Abstract
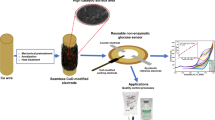
The authors describe a highly efficient photoelectrochemical (PEC) scheme for the determination of hydrogen peroxide (H2O2). BiVO4 microrods were hydrothermally synthesized and deposited on fluorine - doped tin oxide (FTO) glass which acts as the working electrode. Scanning electron microscopy, X-ray powder diffraction and Raman spectroscopy were utilized for the characterization of the microrods. On irradiation with visible light, the holes generated in the microrods are capturing electrons from H2O2 to produce a photocurrent at an operating potential of 0 V vs. Ag/AgCl. Under optimal conditions, the photocurrent increases with the concentration of H2O2 in the range from 50 μmol·L−1 to 1.5 mmol·L−1, and the limit of detection is 8.5 μmol·L−1 (at 3σ). A repeatability and intermediate precision of ≤6.6% was accomplished at H2O2 levels of 0.1, 0.5 and 1.0 mmol·L−1. The method was applied to the determination of H2O2 in spiked sterilized milk samples and gave satisfactory results. As the method works at zero potential, the photocurrent can be measured with simple instrumentation such as digital multimeters, and this will enable expensive electrochemical workstations to be replaced in future.

An enzyme-free photoelectrochemical sensing strategy is described for sensitive determination of hydrogen peroxide in foodstuff using fluorine-doped tin oxide electrode modified with BiVO4 microrods.




Similar content being viewed by others
References
Liu Y, Sun G, Jiang C, Zheng X, Zheng L, Li C (2014) Highly sensitive detection of hydrogen peroxide at a carbon nanotube fiber microelectrode coated with palladium nanoparticles. Microchim Acta 181:63–70
Winterbourn C (2008) Reconciling the chemistry and biology of reactive oxygen species. Nat Chem Biol 4:278–286
Bortolozzi R, von Gradowski S, Ihmels H, Schafer K, Viola G (2014) Selective ratiometric detection of H2O2 in water and in living cells with boronobenzob quinolizinium derivatives. Chem Commun 50:8242–8245
Wang T, Zhu H, Zhuo J, Zhu Z, Papakonstantinou P, Lubarsky G, Lin J, Li M (2013) Biosensor based on ultrasmall MoS2 nanoparticles for electrochemical detection of H2O2 released by cells at the nanomolar level. Anal Chem 85:10289–10295
Yu C, Wang Q, Qian D, Li W, Huang Y, Chen F, Bao N, Gu H (2016) An ITO electrode modified with electrodeposited graphene oxide and gold nanoclusters for detecting the release of H2O2 from bupivacaine-injured neuroblastoma cells. Microchim Acta 183:3167–3175
Tarvin M, McCord B, Mount K, Miller M (2011) Analysis of hydrogen peroxide field samples by HPLC/FD and HPLC/ED in DC mode. Forensic Sci Int 209:166–172
Wang Z, Song H, Zhao H, Lv Y (2013) Graphene-amplified electrogenerated chemiluminescence of CdTe quantum dots for H2O2 sensing. Luminescence 28:259–264
Zhuang J, Tang D, Lai W, Xu M, Tang D (2015) Target-induced nano-enzyme reactor mediated hole-trapping for high-throughput immunoassay based on a split-type photoelectrochemical detection strategy. Anal Chem 87:9473–9480
Zheng Y, Liang W, Yuan Y, Xiong C, Xie S, Wang H, Chai Y, Yuan R (2016) Wavelength-resolved simultaneous photoelectrochemical bifunctional sensor on single interface: a newly in vitro approach for multiplexed DNA monitoring in cancer cells. Biosens Bioelectron 81:423–430
Ge S, Li W, Yan M, Song X, Yu J (2015) Photoelectrochemical detection of tumor markers based on a CdS quantum dot/ZnO nanorod/Au@Pt-paper electrode 3D origami immunodevice. J Mater Chem B 3:2426–2432
Jin D, Xu Q, Yu L, Hu X (2015) Photoelectrochemical detection of the herbicide clethodim by using the modified metal-organic framework amino-MIL-125(Ti)/TiO2. Microchim Acta 182:1885–1892
Han Q, Wang K, Xu L, Yan X, Zhang K, Chen X, Wang Q, Zhang L, Pei R (2015) N-doped TiO2 based visible light activated label-free photoelectrochemical biosensor for detection of Hg2+ through quenching of photogenerated electrons. Analyst 140:4143–4147
Zhao W, Han Y, Zhu Y, Zhang N, Xu J, Chen H (2015) DNA labeling generates a unique amplification probe for sensitive photoelectrochemical immunoassay of HIV-1 p24 antigen. Anal Chem 87:5496–5499
Gong J, Fang T, Peng D, Li A, Zhang L (2015) A highly sensitive photoelectrochemical detection of perfluorooctanic acid with molecularly imprinted polymer-functionalized nanoarchitectured hybrid of AgI-BiOI composite. Biosens Bioelectron 73:256–263
Cao F, Xiong J, Wu F, Liu Q, Shi Z, Yu Y, Wang X, Li L (2016) Enhanced photoelectrochemical performance from rationally designed anatase/rutile TiO2 heterostructures. ACS Appl Mater Interface 8:12239–12245
Li H, Li J, Xu Q, Hu X (2011) Poly(3-hexylthiophene)/TiO2 nanoparticle-functionalized electrodes for visible light and low potential photoelectrochemical sensing of organophosphorus pesticide chlopyrifos. Anal Chem 83:9681–9686
Khan M, Khan M, Cho M (2016) CdS-graphene nanocomposite for efficient visible-light-driven photocatalytic and photoelectrochemical applications. J Colloid Interface Sci 482:221–232
Sahai S, Ikram A, Rai S, Dass S, Shrivastav R, Satsangi V (2014) CdSe quantum dots sensitized nanoporous hematite for photoelectrochemical generation of hydrogen. Int J Hydrogen Energ 39:11860–11866
Shao M, Ning F, Wei M, Evans D, Duan X (2014) Hierarchical nanowire arrays based on ZnO core-layered double hydroxide shell for largely enhanced photoelectrochemical water splitting. Adv Funct Mater 24:580–586
Qamar M, Drmosh Q, Ahmed M, Qamaruddin M, Yamani Z (2015) Enhanced photoelectrochemical and photocatalytic activity of WO3-surface modified TiO2 thin film. Nanoscale Res Lett 10:1–6
Li R, Han H, Zhang F, Wang D, Li C (2014) Highly efficient photocatalysts constructed by rational assembly of dual-cocatalysts separately on different facets of BiVO4. Energ Environ Sci 7:1369–1376
Zhao Z, Li Z, Zou Z (2011) Electronic structure and optical properties of monoclinic clinobisvanite BiVO4. Phys Chem Chem Phys 13:4746–4753
Ke D, Peng T, Ma L, Cai P, Jiang P (2008) Photocatalytic water splitting for O2 production under visible-light irradiation on BiVO4 nanoparticles in different sacrificial reagent solutions. Appl Catal A 350:111–117
Ribeiro F, Moraes F, Pereira E, Marken F, Mascaro L (2015) New application for the BiVO4 photoanode: a photoelectroanalytical sensor for nitrite. Electrochem Commun 61:1–4
Zhou L, Wang W, Zhang L, Xu H, Zhu W (2007) Single-crystalline BiVO4 microtubes with square cross-sections: microstructure, growth mechanism, and photocatalytic property. J Phys Chem C 111:13659–13664
Yu J, Zhang Y, Kudo A (2009) Synthesis and photocatalytic performances of BiVO4 by ammonia co-precipitation process. J Solid State Chem 182:223–228
Hernandez S, Gerardi G, Bejtka K, Fina A, Russo N (2016) Evaluation of the charge transfer kinetics of spin-coated BiVO4 thin films for sun-driven water photoelectrolysis. Appl Catal B 190:66–74
Tu W, Lei J, Wang P, Ju H (2011) Photoelectrochemistry of free-base-porphyrin-functionalized zinc oxide nanoparticles and their applications in biosensing. Eur Chem J 17:9440–9447
Lin Y, Chen X, Lin Y, Zhou Q, Tang D (2015) Non-enzymatic sensing of hydrogen peroxide using a glassy carbon electrode modified with a nanocomposite made from carbon nanotubes and molybdenum disulfide. Microchim Acta 182:1803–1809
Yang Y, Fu R, Yuang J, Wu S, Zhang J, Wang H (2015) Highly sensitive hydrogen peroxide sensor based on a glassy carbon electrode modified with platinum nanoparticles on carbon nanofiber heterostructures. Microchim Acta 182:2241–2249
Wang B, Ju P, Zhang D, Han X, Zheng L, Yin X, Sun C (2016) Colorimetric detection of H2O2 using flower-like Fe2(MoO4)3 microparticles as a peroxidase mimic. Microchim Acta Doi. doi:10.1007/s00604-016-1955-8
Jahanian M, Akbarinejad A, Alizadeh N (2017) Design of a sensing platform with dual performance for detection of hydrogen peroxide and Fe3+ based on a new fluorescent oligo N-phenylpyrrole derivative. Sens Actu B 240:971–978
Sharma V, Mobin S (2017) Cytocompatible peroxidase mimic CuO:graphene nanosphere composite as colorimetric dual sensor for hydrogen peroxide and cholesterol with its logic gate implementation. Sens Acut B 240:338–348
Liu Y, Liu X, Guo Z, Hu Z, Xue Z, Lu X (2017) Horseradish peroxidase supported on porous graphene as a novel sensing platform for detection of hydrogen peroxide in living cells sensitively. Biosens Bioelectron 87:101–107
Thanh T, Balamurugan J, Lee S, Kim N, Lee J (2016) Novel porous gold-palladium nanoalloy network- supported graphene as an advanced catalyst for non-enzymatic hydrogen peroxide sensing. Biosens Bioelectron 85:669–678
Boujakhrout A, Diez P, Sanchez A, Martinez-Ruiz P, Pingarron J, Villalonga J (2016) Gold nanoparticles- decorated silver-bipyridine nanobelts for the construction of mediatorless hydrogen peroxide biosensor. J Colloid Interf Sci 482:105–111
Fang X, Xu X, Hu X, Li Z, Wang G (2016) Native carbon nanodots as fluorescent probe for assays based on the use of glucose oxidase or horseradish peroxidase. Microchim Acta 183:2761–2770
Yao Z, Yang X, Wu F, Wu W, Wu F (2016) Synthesis of differently sized silver nanoparticles on a screen-printed electrode sensitized with a nanocomposites consisting of reduced graphene oxide and cerium (IV) oxide for nonenzymatic sensing of hydrogen peroxide. Microchim Acta 183:2799–2806
Wu Q, Sheng Q, Zheng J (2016) Nonenzymatic amperometric sensing of hydrogen peroxide using a glassy carbon electrode modified with a sandwich-structured nanocomposite consisting of silver nanoparticles. Microchim Acta 183:1943–1951
Yang Z, Qi C, Zheng X, Zheng J (2016) Sensing hydrogen peroxide with a glassy carbon electrode modified with silver nanoparticles, AlOOH and reduced graphene oxide. Microchim Acta 183:1131–1136
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21675029 and 21475025), the National Science Foundation of Fujian Province (2014 J07001), and the Program for Changjiang Scholars and Innovative Research Team in University (IRT15R11).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yu, Z., Lv, S., Ren, R. et al. Photoelectrochemical sensing of hydrogen peroxide at zero working potential using a fluorine-doped tin oxide electrode modified with BiVO4 microrods. Microchim Acta 184, 799–806 (2017). https://doi.org/10.1007/s00604-016-2071-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-016-2071-5