Abstract
Background
The use of five-aminolevulinic acid (5-ALA) in the staining of malignant glioma cells has significantly improved intraoperative radicality in the resection of gliomas in the last decade. Currently, there is no comparable selective fluorescent substance available for meningiomas. There is however a demand for intraoperative fluorescent identification of, e.g., invasive skull base meningiomas to help improve safe radical resection. Meningiomas show high expression of the somatostatin receptor type 2, offering the possibility of receptor-targeted imaging. The authors used a somatostatin receptor-labeled fluorescence dye in the identification of meningiomas in vitro. The aim of this study was to evaluate the possibility of selective identification of meningioma cells with fluorescent techniques.
Methods
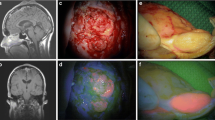
Twenty-four primary human meningioma cell cultures were analyzed. The tumor cells were incubated with FAM-TOC (5,6-Carboxyfluoresceine-Tyr3-Octreotide). As a negative control, four human dura tissues were cultured as well as a mixed cell culture in vitro and incubated with the same somatostatin receptor-labeled fluorescence substance. After incubation, fluorescence signal and intensity in all cell cultures were analyzed at three different time points using a fluorescence microscope with 488 nm epi-illumination.
Results
Sixteen WHO I, six WHO II, two WHO III meningioma primary cell cultures, and four dura cell cultures were analyzed. Fluorescence was detected in all meningioma cell cultures (22 cell culture stained strongly, 2 cell cultures moderately) directly after incubation up until 4 h later. There were no differences in the quality and quantity of fluorescence signal between the various meningioma grades. The fluorescence signal persisted unchanged during the analyzed period. In the negative control, dura cell cultures remained unstained.
Conclusions
This study demonstrates the use of FAM-TOC in the selective fluorescent identification of meningioma cells in vitro. Further evaluation of the chemical kinetics of the applied somatostatin receptor ligand and fluorescence dye is warranted. As a next step, an experimental animal model is needed to evaluate these promising results in vivo.



Similar content being viewed by others
Abbreviations
- 5-ALA:
-
Five-aminolevulinic acid
- CO2 :
-
Carbon dioxide
- DAPI:
-
4′,6-diamidin-2-phenylindol
- DMEM:
-
Dulbecco’s modified Eagle medium
- FAM-TOC:
-
6-Carboxyfluorescein-Tyrosin3-Octreotide
- FCS:
-
Fetal calf serum
- HE:
-
Hematoxylin and eosin
- Na-Fl:
-
Sodium fluorescein
- PBS:
-
Phosphate-buffered saline
- PFA:
-
Paraformaldehyde
- ROI:
-
Region of interest
- SPECT:
-
Single-photon emission computed tomography
- SSTR:
-
Somatostatin receptor
- WHO:
-
World Health Organization
References
Akcakaya MO, Goker B, Kasimcan MO, Hamamcioglu MK, Kiris T (2017) Use of sodium fluorescein in meningioma surgery performed under the YELLOW-560 nm surgical microscope filter: feasibility and preliminary results. World Neurosurg 107:966–973
Bedwell J, MacRobert AJ, Phillips D, Bown SG (1992) Fluorescence distribution and photodynamic effect of ALA-induced PP IX in the DMH rat colonic tumour model. Br J Cancer 65:818–824
Beks JW, de Windt HL (1988) The recurrence of supratentorial meningiomas after surgery. Acta Neurochir 95:3–5
Borovich B, Doron Y (1986) Recurrence of intracranial meningiomas: the role played by regional multicentricity. J Neurosurg 64:58–63
Borovich B, Doron Y, Braun J, Guilburd JN, Zaaroor M, Goldsher D, Lemberger A, Gruszkiewicz J, Feinsod M (1986) Recurrence of intracranial meningiomas: the role played by regional multicentricity. Part 2: clinical and radiological aspects. J Neurosurg 65:168–171
da Silva CE, Peixoto de Freitas PE (2016) Recurrence of skull base meningiomas: the role of aggressive removal in surgical treatment. J Neurol Surg B Skull Base 77:219–225
da Silva CE, da Silva JL, da Silva VD (2010) Use of sodium fluorescein in skull base tumors. Surg Neurol Int 1:70
Ding D, Starke RM, Hantzmon J, Yen CP, Williams BJ, Sheehan JP (2013) The role of radiosurgery in the management of WHO Grade II and III intracranial meningiomas. Neurosurg Focus 35:E16
Graillon T, Romano D, Defilles C, Saveanu A, Mohamed A, Figarella-Branger D, Roche PH, Fuentes S, Chinot O, Dufour H, Barlier A (2017) Octreotide therapy in meningiomas: in vitro study, clinical correlation, and literature review. J Neurosurg 127:660–669
Hellwig D, Samnick S, Reif J, Romeike BF, Reith W, Moringlane JR, Kirsch CM (2002) Comparison of tc-99m depreotide and in-111 octreotide in recurrent meningioma. Clin Nucl Med 27:781–784
Henn W, Cremerius U, Heide G, Lippitz B, Schroder JM, Gilsbach JM, Bull U, Zang KD (1995) Monosomy 1p is correlated with enhanced in vivo glucose metabolism in meningiomas. Cancer Genet Cytogenet 79:144–148
Kamp MA, Fischer I, Buhner J, Turowski B, Cornelius JF, Steiger HJ, Rapp M, Slotty PJ, Sabel M (2016) 5-ALA fluorescence of cerebral metastases and its impact for the local-in-brain progression. Oncotarget 7:66776–66789
Kawahara Y, Niiro M, Yokoyama S, Kuratsu J (2001) Dural congestion accompanying meningioma invasion into vessels: the dural tail sign. Neuroradiology 43:462–465
Ketter R, Kim YJ, Storck S, Rahnenfuhrer J, Romeike BF, Steudel WI, Zang KD, Henn W (2007) Hyperdiploidy defines a distinct cytogenetic entity of meningiomas. J Neuro-Oncol 83:213–221
Ketter R, Urbschat S, Henn W, Feiden W, Beerenwinkel N, Lengauer T, Steudel WI, Zang KD, Rahnenfuhrer J (2007) Application of oncogenetic trees mixtures as a biostatistical model of the clonal cytogenetic evolution of meningiomas. Int J Cancer 121:1473–1480
Ketter R, Rahnenfuhrer J, Henn W, Kim YJ, Feiden W, Steudel WI, Zang KD, Urbschat S (2008) Correspondence of tumor localization with tumor recurrence and cytogenetic progression in meningiomas. Neurosurgery 62:61–69 discussion 69-70
Knipps J, Beseoglu K, Kamp M, Fischer I, Felsberg J, Neumann LM, Steiger HJ, Cornelius JF (2017) Fluorescence behavior and dural infiltration of meningioma analyzed by 5-aminolevulinic acid-based fluorescence: operating microscope versus mini-spectrometer. World Neurosurg 108:118–127
Linsler S, Kraemer D, Driess C, Oertel J, Kammers K, Rahnenfuhrer J, Ketter R, Urbschat S (2014) Molecular biological determinations of meningioma progression and recurrence. PLoS One 9:e94987
Linsler S, Keller C, Urbschat S, Ketter R, Oertel J (2016) Prognosis of meningiomas in the early 1970s and today. Clin Neurol Neurosurg 149:98–103
Linsler S, Fischer G, Skliarenko V, Stadie A, Oertel J (2017) Endoscopic assisted supraorbital keyhole approach or endoscopic endonasal approach in case of tuberculum sellae meningioma: which surgical route should be favoured? World Neurosurg 104:601–611
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820
Millesi M, Kiesel B, Mischkulnig M, Martinez-Moreno M, Wohrer A, Wolfsberger S, Knosp E, Widhalm G (2016) Analysis of the surgical benefits of 5-ALA-induced fluorescence in intracranial meningiomas: experience in 204 meningiomas. J Neurosurg 125:1408–1419
Morofuji Y, Matsuo T, Hayashi Y, Suyama K, Nagata I (2008) Usefulness of intraoperative photodynamic diagnosis using 5-aminolevulinic acid for meningiomas with cranial invasion: technical case report. Neurosurgery 62:102–103 discussion 103-104
Rokni-Yazdi H, Azmoudeh Ardalan F, Asadzandi Z, Sotoudeh H, Shakiba M, Adibi A, Ayatollahi H, Rahmani M (2009) Pathologic significance of the “dural tail sign”. Eur J Radiol 70:10–16
Scheichel F, Ungersboeck K, Kitzwoegerer M, Marhold F (2017) Fluorescence-guided resection of extracranial soft tissue tumour infiltration in atypical meningioma. Acta Neurochir 159:1027–1031
Schick B, Wolf G, Romeike BF, Mestres P, Praetorius M, Plinkert PK (2003) Dural cell culture. A new approach to study duraplasty. Cells Tissues Organs 173:129–137
Silva CB, Ongaratti BR, Trott G, Haag T, Ferreira NP, Leaes CG, Pereira-Lima JF, Oliveira Mda C (2015) Expression of somatostatin receptors (SSTR1-SSTR5) in meningiomas and its clinicopathological significance. Int J Clin Exp Pathol 8:13185–13192
Simpson D (1957) The recurrence of intracranial meningiomas after surgical treatment. J Neurol Neurosurg Psychiatry 20:22–39
Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93:1003–1013
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ, Group AL-GS (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7:392–401
Valery CA, Faillot M, Lamproglou I, Golmard JL, Jenny C, Peyre M, Mokhtari K, Mazeron JJ, Cornu P, Kalamarides M (2016) Grade II meningiomas and Gamma Knife radiosurgery: analysis of success and failure to improve treatment paradigm. J Neurosurg 125:89–96
Wohrer A, Waldhor T, Heinzl H, Hackl M, Feichtinger J, Gruber-Mosenbacher U, Kiefer A, Maier H, Motz R, Reiner-Concin A, Richling B, Idriceanu C, Scarpatetti M, Sedivy R, Bankl HC, Stiglbauer W, Preusser M, Rossler K, Hainfellner JA (2009) The Austrian Brain Tumour Registry: a cooperative way to establish a population-based brain tumour registry. J Neuro-Oncol 95:401–411
Acknowledgements
The authors thank Mrs. Sigrid Welsch for technical assistance during cell culture and fluorescence labeling of the cell cultures, and Mr. David Breuskin and Mr. Sam Orie for proofreading of the manuscript.
Funding
The Saarland University provided financial support in the form of the grant HOMFOR 2016-201000790. The sponsor had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Comments
Very interesting if this flourescence could be of clinical use in the future. These results are very early but if proven specific enough with respect to per example invasion of dura there is a big potential.
Jane Skjoth-Rasmussen
Copenhagen, Denmark
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Tumor - Meningioma
Rights and permissions
About this article
Cite this article
Linsler, S., Ketter, R., Oertel, J. et al. Fluorescence imaging of meningioma cells with somatostatin receptor ligands: an in vitro study. Acta Neurochir 161, 1017–1024 (2019). https://doi.org/10.1007/s00701-019-03872-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-019-03872-x