44:99–109
Nedergaard M, Dirnagl U (2005) Role of glial cells in cerebral ischemia. Glia 50:281–286
Newpher TM, Ehlers MD (2008) Glutamate receptor dynamics in dendritic microdomains. Neuron 58:472–497
Oliet SH, Piet R, Poulain DA et al (2004) Glial modulation of synaptic transmission: Insights from the supraoptic nucleus of the hypothalamus. Glia 47:258–267
Papadia S, Hardingham GE (2007) The dichotomy of NMDA receptor signaling. Neuroscientist 13:572–579
Parpura V, Basarsky TA, Liu F et al (1994) Glutamate-mediated astrocyte-neuron signalling. Nature 369:744–747
Persson M, Rönnbäck L (2011) Microglial self defence mediated through GLT-1 and glutathione. Amino Acids (this issue)
Piet R, Vargova L, Sykova E et al (2004) Physiological contribution of the astrocytic environment of neurons to intersynaptic crosstalk. Proc Natl Acad Sci USA 101:2151–2155
Pines G, Danbolt NC, Bjoras M et al (1992) Cloning and expression of a rat brain l-glutamate transporter. Nature 360:464–467
Pinheiro PS, Mulle C (2008) Presynaptic glutamate receptors: physiological functions and mechanisms of action. Nat Rev Neurosci 9:423–436
Pow DV, Barnett NL (2000) Developmental expression of excitatory amino acid transporter 5: a photoreceptor and bipolar cell glutamate transporter in rat retina. Neurosci Lett 280:21–24
Re DB, Boucraut J, Samuel D et al (2003) Glutamate transport alteration triggers differentiation-state selective oxidative death of cultured astrocytes: a mechanism different from excitotoxicity depending on intracellular GSH contents. J Neurochem 85:1159–1170
Re DB, Nafia I, Melon C et al (2006) Glutamate leakage from a compartmentalized intracellular metabolic pool and activation of the lipoxygenase pathway mediate oxidative astrocyte death by reversed glutamate transport. Glia 54:47–57
Reichelt W, Stabel-Burow J, Pannicke T et al (1997) The glutathione level of retinal Muller glial cells is dependent on the high-affinity sodium-dependent uptake of glutamate. Neuroscience 77:1213–1224
Richman PG, Meister A (1975) Regulation of gamma-glutamyl-cysteine synthetase by nonallosteric feedback inhibition by glutathione. J Biol Chem 250:1422–1426
Robertson RT, Zimmer J, Gahwiler BH (1989) Dissection procedures for preparation of slice cultures. In A dissection and tissue culture manual of the nervous system. Alan R. Liss, New York pp 1–15
Robinson MB, Coyle JT (1987) Glutamate and related acidic excitatory neurotransmitters: from basic science to clinical application. FASEB J 1:446–455
Rothstein JD, Martin L, Levey AI et al (1994) Localization of neuronal and glial glutamate transporters. Neuron 13:713–725
Rothstein JD, Dykes-Hoberg M, Pardo CA et al (1996) Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 16:675–686
Rouach N, Avignone E, Meme W et al (2002) Gap junctions and connexin expression in the normal and pathological central nervous system. Biol Cell 94:457–475
Rutten EP, Engelen MP, Schols AM et al (2005) Skeletal muscle glutamate metabolism in health and disease: state of the art. Curr Opin Clin Nutr Metab Care 8:41–51
Sagara JI, Miura K, Bannai S (1993) Maintenance of neuronal glutathione by glial cells. J Neurochem 61:1672–1676
Sato H, Shiiya A, Kimata M et al (2005) Redox imbalance in cystine/glutamate transporter-deficient mice. J Biol Chem 280:37423–37429
Scemes E, Giaume C (2006) Astrocyte calcium waves: what they are and what they do. Glia 54:716–725
Schlag BD, Vondrasek JR, Munir M et al (1998) Regulation of the glial Na+-dependent glutamate transporters by cyclic AMP analogs and neurons. Mol Pharmacol 53:355–369
Schousboe A, Westergaard N, Sonnewald U et al (1993) Glutamate and glutamine metabolism and compartmentation in astrocytes. Dev Neurosci 15:359–366
Seiler A, Schneider M, Förster H et al (2008) Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab 8:237–248
Sem’yanov AV (2005) Diffusional extrasynaptic neurotransmission via glutamate and GABA. Neurosci Behav Physiol 35:253–266
Sepkuty JP, Cohen AS, Eccles C et al (2002) A neuronal glutamate transporter contributes to neurotransmitter GABA synthesis and epilepsy. J Neurosci 22:6372–6379
Shain W, Forman DS, Madelian V, Turner JN (1987) Morphology of astroglial cells is controlled by beta-adrenergic receptors. J Cell Biol 105:2307–2314
Shank RP, Bennett GS, Freytag SO et al (1985) Pyruvate carboxylase: an astrocyte-specific enzyme implicated in the replenishment of amino acid neurotransmitter pools. Brain Res 329:364–367
Shanker G, Aschner M (2001) Identification and characterization of uptake systems for cystine and cysteine in cultured astrocytes and neurons: evidence for methylmercury-targeted disruption of astrocyte transport. J Neurosci Res 66:998–1002
Shigeri Y, Seal RP, Shimamoto K (2004) Molecular pharmacology of glutamate transporters, EAATs and VGLUTs. Brain Res Brain Res Rev 45:250–265
Shine HD, Haber B (1981) Immunocytochemical localization of gamma-glutamyl transpeptidase in the rat CNS. Brain Res 217:339–349
Storck T, Schulte S, Hofmann K et al (1992) Structure, expression, and functional analysis of a Na(+) -dependent glutamate/aspartate transporter from rat brain. Proc Natl Acad Sci USA 89:10955–10959
Swanson RA, Liu J, Miller JW et al (1997) Neuronal regulation of glutamate transporter subtype expression in astrocytes. J Neurosci 17:932–940
Szatkowski M, Barbour B, Attwell D (1990) Non-vesicular release of glutamate from glial cells by reversed electrogenic glutamate uptake. Nature 348:443–446
Takamori S (2006) VGLUTs: ‘exciting’ times for glutamatergic research? Neurosci Res 55:343–351
Takamori S, Rhee JS, Rosenmund C et al (2000) Identification of a vesicular glutamate transporter that defines a glutamatergic phenotype in neurons. Nature 407:189–194
Tan S, Schubert D, Maher P (2001) Oxytosis: a novel form of programmed cell death. Curr Top Med Chem 1:497–506
Tibbs GR, Barrie AP, Van Mieghem FJ et al (1989) Repetitive action potentials in isolated nerve terminals in the presence of 4-aminopyridine: effects on cytosolic free Ca2+ and glutamate release. J Neurochem 53:1693–1699
Tobaben S, Grohm J, Seiler A et al (2011) Bid-mediated mitochondrial damage is a key mechanism in glutamate-induced oxidative stress and AIF-dependent cell death in immortalized HT-22 hippocampal neurons. Cell Death Differ 18:282–292
Trotti D, Rolfs A, Danbolt NC et al (1999) SOD1 mutants linked to amyotrophic lateral sclerosis selectively inactivate a glial glutamate transporter. Nat Neurosci 2:848
Verkhratsky A (2009) Neuronismo y reticulismo: neuronal-glial circuits unify the reticular and neuronal theories of brain organization. Acta Physiol (Oxf) 195:111–122
Verkhratsky A, Kirchhoff F (2007) NMDA receptors in glia. Neuroscientist 13:28–37
Volterra A, Meldolesi J (2005) Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci 6:626–640
Watabe M, Aoyama K, Nakaki T (2008) A dominant role of GTRAP3–18 in neuronal glutathione synthesis. J Neurosci 28:9404–9413
Watkins JC (2000) l-glutamate as a central neurotransmitter: looking back. Biochem Soc Trans 28:297–309
Yamada K, Watanabe M, Shibata T et al (1996) EAAT4 is a post-synaptic glutamate transporter at Purkinje cell synapses. Neuroreport 7:2013–2017
Yernool D, Boudker O, Jin Y et al (2004) Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431:811–818
Yi Z, Petralia RS, Fu Z et al (2007) The role of the PDZ protein GIPC in regulating NMDA receptor trafficking. J Neurosci 27:11663–11675
Acknowledgments
The work described in this review was supported by grants from different French research agencies and foundations: CNRS, Aix-Marseille University, DGA, “NRJ-Institut de France”, “Fédération Française des Groupements de Parkinsoniens” and “France Parkinson”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Had-Aissouni, L. Toward a new role for plasma membrane sodium-dependent glutamate transporters of astrocytes: maintenance of antioxidant defenses beyond extracellular glutamate clearance. Amino Acids 42, 181–197 (2012). https://doi.org/10.1007/s00726-011-0863-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-0863-9