Abstract
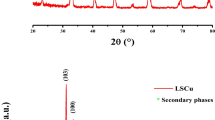
A new electrochemical sensor for H2O2 was constructed by depositing copper doped CuO nanosheets on a glassy carbon electrode (GCE). The morphology and composition of the modified electrode were characterized via scanning electron microscopy, energy dispersive X-ray spectroscopy and X-ray diffraction. The electrochemical properties of the electrode were studied using cyclic voltammetry and electrochemical impedance spectroscopy. The catalytic performance of the sensor was studied in 100 mM NaOH solution via differential pulse voltammetry and revealed the sensor to display significantly improved electrocatalytic activity with respect to the analysis of the H2O2 in comparison to a plain GCE or a GCE modified with copper only. The response to H2O2 at a working voltage of −0.46 V (vs. Ag/AgCl) is linear in the 0.003 – 8 mM concentration range, and the detection limit is 0.21 mM (at an S/N ratio of 3). Satisfactory results were obtained in the analysis of tap, rain and river waters spiked with H2O2. The analytical performance of this electrode compares favorably to the results obtained with other commonly used techniques for analysis of H2O2.

Copper doped CuO nanosheets have been synthesized by the electro-deposition method, and then it was further used for the construction of a hydrogen peroxide sensor.






Similar content being viewed by others
References
Xuan J, Jia XD, Jiang LP, Abdel-Halim ES, Zhu JJ (2012) Gold nanoparticle-assembled capsules and their application as hydrogen peroxide biosensor based on hemoglobin. Bioelectrochemistry 84:32–37
Kim M, Lee HJ, Oh S, Kim Y, Jung H, Oh MK, Yoon YJ, Yoo TH, Yoon TS, Lee HH (2014) Robust ZnO nanoparticle embedded memory device using vancomycin conjugate and its biorecognition for electrical charging node. Biosens Bioelectron 56:33–38
Teimuri-Mofrad R, Safa KD, Rahimpour K (2014) Synthesis, characterization and electrochemical properties of novel trinuclear ferrocenyl based organosilane compounds. J Org Chem 758:36–44
Wang F, Xie Z, Zhang B, Liu Y, Yang WD, Liu CY (2014) Down-and up-conversion luminescent carbon dot fluid: inkjet printing and gel glass fabrication. Nanoscale 6:3818–3823
Veerapandian M, Seo YT, Yun K, Lee MH (2014) Graphene oxide functionalized with silver@silica-polyethylene glycol hybrid nanoparticles for direct electrochemical detection of quercetin. Biosens Bioelectron 58:200–204
Wang XJ, Wu W, Xiang XD, Li WS (2014) Pore-arrayed hydrogen molybdenum bronze: preparation and performance as support of platinum nanoparticles for methanol oxidation. J Power Sources 259:255–261
Jiang BB, Wei XW, Wu FH (2014) A non-enzymatic hydrogen peroxide sensor based on a glassy carbon electrodemodified with cuprous oxide and nitrogen-doped graphene in a nafion matrix. Microchim Acta 181:1463–1470
Zhao WW, Ma ZY, Xu JJ, Chen HY (2013) In situ modification of a semiconductor surface by an enzymatic process: a general strategy for photoelectrochemical bioanalysis. Anal Chem 85:8503–8506
Ensafi AA, Abarghoui MM, Rezaei B (2014) Electrochemical determination of hydrogen peroxide using copper/porous silicon basednon-enzymatic sensor. Sens Actuators B-Chem 196:398–405
Xiong W, Qu Q, Liu ST (2014) Self-assembly of ultra-small gold nanoparticles on an indium tin oxide electrode for the enzyme-free detection of hydrogen peroxide. Microchim Acta 181:983–989
Zhao WJ, Wang ZH, Li QM (2012) Fabrication of a nichrome electrode coated with silver microcrystals, and its application to sensing hydrogen peroxide. Anal Methods 4:1105–1109
Qian L, Yang XR (2008) Dendrimer films as matrices for electrochemical fabrication of novel gold/palladium bimetallic nanostructures. Talanta 74:1649–1653
Annamalai SK, Palani B, Pillai KC (2012) Highly stable and redox active nano copper species stabilized functionalized-multiwalled carbon nanotube/chitosan modified electrode for efficient hydrogen peroxide detection. Colloids Surf A 395:207–216
Zeng AP, Jin CY, Cho SJ, Seo HO, Kim YD, Lim DC, Kim DH, Hong BY, Boo JH (2012) Nickel nano-particle modified nitrogen-doped amorphous hydrogenated diamond-like carbon film for glucose sensing. Mater Res Bull 47:2713–2716
Cao F, Guo S, Ma HY, Yang GC, Yang SX, Gong J (2011) Highly sensitive nonenzymatic glucose sensor based on electrospun copper oxide-doped nickel oxide composite microfibers. Talanta 86:214–220
Tian LL, Liu B (2013) Fabrication of CuO nanosheets modified Cu electrode and its excellent electrocatalytic performance towards glucose. Appl Surf Sci 283:947–953
Zhu L, Shao G, Luo JK (2012) Numerical study of metal oxide Schottky type solar cells. Solid State Sci 14:857–863
Singh I, Bedi RK (2011) Influence of pH on the synthesis and characterization of CuO powder for thick film room-temperature NH3 gas sensor. J Mater Sci 46:5568–5580
Verma MK, Gupta V (2012) A highly sensitive SnO-CuO multilayered sensor structure for detection of H2S gas. Sens. Actuators B 166–167:378–385
Priya S, Berchmans S (2012) Copper oxide-modified glassy carbon electrode prepared through copper hexacyanoferrate-G5-PAMAM dendrimer templates as electrocatalyst for carbohydrate and alcohol oxidation. J Solid State Electrochem 16:1527–1535
Vaseem M, Hong AR, Kim RT, Hahn YB (2013) Copper oxide quantum dot ink for inkjet-driven digitally controlled high mobility field effecttransistors. J Mater Chem C 1:2112–2120
Zhang YC, Liu YX, Su L, Zhang ZH, Huo DQ, Hou CJ, Lei Y (2014) CuO nanowires based sensitive and selective non-enzymatic glucose detection. Sens Actuators B 191:86–93
Park CM, Ahn WJ, Jo WK, Song JH, Oh CY, Jeong YS, Kim KJ, Chung MC, Jeong WJ, Park KP (2013) Supported CuO and ZnO catalyst for hydrogenation of carbon dioxide to methyl alcohol. Adv Mater Res 550–553:396–399
Han X, Yu YB, He H, Zhao JJ (2013) Low CO content hydrogen production from oxidative steam reforming of ethanol over CuO-CeO2 catalysts at low-temperature. J Energy Chem 22:861–868
Li H, Liao JY, Zeng T (2013) A facile synthesis of CuO nanowires and nanorods, and their catalytic activity in the oxidative degradation of rhodamine B with hydrogen peroxide. Catal Commun 46:169–173
Zheng ZK, Huang BB, Wang ZY, Guo M, Qin XY, Zhang XY, Wang P, Dai Y (2009) Crystal faces of Cu2O and their stabilities in photocatalytic reactions. J Phys Chem C 113:14448–14453
Song HY, Ni YN, Kokot S (2013) A glassy carbon electrode modified with poly(anthranilic acid), poly(diphenylamine sulfonate) and CuO nano-particles for the sensitive determination of hydrogen peroxide. Microchim Acta 180:1263–1270
Yang J, Jiang LC, Zhang WD, Gunasekaran S (2010) A highly sensitive non-enzymatic glucose sensor based on a simple two-step electrodeposition of cupric oxide (CuO) nanoparticles onto multi-walled carbon nanotube arrays. Talanta 82:25–33
Jin CY, Zeng AP, Cho SJ, Boo JH (2012) Investigation of copper and silver nanoparticles deposited on a nitrogen-doped diamond-like carbon (N-DLC) film electrode for Bio-sensing. J Korean Phys Soc 60:912–915
Erdogan IY, Gullu O (2010) Optical and structural properties of CuO nanofilm: its diode application. J Alloys Compd 492:378–383
Ahmad R, Mehrnaz G (2012) Preparation and properties of semiconductor CuO nanoparticles via a simple precipitation method at different reaction temperatures. Opt Quant Electron 44:313–322
Song HY, Ni YN, Kokot S (2013) A novel electrochemical biosensor based on the hemin-graphene nanosheets and gold nanoparticles hybrid film for the analysis of hydrogen peroxide. Anal Chim Acta 788:24–31
Nazari A, Rafieipour MH, Riahi S (2011) The effects of CuO nanoparticles on properties of self compacting concrete with GGBFS as binder, materials research-ibero-american. J Mater 14:307–316
Liu RJ, Li SW, Yu XL, Zhang GJ, Zhang SJ, Yao JN, Keita B, Nadjo L, Zhi LJ (2012) Facile synthesis of Au-nanoparticle/Polyoxometalate/Graphene tricomponent nanohybrids: an enzyme-free electrochemical biosensor for hydrogen peroxide. Small 8:1398–1406
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, H., Ma, C., You, L. et al. Electrochemical hydrogen peroxide sensor based on a glassy carbon electrode modified with nanosheets of copper-doped copper(II) oxide. Microchim Acta 182, 1543–1549 (2015). https://doi.org/10.1007/s00604-015-1485-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-015-1485-9