Abstract
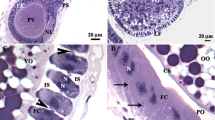
Vitellogenin receptor (VgR) is a low-density lipoprotein receptor responsible for the mediated endocytosis of vitellogenin (Vg) during egg formation in insects. The maturing oocyte is enveloped by a follicular epithelium, which has large intercellular spaces during Vg accumulation (patency). However, Vg has been reported in the cytoplasm of follicular cells, indicating that there may be a transcellular route for its transport. This study verified the presence of VgR in the follicular cells of the ovaries of the honeybee Apis mellifera and the wasp Polistes simillimus in order to evaluate if Vg is transported via transcytosis in these insects. Antibodies specific for vitellogenin receptor (anti-VgR), vitellogenin (anti-Vg), and clathrin (anti-Clt) were used for immunolocalization. The results showed the presence of VgR on the apical and basal plasma membranes of follicular cells of the vitellogenic follicles in both species, indicating that VgR may have been transported from the basal to the apical cell domain, followed by its release into the perivitelline space, evidenced by the presence of apical plasma membrane projections containing VgR. Co-localization proved that Vg bind to VgR and that the transport of this protein is mediated by clathrin. These data suggest that, in these social insects, Vg is transported via clathrin-mediated VgR transcytosis in follicular cells.






Similar content being viewed by others
References
Alberts B, Bray D, Lewis J, Raff M, Roberts K, Walter P (2014) Molecular biology of the cell. Gerland Science, New York
Anderson LM, Telfer WH (1970) Extracellular concentration of proteins in the Cecropia moth follicle. J Cell Physiol 76:37–53. https://doi.org/10.1002/jcp.1040760108
Azevedo DO, Zanuncio JC, Delabie JHC, Serrão JE (2011) Temporal variation of vitellogenin synthesis in Ectatomma tuberculatum (Formicidae: Ectatomminae) workers. J Ins Physiol 57:972–977. https://doi.org/10.1016/j.jinsphys.2011.04.015
Chapman RF (2013) The insect: structure and function. Cambrigde University Press, Cambridge
Ciudad L, Piulachs MD, Bellés X (2006) Systemic RNAi of the cockroach vitellogenin receptor results in a phenotype similar to that Drosophila yolkless mutant. FEBS J 273:325–335. https://doi.org/10.1111/j.1742-4658.2005.05066.x
Cruz-Landim C, Roat TC, Berger B (2013) Fat body, hemolymph and ovary routes for delivery of substances to ovary in Melipona quadrifasciata anthidioides: differences among castes through the use of electron-opaque tracers. Microscopy 62:457–466. https://doi.org/10.1093/jmicro/dft018
Davey KG (1981) Hormonal control of vitellogenin uptake in Rhodnius prolixus Stal. Am Zool 21:701–705
Dhadialla TS, Hays AR, Raikhel AS (1992) Characterization of the solubilized mosquito vitellogenin receptor. Ins Biochem Molec Biol 22:803–816. https://doi.org/10.1016/0965-1748(92)90107-P
Fleig R (1995) Role of the follicle cells for yolk uptake in ovarian follicles of the honey bee Apis mellifera L. (Hymenoptera: Apidae). Int J Ins Morphol Embryol 24:427–433. https://doi.org/10.1016/0020-7322(95)98841-Z
Goldstein JL, Brown MS, Anderson RGW, Russel DW, Scheneider WJ (1985) Receptor-mediated endocytosis: concepts emerging from de LDL receptor system. Ann Rev Cell Biol 1:1–39. https://doi.org/10.1146/annurev.cb.01.110185
Han C, Chen E, Shen G, Peng Z, Xu Y, Zhang H, Liu H, Zhang Y, Wu J, Lin Y, Xia Q (2017) Vitellogenin receptor selectively endocytoses female-specific and highly-expressed hemolymph proteins in the silkworm Bombyx mori. Biochem Cell Biol 95:510–516. https://doi.org/10.1139/bcb-2016-0255
Herz J, Clouthier DE, Hammer RE (1992) LDL receptor-related protein internalizes and degrades uPA-Pal-1 complexes and is essential embryo implantation. Cell 71:411–421. https://doi.org/10.1016/0092-8674(92)90511-A
Laemmli UK (1970) Clavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lauverjat S, Szollosi A, Marcaillou C (1984) Permeability of the ovarian follicle during oogenesis in Locusta migratoria L. (insect: Orthoptera). J Ultrastruct Res 87:197–211. https://doi.org/10.1016/S0022-5320(84)80060-X
Lisboa LCO, Serrão JE, Cruz-Landim C, Campos LAO (2005) Effect of larval food amount on ovariole development in queens of Trigona spinipes (Hymenoptera, Apinae). Anat Hist Embryol 34:179–184. https://doi.org/10.1111/j.1439-0264.2005.00591.x
Lu AL, Vinson SB, Pietrantonio PV (2009) Oocyte membrane localization of vitellogenin receptor coincides with queen flying age, and receptor silencing by RNAi disrupts egg formation in fire ant virgin queens. FEBS J 276:3110–3123. https://doi.org/10.1111/j.1742-4658.2009.07029.x
Mukherjee S, Ghosh RN, Maxfield FR (1997) Endocytosis. Physiol Rev 77:759–803. https://doi.org/10.1152/physrev.1997.77.3.759
Raikhel AS, Dhadialla TS (1992) Accumulation of yolk proteins in insects oocytes. Ann Rev Ent 37:217–251. https://doi.org/10.1146/annurev.en.37.010192.001245
Raikhel AS, Lea AO (1991) Control of follicular epithelium developmental and vitelline envelope formation in the mosquito; role of juvenile hormone and 20-hydroxyecdysone. Tiss Cell 23:577–591. https://doi.org/10.1016/0040-8166(91)90015-L
Ronnau M, Azevedo DO, Fialho MCQ, Gonçalves WG, Zanuncio JC, Serrão JE (2015) Changes in follicular cells architecture during vitellogenin transport in ovary of social Hymenoptera. Protoplasma 253:815–829. https://doi.org/10.1007/s00709-015-0843-0
Roth TF, Porter KR (1964) Yolk protein uptake in the oocyte of the mosquito Aedes aegypti L. J Cell Biol 20:313–332. https://doi.org/10.1083/jcb.20.2.313
Sappington TW, Raikhel AS (1998) Molecular characteristics of insect vitellogenins and vitellogenin receptor. Ins Biochem Molec Biol 28:277–300
Sappington TW, Hays AR, Raikhel AS (1995) Mosquito vitellogenin: purification, developmental and biochemical characterization. Ins Biochem Molecr Biol 25:807–817. https://doi.org/10.1016/0965-1748(95)00016-O
Schmidt-Capella IC, Hartfelder K (1998) Juvenile hormone effect on DNA synthesi and apoptosis in caste-specific differentiation larval honeybee (Apis mellifera L.) ovary. J Ins Physiol 44:385–391. https://doi.org/10.1016/S0022-1910(98)00027-4
Schonbaum CP, Lee S, Mahowald AP (1995) The Drosophila yolkless gene encodes a vitellogenin receptor belonging to the low density lipoprotein receptor superfamily. Proc Natl Acad Sci U S A 92:1485–1489
Schonbaum CP, Perrino JJ, Mahowald AP (2000) Regulation of the vitellogenin receptor during Drosophila melanogaster oogenesis. Mol Biol Cell 11:511–521
Seehuus SC, Norberg K, Krekling T, Fondrk K, Amdam GV (2007) Immunogold localization of vitellogenin in the ovaries, hypopharyngeal glands and head fat bodies of honeybee workers, Apis mellifera. Int J Insect Sci 7(52):1–14. https://doi.org/10.1673/031.007.5201
Telfer WH (1961) The route of entry and localization of blood protein in the oocytes of saturniid moths. J Biophys Biochem Citol 9:747–753. https://doi.org/10.1083/jcb.9.4.747
Telfer WH, Huebner E, Smith DS (1982) The cell biology of vitellogenic follicles in Hyalophora and Rhodnius. In: King RC, Akai H (eds) Insect Ultrastructure, Vol 1. Plenum Press, New York, pp 118–149
Tufail M, Takeda M (2005) Molecular cloning characterization and regulation of the cockroach vitellogenin receptor during oogenesis. Ins Molec Biol 14:389–401. https://doi.org/10.1111/j.1365-2583.2005.00570.x
Tufail M, Takeda M (2007) Molecular cloning and developmental expression pattern of the vitellogenin receptor from the cockroach, Leucophaea maderae. Ins Biochem Molec Biol 37:235–245. https://doi.org/10.1016/j.ibmb.2006.11.007
Upadhyay SK, Singh H, Dixit S, Mendu V, Verma PC (2016) Molecular characterization of vitellogenin and vitellogenin receptor of Bemisia tabaci. PLoS One 11(5):e0155306. https://doi.org/10.1371/journal.pone.0155306
Zhang W, Ma L, Xiao H, Xie B, Smagghe G, Guo Y, Liang G (2016) Molecular characterization and function analysis of the vitellogenin receptor from the cotton bollworm Helicoperva armigera (Hübner) (Lepidoptera, Nuctuidae). PLoS One 11(5):e0155785. https://doi.org/10.1371/journal.pone.0155785
Acknowledgements
We thank the Center of Microscopy and Microanalysis of the Federal University of Viçosa for providing the microscopes used in this study and HandJ Editing Services, Brazil, for the English revision.
Funding
This study was funded by Brazilian research agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico CNPq (grant number 3015165/2013-5), Coordenação de Aperfeiçoamento de Pessoal de Nível Supeiror CAPES and Fundação de Amparo à Pesquisa do Estado de Minas Gerais FAPEMIG (grant number APQ-00508-16).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Virginia Teles Dohanik declares that she has no conflict of interest. Wagner Gonzaga Gonçalves declares that she has no conflict of interest. Leandro Licursi Oliveira declares that he has no conflict of interest. José Cola Zanuncio declares that he has no conflict of interest. José Eduardo Serrão declares that he has no conflict of interest.
Human and animal rights and informed consent
All procedures performed in studies involving animals were in accordance with ethical standards of the institution or practice at which the studies were conducted.
Additional information
Handling Editor: Jan Raoul De Mey
Electronic supplementary material
Suppl. Figure 1
SDS-PAGE and Western blots of vitellogenin receptor protein [1] VgR with 39 kDa (white arrow) purified using a nickel column (SDS-PAGE). [2] Western blot with a positive reaction for anti-VgR antibody (black arrow). (GIF 28 kb)
Suppl. Figure 2
Negative control of immunofluorescence for antibodies anti-VgR and anti-clathrin in the vitellogenic follicle of Apis mellifera. N - nucleus, FC - follicular cells. (GIF 23 kb)
Suppl. Figure 3
Negative control of immunogold for antibodies anti-VgR and anti-clathrin in the follicular cells of Polistes simillimus. AR - Apical region, OO – Oocyte, * - perivitelline space. (GIF 218 kb)
Rights and permissions
About this article
Cite this article
Dohanik, V.T., Gonçalves, W.G., Oliveira, L.L. et al. Vitellogenin transcytosis in follicular cells of the honeybee Apis mellifera and the wasp Polistes simillimus. Protoplasma 255, 1703–1712 (2018). https://doi.org/10.1007/s00709-018-1260-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1260-y