Abstract
Background
The technique of 5-aminolevulinic acid (5-ALA) tumor fluorescence is increasingly used to improve visualization of tumor tissue and thereby to increase the rate of patients with gross total resections. In this study, we measured the resection volumes in patients who underwent 5-ALA-guided surgery for non-eloquent glioblastoma and compared them with the preoperative tumor volume.
Methods
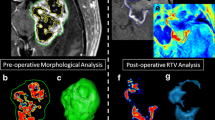
We selected 13 patients who had received a complete resection according to intraoperative 5-ALA induced fluorescence and CRET according to post-operative T1 contrast-enhanced MRI. The volumes of pre-operative contrast enhancing tissue, post-operative resection cavity and resected tissue were determined through shift-corrected volumetric analysis.
Results
The mean resection cavity (29 cm3) was marginally smaller than the pre-operative contrast-enhancing tumor (39 cm3, p = 0.32). However, the mean overall resection volume (84 cm3) was significantly larger than the pre-operative contrast-enhancing tumor (39 cm3, p = 0.0087). This yields a mean volume of resected 5-ALA positive, but radiological non-enhancing tissue of 45 cm3. The mean calculated rim of resected tissue surpassed pre-operative tumor diameter by 6 mm (range 0–10 mm).
Conclusions
Results of the current study imply that (i) the resection cavity underestimates the volume of resected tissue and (ii) 5-ALA complete resections go significantly beyond the volume of pre-operative contrast-enhancing tumor bulk on MRI, indicating that 5-ALA also stains MRI non-enhancing tumor tissue. Use of 5-ALA may thus enable extension of coalescent tumor resection beyond radiologically evident tumor. The impact of this more extended resection method on time to progression and overall survival has not been determined, and potentially puts adjacent and functionally intact tissue at risk.



Similar content being viewed by others
References
Aldave G, Tejada S, Pay E, Marigil M, Bejarano B, Idoate MA, Diez-Valle R (2013) Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic Acid-guided surgery. Neurosurgery 72:915–920, discussion 920–921
Berger MS (2011) Glioma surgery: a century of challenge. Clin Neursurg 58:7–9
Collaud S, Juzeniene A, Moan J, Lange N (2004) On the selectivity of 5-aminolevulinic acid-induced protoporphyrin IX formation. Current medicinal chemistry. Curr Med Chem Anticancer Agents 4:301–316
Konukoglu E, Clatz O, Bondiau PY, Delingette H, Ayache N (2010) Extrapolating glioma invasion margin in brain magnetic resonance images: suggesting new irradiation margins. Med Image Anal 14:111–125
Ku HH (1969) Notes on the use of propagation of error formulas. J Res NBS C Eng Inst 70C:263–273
Kuhnt D, Becker A, Ganslandt O, Bauer M, Buchfelder M, Nimsky C (2011) Correlation of the extent of tumor volume resection and patient survival in surgery of glioblastoma multiforme with high-field intraoperative MRI guidance. Neuro-oncology 13:1339–1348
Scherer HJ (1940) The forms of growth in gliomas and their practical significance. Brain 63:1–35
Schucht P, Beck J, Abu-Isa J, Andereggen L, Murek M, Seidel K, Stieglitz L, Raabe A (2012) Gross total resection rates in contemporary glioblastoma surgery: results of an institutional protocol combining 5-aminolevulinic acid intraoperative fluorescence imaging and brain mapping. Neurosurgery 71:927–935, discussion 935–936
Seidel K, Beck J, Stieglitz L, Schucht P, Raabe A (2013) The warning-sign hierarchy between quantitative subcortical motor mapping and continuous motor evoked potential monitoring during resection of supratentorial brain tumors. J Neurosurg 118:287–296
Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93:1003–1013
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ, Group AL-GS (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7:392–401
Stummer W, Reulen HJ, Meinel T, Pichlmeier U, Schumacher W, Tonn JC, Rohde V, Oppel F, Turowski B, Woiciechowsky C, Franz K, Pietsch T, Group AL-GS (2008) Extent of resection and survival in glioblastoma multiforme: identification of and adjustment for bias. Neurosurgery 62:564–576
Stummer W, Tonn JC, Mehdorn HM, Nestler U, Franz K, Goetz C, Bink A, Pichlmeier U, Group AL-GS (2011) Counterbalancing risks and gains from extended resections in malignant glioma surgery: a supplemental analysis from the randomized 5-aminolevulinic acid glioma resection study. Clinical article. J Neurosurg 114:613–623
Van Leemput KM, Maes F, Vandermeulen D, Suetens P (1999) Automated model-based tissue classification of MR images of the brain. IEEE Trans Med Imaging 18:897–908
Conflicts of interest
The authors report no conflicts of interest concerning the materials or methods used or the findings specified in this paper. No specific funding was provided for this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
This paper by Schucht et al. addresses an important aspect of fluorescence-guided resections, which is that the volume of ALA-derived tumor fluorescence during surgery of malignant gliomas markedly surpasses the volume of Gd-enhancement. The volume of Gd-enhancing tissue has always been the traditional target tissue in surgery for these diffusely infiltrating lesions and complete resection of Gd-enhancing tumor is one of the strongest prognostic indicators. Schucht and co-workers quantified the volume of fluorescing tissue by subtracting brain volume after surgery from brain volume prior to surgery, and correcting for CSF spaces. Nevertheless, this elegant approach has methodological limitations, since changes in brain blood volume or edema resolutions after resection of the tumor mass were not quantified and possibly cannot be quantified. Still, the approximation is probably quite good.
This paper complements recent data from Pamplona (1), in which Aldave and co-workers demonstrate that patients have better survival if fluorescing tissue is removed completely rather than leaving fluorescing tissue behind, even if residual tumor does not show up as GD-enhancement on the MRI. With this experience in mind, one might consider redefining the aim of surgery for malignant gliomas to extend resections to include all fluorescing tissue. Of course, maintaining functionality in patients is of utmost importance, and neurophysiological monitoring and mapping techniques have become indispensible adjuncts in the surgical therapy of patients with malignant gliomas when using the potential afforded by ALA-induced tumor fluorescence.
Walter Stummer
Münster, Germany
Reference:
(1) Aldave G, Tejada S, Pay E, Marigil M, Bejarano B, Idoate MA, Díez-Valle R (2013) Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic Acid-guided surgery. Neurosurgery. 72(6):915–20
Rights and permissions
About this article
Cite this article
Schucht, P., Knittel, S., Slotboom, J. et al. 5-ALA complete resections go beyond MR contrast enhancement: shift corrected volumetric analysis of the extent of resection in surgery for glioblastoma. Acta Neurochir 156, 305–312 (2014). https://doi.org/10.1007/s00701-013-1906-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-013-1906-7