Abstract
The authors describe a method for solvent-free mechano-chemical synthesis of a bioinspired sorbent. A 2D ultra-thin carbon sheet similar to graphene oxide was prepared using a natural waste (onion sheet). The formation of 2D carbon sheets was confirmed by Raman spectroscopy, X-ray photoelectron spectroscopy and ATR-IR. The surface morphology was characterized by field emission scanning electron microscopy and high-resolution tunneling electron microscopy. The carbon sheets were decorated with crystalline MnFe2O4 nanoparticles by solid-state reaction at room temperature. The presence of magnetic particles in the final product was confirmed by vibrating sample magnetometry and electron microscopy. The synergistic effect of carbon sheets and MnFe2O4 led to an enhanced sorption of arsenic species compared to bare carbon sheets or to MnFe2O4 nanoparticles. A column was prepared for the simultaneous preconcentration and determination of trace levels of As(III) and As(V) from water samples. The preconcentration factors are between 900 and 833 for As(III) and As(V) species, respectively. The linearity of the calibration plot ranges from 0.4–10 ng mL−1. The detection limits (at 3σ) for both As(III) and As(V) are 30 pg mL−1. The Student’s t values for the analysis of spiked samples are lower than the critical Student’s t values at a 95% confidence level. The recoveries from spiked water samples range between 99 and 102.8%.

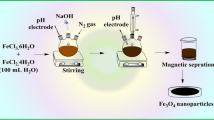
Schematic representation of the preparation of carbon sheets similar to graphene oxide from onion sheaths after pyrolysis at 800 °C. The prepared carbon sheet-MnFe2O4 composite shows excellent arsenic sorption and preconcentration down to the pg mL−1 concentration.







Similar content being viewed by others
References
Zhang J, Chen Y, Zhao W, Li Y (2018) Arsenic removal from aqueous solutions by diethylenetriamine-functionalized resin: isotherm, kinetics, selectivity and mechanism. R Soc Open Sci 5(9):181013. https://doi.org/10.1098/rsos.181013
Wei Z, Wang Z, Yan J, Liu Y, Wu Y, Fang Y, Yu L, Cheng G, Pan Z, Hu G (2019) Adsorption and oxidation of arsenic by two kinds of beta-MnO2. J Hazard Mater 373:232–242. https://doi.org/10.1016/j.jhazmat.2019.03.071
Kalaruban M, Loganathan P, Nguyen TV, Nur T, Hasan Johir MA, Nguyen TH, Trinh MV, Vigneswaran S (2019) Iron-impregnated granular activated carbon for arsenic removal: application to practical column filters. J Environ Manag 239:235–243. https://doi.org/10.1016/j.jenvman.2019.03.053
Paszkiewicz M, Caban M, Bielicka-Gieldon A, Stepnowski P (2017) Optimization of a procedure for the simultaneous extraction of polycyclic aromatic hydrocarbons and metal ions by functionalized and non-functionalized carbon nanotubes as effective sorbents. Talanta 165:405–411. https://doi.org/10.1016/j.talanta.2016.10.049
Liu Z, Wang H, Liu C, Jiang Y, Yu G, Mu X, Wang X (2012) Magnetic cellulose-chitosan hydrogels prepared from ionic liquids as reusable adsorbent for removal of heavy metal ions. Chem Commun (Camb) 48(59):7350–7352. https://doi.org/10.1039/c2cc17795a
Mututuvari TM, Tran CD (2014) Synergistic adsorption of heavy metal ions and organic pollutants by supramolecular polysaccharide composite materials from cellulose, chitosan and crown ether. J Hazard Mater 264:449–459. https://doi.org/10.1016/j.jhazmat.2013.11.007
Zhou Y, Fu S, Zhang L, Zhan H, Levit MV (2014) Use of carboxylated cellulose nanofibrils-filled magnetic chitosan hydrogel beads as adsorbents for Pb(II). Carbohydr Polym 101:75–82. https://doi.org/10.1016/j.carbpol.2013.08.055
Luo X, Zeng J, Liu S, Zhang L (2015) An effective and recyclable adsorbent for the removal of heavy metal ions from aqueous system: magnetic chitosan/cellulose microspheres. Bioresour Technol 194:403–406. https://doi.org/10.1016/j.biortech.2015.07.044
Vincent C, Barre Y, Vincent T, Taulemesse JM, Robitzer M, Guibal E (2015) Chitin-Prussian blue sponges for Cs(I) recovery: from synthesis to application in the treatment of accidental dumping of metal-bearing solutions. J Hazard Mater 287:171–179. https://doi.org/10.1016/j.jhazmat.2015.01.041
Wang M, Xu L, Peng J, Zhai M, Li J, Wei G (2009) Adsorption and desorption of Sr(II) ions in the gels based on polysaccharide derivates. J Hazard Mater 171(1–3):820–826. https://doi.org/10.1016/j.jhazmat.2009.06.071
Baranik A, Sitko R, Gagor A, Queralt I, Margui E, Zawisza B (2018) Graphene oxide decorated with cerium(IV) oxide in determination of Ultratrace metal ions and speciation of selenium. Anal Chem 90(6):4150–4159. https://doi.org/10.1021/acs.analchem.8b00137
Chen D, Zhang H, Yang K, Wang H (2016) Functionalization of 4-aminothiophenol and 3-aminopropyltriethoxysilane with graphene oxide for potential dye and copper removal. J Hazard Mater 310:179–187. https://doi.org/10.1016/j.jhazmat.2016.02.040
Chen M, Li Z, Li J, Li J, Li Q, Zhang L (2017) The extraction of uranium using graphene aerogel loading organic solution. Talanta 166:284–291. https://doi.org/10.1016/j.talanta.2017.01.070
Wei MP, Chai H, Cao YL, Jia DZ (2018) Sulfonated graphene oxide as an adsorbent for removal of Pb(2+) and methylene blue. J Colloid Interface Sci 524:297–305. https://doi.org/10.1016/j.jcis.2018.03.094
Yan H, Li H, Tao X, Li K, Yang H, Li A, Xiao S, Cheng R (2014) Rapid removal and separation of iron(II) and manganese(II) from micropolluted water using magnetic graphene oxide. ACS Appl Mater Interfaces 6(12):9871–9880. https://doi.org/10.1021/am502377n
Scida K, Stege PW, Haby G, Messina GA, Garcia CD (2011) Recent applications of carbon-based nanomaterials in analytical chemistry: critical review. Anal Chim Acta 691(1–2):6–17. https://doi.org/10.1016/j.aca.2011.02.025
Chua CK, Pumera M (2014) Chemical reduction of graphene oxide: a synthetic chemistry viewpoint. Chem Soc Rev 43(1):291–312. https://doi.org/10.1039/c3cs60303b
Malesevic A, Vitchev R, Schouteden K, Volodin A, Zhang L, Tendeloo GV, Vanhulsel A, Haesendonck CV (2008) Synthesis of few-layer graphene via microwave plasma-enhanced chemical vapour deposition. Nanotechnology 19(30):305604. https://doi.org/10.1088/0957-4484/19/30/305604
Dreyer DR, Park S, Bielawski CW, Ruoff RS (2010) The chemistry of graphene oxide. Chem Soc Rev 39(1):228–240. https://doi.org/10.1039/b917103g
Ruan G, Sun Z, Peng Z, Tour JM (2011) Growth of graphene from food, insects, and waste. ACS Nano 5(9):7601–7607. https://doi.org/10.1021/nn202625c
Panmand RP, Patil P, Sethi Y, Kadam SR, Kulkarni MV, Gosavi SW, Munirathnam NR, Kale BB (2017) Unique perforated graphene derived from Bougainvillea flowers for high-power supercapacitors: a green approach. Nanoscale 9(14):4801–4809. https://doi.org/10.1039/c7nr00583k
Benitez V, Molla E, Martin-Cabrejas MA, Aguilera Y, Lopez-Andreu FJ, Cools K, Terry LA, Esteban RM (2011) Characterization of industrial onion wastes (Allium cepa L.): dietary fibre and bioactive compounds. Plant Foods Hum Nutr 66(1):48–57. https://doi.org/10.1007/s11130-011-0212-x
Choi IS, Cho EJ, Moon JH, Bae HJ (2015) Onion skin waste as a valorization resource for the by-products quercetin and biosugar. Food Chem 188:537–542. https://doi.org/10.1016/j.foodchem.2015.05.028
Budde H, Coca-Lopez N, Shi X, Ciesielski R, Lombardo A, Yoon D, Ferrari AC, Hartschuh A (2016) Raman radiation patterns of graphene. ACS Nano 10(2):1756–1763. https://doi.org/10.1021/acsnano.5b06631
Rabinovich D (1998) Descriptive inorganic chemistry (Rayner-Canham, Geoff). J Chem Educ 75(6):697. https://doi.org/10.1021/ed075p697
Boyacı E, Çağır A, Shahwan T, Eroğlu AE (2011) Synthesis, characterization and application of a novel mercapto- and amine-bifunctionalized silica for speciation/sorption of inorganic arsenic prior to inductively coupled plasma mass spectrometric determination. Talanta 85(3):1517–1525. https://doi.org/10.1016/j.talanta.2011.06.021
Hao J, Han M-J, Meng X (2009) Preparation and evaluation of thiol-functionalized activated alumina for arsenite removal from water. J Hazard Mater 167(1):1215–1221. https://doi.org/10.1016/j.jhazmat.2009.01.124
Chen M-L, Sun Y, Huo C-B, Liu C, Wang J-H (2015) Akaganeite decorated graphene oxide composite for arsenic adsorption/removal and its proconcentration at ultra-trace level. Chemosphere 130:52–58. https://doi.org/10.1016/j.chemosphere.2015.02.046
Barros H, Marcó Parra L-M, Bennun L, Greaves ED (2010) Determination of arsenic in water samples by Total reflection X-ray fluorescence using pre-concentration with alumina. Spectrochim Acta B At Spectrosc 65(6):489–492. https://doi.org/10.1016/j.sab.2010.04.004
Dados A, Kartsiouli E, Chatzimitakos T, Papastephanou C, Stalikas CD (2014) In situ trapping of As, Sb and Se hydrides on nanometer-sized ceria-coated iron oxide-silica and slurry suspension introduction to ICP-OES. Talanta 130:142–147. https://doi.org/10.1016/j.talanta.2014.06.046
Baranik A, Gagor A, Queralt I, Marguí E, Sitko R, Zawisza B (2018) Ceria nanoparticles deposited on graphene nanosheets for adsorption of copper(II) and lead(II) ions and of anionic species of arsenic and selenium. Mikrochim Acta 185(5):264–264. https://doi.org/10.1007/s00604-018-2806-6
Montoro Leal P, Vereda Alonso E, López Guerrero MM, Cordero MTS, Cano Pavón JM, García de Torres A (2018) Speciation analysis of inorganic arsenic by magnetic solid phase extraction on-line with inductively coupled mass spectrometry determination. Talanta 184:251–259. https://doi.org/10.1016/j.talanta.2018.03.019
Yang X-A, Shi M-T, Leng D, Zhang W-B (2018) Fabrication of a porous hydrangea-like Fe3O4@MnO2 composite for ultra-trace arsenic preconcentration and determination. Talanta 189:55–64. https://doi.org/10.1016/j.talanta.2018.06.065
dos Santos QO, Silva Junior MM, Lemos VA, Ferreira SLC, de Andrade JB (2018) An online preconcentration system for speciation analysis of arsenic in seawater by hydride generation flame atomic absorption spectrometry. Microchem J 143:175–180. https://doi.org/10.1016/j.microc.2018.08.004
Munonde TS, Maxakato NW, Nomngongo PN (2018) Preparation of magnetic Fe3O4 nanocomposites modified with MnO2, Al2O3, Au and their application for preconcentration of arsenic in river water samples. J Environ Chem Eng 6(2):1673–1681. https://doi.org/10.1016/j.jece.2018.02.017
Author information
Authors and Affiliations
Contributions
H.A and U.H designed the study, analysed the data and wrote the manuscript. K.U and M.S.A performed the experiments and provided the results. H.A, K.U and M.N.A.I discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 153 kb)
Rights and permissions
About this article
Cite this article
Ahmad, H., Haseen, U., Umar, K. et al. Bioinspired 2D carbon sheets decorated with MnFe2O4 nanoparticles for preconcentration of inorganic arsenic, and its determination by ICP-OES. Microchim Acta 186, 649 (2019). https://doi.org/10.1007/s00604-019-3753-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3753-6