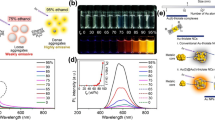
Abstract
Based on the resonance energy transfer between CdS@CeO2 and Cu2O@PdAg, a quenching immunosensor for sensitive detection of prostate specific antigen (PSA) was constructed. The CdS@CeO2 heterostructure was obtained by in situ growth of CeO2 particles on the surface of CdS nanorods, and stable cathodic ECL emission was achieved using K2S2O8 as coreactant. Cu2O@PdAg was composed of Cu2O with tetradecahedral structure and bimetallic PdAg nanospheres and has a UV–V is absorption range between 600 and 800 nm. It overlaps with the ECL emission spectrum of CdS@CeO2, realizing the effective quenching of the ECL signal, which provides feasibility for subsequent practical application. The immunosensor exhibited good linearity in the concentration range 10 fg·mL−1 ~ 100 ng·mL−1, with a detection limit of 5.6 fg·mL−1. In sample analysis, the recoveries were 99.8–101%, and the relative standard deviation (RSD) was 0.85–1.6% showing great potential and development value for the sensitive detection of prostate cancer.
Graphical Abstract







Similar content being viewed by others
References
Numan A, Singh S, Zhan Y, Li L, Khalid M, Rilla K, Ranjan S, Cinti S (2022) Advanced nanoengineered-customized point-of-care tools for prostate-specific antigen. Mikrochim Acta 189:27. https://doi.org/10.1007/s00604-021-05127-y
Usman A (2022) Nanoparticle enhanced optical biosensing technologies for prostate specific antigen biomarker detection. IEEE Rev Biomed Eng 15:122–137. https://doi.org/10.1109/Rbme.2020.3035273
Merriel SWD, Pocock L, Gilbert E, Creavin S, Walter FM, Spencer A, Hamilton W (2022) Systematic review and meta-analysis of the diagnostic accuracy of prostate-specific antigen (PSA) for the detection of prostate cancer in symptomatic patients. BMC Med 20:54. https://doi.org/10.1186/s12916-021-02230-y
Liu PK, Meng H, Zhang G, Song L, Han Q, Wang C, Fu YZ (2021) Ultrasensitive dual-quenching electrochemiluminescence immunosensor for prostate specific antigen detection based on graphitic carbon nitride quantum dots as an emitter. Mikrochim Acta 188:350. https://doi.org/10.1007/s00604-021-05015-5
Balk SP, Ko YJ, Bubley GJ (2003) Biology of prostate-specific antigen. J Clin Oncol 21:383–391. https://doi.org/10.1200/JCO.2003.02.083
Song L, Wu JL, Zhang G, Liu PK, Kuang GR, Fu YZ (2021) Novel gold nanoparticles functionalized Mo-polydopamine hollow sphere as an efficient quencher in conjugated microporous polymer electrochemiluminescent system. Sens Actuators B Chem 344:130130. https://doi.org/10.1016/j.snb.2021.130130
Ahmed HA, Azzazy HME (2013) Power-free chip enzyme immunoassay for detection of prostate specific antigen (PSA) in serum. Biosens Bioelectron 49:478–484. https://doi.org/10.1016/j.bios.2013.05.058
Zhang LJ, Luo ZB, Zeng RJ, Zhou Q, Tang DP (2019) All-solid-state metal-mediated Z-scheme photoelectrochemical immunoassay with enhanced photoexcited charge-separation for monitoring of prostate-specific antigen. Biosens Bioelectron 134:1–7. https://doi.org/10.1016/j.bios.2019.03.052
Zhao YT, Gao W, Ge XX, Li SQ, Du D, Yang HP (2019) CdTe@SiO2 signal reporters-based fluorescent immunosensor for quantitative detection of prostate specific antigen. Anal Chim Acta 1057:44–50. https://doi.org/10.1016/j.aca.2019.01.019
Zhou ZM, Feng Z, Zhou J, Fang BY, Qi XX, Ma ZY, Liu B, Zhao YD, Hu XB (2015) Capillary electrophoresis-chemiluminescence detection for carcino-embryonic antigen based on aptamer/graphene oxide structure. Biosens Bioelectron 64:493–498. https://doi.org/10.1016/j.bios.2014.09.050
Feng SY, Zheng ZC, Xu YJ, Lin JY, Chen GN, Weng CC, Lin D, Qiu SF, Cheng M, Huang ZF, Wang L, Chen R, Xie SS, Zeng HS (2017) A noninvasive cancer detection strategy based on gold nanoparticle surface-enhanced Raman spectroscopy of urinary modified nucleosides isolated by affinity chromatography. Biosens Bioelectron 91:616–622. https://doi.org/10.1016/j.bios.2017.01.006
Dai L, Li YY, Wang YG, Luo XL, Wei D, Feng R, Yan T, Ren X, Du B, Wei Q (2019) A prostate-specific antigen electrochemical immunosensor based on Pd NPs functionalized electroactive Co-MOF signal amplification strategy. Biosens Bioelectron 132:97–104. https://doi.org/10.1016/j.bios.2019.02.055
Chen MM, Xu CH, Zhao W, Chen HY, Xu JJ (2022) Single cell imaging of electrochemiluminescence-driven photodynamic therapy. Angew Chem Int Edit 61:e202117401. https://doi.org/10.1002/anie.202117401
Xiao SY, Wang XY, Yang CP, Jiang YJ, Zhen SJ, Huang CZ, Li YF (2022) Electrochemiluminescence resonance energy transfer system based on silver metal–organic frameworks as a double-amplified emitter for sensitive detection of miRNA-107. Anal Chem 94:1178–1186. https://doi.org/10.1021/acs.analchem.1c04368
Ma XM, Kang Q, Li MM, Fu L, Zou GZ, Shen DZ (2022) Sensitive, signal-modulation strategy for discrimination of ECL spectra and investigation of mutual interactions of emitters. Anal Chem 94:3637–3644. https://doi.org/10.1021/acs.analchem.1c05217
Tu TT, Sun Y, Lei YM, Chai YQ, Zhuo Y, Yuan R (2022) Pyrenecarboxaldehyde encapsulated porous TiO2 nanoreactors for monitoring cellular GSH levels. Nanoscale 14:5751–5757. https://doi.org/10.1039/d2nr00784c
Zhang JL, Yao LY, Yang Y, Liang WB, Yuan R, Xiao DR (2022) Conductive covalent organic frameworks with conductivity- and pre-reduction-enhanced electrochemiluminescence for ultrasensitive biosensor construction. Anal Chem 94:3685–3692. https://doi.org/10.1021/acs.analchem.1c05436
Lu HJ, Xu JJ, Zhou H, Chen HY (2020) Recent advances in electrochemiluminescence resonance energy transfer for bioanalysis: fundamentals and applications. Trends Anal Chem 122:115746. https://doi.org/10.1016/j.trac.2019.115746
Chen C, Hildebrandt N (2020) Resonance energy transfer to gold nanoparticles: NSET defeats FRET. Trends Anal Chem 123:115748. https://doi.org/10.1016/j.trac.2019.115748
Jeong D, Jo W, Jeong J, Kim T, Han S, Son MK, Jung H (2022) Characterization of Cu2O/CuO heterostructure photocathode by tailoring CuO thickness for photoelectrochemical water splitting. RSC Adv 12:2632–2640. https://doi.org/10.1039/d1ra08863g
Sundarapandi M, Shanmugam S, Ramaraj R (2022) Tuning Cu2O shell on gold nanocube core employing amine-functionalized silane for electrocatalytic nitrite detection. ACS Appl Nano Mater 5:1674–1682. https://doi.org/10.1021/acsanm.1c04459
Xing B, Zhang T, Han Q, Wei Q, Wu D (2019) Electrochemiluminescent immunoassay for insulin by using a quencher pair consisting of CdS: Eu nanoclusters loaded with multiwalled carbon nanotubes on reduced graphene oxide nanoribbons and gold nanoparticle-loaded octahedral Cu2O. Microchim Acta 186:500. https://doi.org/10.1007/s00604-019-3640-1
Oh JW, Takaloo AV, Baek SD, Myoung JM (2022) Efficient electrochemiluminescence devices using Pd nanoparticle-anchored TiO2 nanorod electrodes with high catalytic activity. Appl Surf Sci 586:152864. https://doi.org/10.1016/j.apsusc.2022.152864
Liu SH, Jia Y, Xue JW, Li YW, Wu ZL, Ren X, Ma H, Li Y, Wei Q (2020) Bifunctional peptide-biomineralized gold nanoclusters as electrochemiluminescence probe for optimizing sensing interface. Sens Actuators B Chem 318:128278. https://doi.org/10.1016/j.snb.2020.128278
Yang L, Fan DW, Zhang Y, Ding CF, Wu D, Wei Q, Ju HX (2019) Ferritin-based electrochemiluminescence nanosurface energy transfer system for procalcitonin detection using HWRGWVC heptapeptide for site-oriented antibody immobilization. Anal Chem 91:7145–7152. https://doi.org/10.1021/acs.analchem.9b00325
Song XZ, Zhao L, Luo CN, Ren X, Yang L, Wei Q (2021) Peptide-based biosensor with a luminescent copper-based metal–organic framework as an electrochemiluminescence emitter for trypsin assay. Anal Chem 93:9704–9710. https://doi.org/10.1021/acs.analchem.1c00850
Yang L, Jia Y, Wu D, Zhang Y, Ju HX, Du Y, Ma HM, Wei Q (2019) Synthesis and application of CeO2/SnS2 heterostructures as a highly efficient coreaction accelerator in the luminol–dissolved O2 system for ultrasensitive biomarkers immunoassay. Anal Chem 91:14066–14073. https://doi.org/10.1021/acs.analchem.9b03796
Zhao JE, Wang SP, Zhang SB, Zhao PN, Wang JR, Yan M, Ge SG, Yu JH (2020) Peptide cleavage-mediated photoelectrochemical signal on-off via CuS electronic extinguisher for PSA detection. Biosens Bioelectron 150:111958. https://doi.org/10.1016/j.bios.2019.111958
Liang H, Xu HB, Zhao YT, Zheng J, Zhao H, Li GL, Li CP (2019) Ultrasensitive electrochemical sensor for prostate specific antigen detection with a phosphorene platform and magnetic covalent organic framework signal amplifier. Biosens Bioelectron 144:111691. https://doi.org/10.1016/j.bios.2019.111691
Xu DD, Deng YL, Li CY, Lin Y, Tang HW (2017) Metal-enhanced fluorescent dye-doped silica nanoparticles and magnetic separation: a sensitive platform for one-step fluorescence detection of prostate specific antigen. Biosens Bioelectron 87:881–887. https://doi.org/10.1016/j.bios.2016.09.034
Fu L, Fu KN, Gao XW, Dong ST, Zhang B, Fu SJ, Hsu H-Y, Zou GZ (2021) Enhanced near-infrared electrochemiluminescence from trinary Ag−In−S to multinary Ag−Ga−In−S nanocrystals via doping-in-growth and its immunosensing applications. Anal Chem 93:2160–2165. https://doi.org/10.1021/acs.analchem.0c03975
Yang XL, We YX, Wang ZM, Wang J, Qi HL, Gao Q, Zhang CX (2022) Highly efficient electrogenerated chemiluminescence quenching on lipid-coated multifunctional magnetic nanoparticles for the determination of proteases. Anal Chem 94:2305–2312. https://doi.org/10.1021/acs.analchem.1c05033
Funding
This research was financially supported by the Innovation Team Project of Colleges and Universities in Jinan (No. 2019GXRC027), the National Natural Science Foundation of China (Nos. 22274063, 51904114, 21777056, 21675063), and the Special Foundation for Taishan Scholar Professorship of Shandong Province.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, Z., Shao, X., Luo, J. et al. Cu2O@PdAg-quenched CdS@CeO2 heterostructure electrochemiluminescence immunosensor for determination of prostate-specific antigen. Microchim Acta 190, 59 (2023). https://doi.org/10.1007/s00604-023-05635-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-05635-z