Abstract
Purpose
The present study assessed postoperative bowel dysfunction in Japanese patients with rectal cancer, including patients who underwent preoperative radiotherapy (RT).
Methods
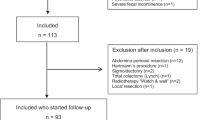
A total of 277 rectal cancer patients who underwent primary resection were included in the analyses. A questionnaire survey was administered using the low anterior resection syndrome (LARS) score and Wexner score. Scores were determined one year after rectal surgery or diverting ileostomy closure. The LARS score was categorized as minor LARS (21–29) and major LARS (30–42).
Results
The proportions of patients with minor and major LARS were significantly larger and Wexner scores significantly higher in patients with distal tumors and a lower anastomosis level than in those with proximal tumors and a higher anastomosis level. Among the patients with lower rectal cancer, the proportions with minor and major LARS were similar between those with and without preoperative RT. The Wexner scores in patients with preoperative RT were significantly higher than in patients without RT. A distal tumor location and lower anastomosis level were independent risk factors of major LARS in multivariate analyses.
Conclusion
A distal tumor location, low anastomosis level, and preoperative RT might be associated with postoperative bowel dysfunction in rectal cancer patients.




Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209–49.
Kasi A, Abbasi S, Handa S, Al-Rajabi R, Saeed A, Baranda J, et al. Total neoadjuvant therapy vs standard therapy in locally advanced rectal cancer: a systematic review and meta-analysis. JAMA Netw Open. 2020;3(12): e2030097.
Morris E, Quirke P, Thomas JD, Fairley L, Cottier B, Forman D. Unacceptable variation in abdominoperineal excision rates for rectal cancer: time to intervene? Gut. 2008;57(12):1690–7.
Henley SJ, Ward EM, Scott S, Ma J, Anderson RN, Firth AU, et al. Annual report to the nation on the status of cancer, part I: national cancer statistics. Cancer. 2020;126(10):2225–49.
Saito N, Moriya Y, Shirouzu K, Maeda K, Mochizuki H, Koda K, et al. Intersphincteric resection in patients with very low rectal cancer: a review of the Japanese experience. Dis Colon Rectum. 2006;49(10 Suppl):S13-22.
Cornish JA, Tilney HS, Heriot AG, Lavery IC, Fazio VW, Tekkis PP. A meta-analysis of quality of life for abdominoperineal excision of rectum versus anterior resection for rectal cancer. Ann Surg Oncol. 2007;14(7):2056–68.
Colquhoun P, Kaiser R Jr, Efron J, Weiss EG, Nogueras JJ, Vernava AM 3rd, et al. Is the quality of life better in patients with colostomy than patients with fecal incontience? World J Surg. 2006;30(10):1925–8.
Williamson ME, Lewis WG, Holdsworth PJ, Finan PJ, Johnston D. Decrease in the anorectal pressure gradient after low anterior resection of the rectum. A study using continuous ambulatory manometry. Dis Colon Rectum. 1994;37(12):1228–31.
Bryant CL, Lunniss PJ, Knowles CH, Thaha MA, Chan CL. Anterior resection syndrome. Lancet Oncol. 2012;13(9):e403-408.
Juul T, Ahlberg M, Biondo S, Emmertsen KJ, Espin E, Jimenez LM, et al. International validation of the low anterior resection syndrome score. Ann Surg. 2014;259(4):728–34.
Emmertsen KJ, Laurberg S. Low anterior resection syndrome score: development and validation of a symptom-based scoring system for bowel dysfunction after low anterior resection for rectal cancer. Ann Surg. 2012;255(5):922–8.
Bretagnol F, Rullier E, Laurent C, Zerbib F, Gontier R, Saric J. Comparison of functional results and quality of life between intersphincteric resection and conventional coloanal anastomosis for low rectal cancer. Dis Colon Rectum. 2004;47(6):832–8.
Chamlou R, Parc Y, Simon T, Bennis M, Dehni N, Parc R, et al. Long-term results of intersphincteric resection for low rectal cancer. Ann Surg. 2007;246(6):916–21.
Ito M, Saito N, Sugito M, Kobayashi A, Nishizawa Y, Tsunoda Y. Analysis of clinical factors associated with anal function after intersphincteric resection for very low rectal cancer. Dis Colon Rectum. 2009;52(1):64–70.
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351(17):1731–40.
Fujita S, Mizusawa J, Kanemitsu Y, Ito M, Kinugasa Y, Komori K, et al. Mesorectal excision with or without lateral lymph node dissection for clinical stage II/III lower rectal cancer (JCOG0212): a multicenter, randomized controlled. Noninferiority Trial Ann Surg. 2017;266(2):201–7.
Shinagawa T, Tanaka T, Nozawa H, Emoto S, Murono K, Kaneko M, et al. Comparison of the guidelines for colorectal cancer in Japan, the USA and Europe. Ann Gastroenterol Surg. 2018;2(1):6–12.
Honda M, Akiyoshi T, Noma H, Ogura A, Nagasaki T, Konishi T, et al. Patient-centered outcomes to decide treatment strategy for patients with low rectal cancer. J Surg Oncol. 2016;114(5):630–6.
Ribas Y, Aguilar F, Jovell-Fernandez E, Cayetano L, Navarro-Luna A, Munoz-Duyos A. Clinical application of the LARS score: results from a pilot study. Int J Colorectal Dis. 2017;32(3):409–18.
Jorge JM, Wexner SD. Etiology and management of fecal incontinence. Dis Colon Rectum. 1993;36(1):77–97.
Haanstra JF, de Vos Tot Nederveen Cappel WH, Gopie JP, Vecht J, Vanhoutvin SA, Cats A, et al. Quality of life after surgery for colon cancer in patients with Lynch syndrome: partial versus subtotal colectomy. Dis Colon Rectum. 2012;55(6):653–9.
You YN, Chua HK, Nelson H, Hassan I, Barnes SA, Harrington J. Segmental vs. extended colectomy: measurable differences in morbidity, function, and quality of life. Dis Colon Rectum. 2008;51(7):1036–43.
Japanese Society for Cancer of thr, REctum. Japanese classification of colorectal, appendiceal, and anal carcinoma: the 3d english edition [secondary publication]. J Anus Rectum Colon. 2019;3:175–95.
Sasaki S, Nagasaki T, Oba K, Akiyoshi T, Mukai T, Yamaguchi T, et al. Risk factors for outlet obstruction after laparoscopic surgery and diverting ileostomy for rectal cancer. Surg Today. 2021;51(3):366–73.
Akiyoshi T, Toda S, Tominaga T, Oba K, Tomizawa K, Hanaoka Y, et al. Prognostic impact of residual lateral lymph node metastasis after neoadjuvant (chemo)radiotherapy in patients with advanced low rectal cancer. BJS Open. 2019;3(6):822–9.
Konishi T, Shinozaki E, Murofushi K, Taguchi S, Fukunaga Y, Nagayama S, et al. Phase II trial of neoadjuvant chemotherapy, chemoradiotherapy, and laparoscopic surgery with selective lateral node dissection for poor-risk low rectal cancer. Ann Surg Oncol. 2019;26(8):2507–13.
Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020;25(1):1–42.
Peeters KC, Marijnen CA, Nagtegaal ID, Kranenbarg EK, Putter H, Wiggers T, et al. The TME trial after a median follow-up of 6 years: increased local control but no survival benefit in irradiated patients with resectable rectal carcinoma. Ann Surg. 2007;246(5):693–701.
van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12(6):575–82.
Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345(9):638–46.
Haddock MG, Sloan JA, Bollinger JW, Soori G, Steen PD, Martenson JA, et al. Patient assessment of bowel function during and after pelvic radiotherapy: results of a prospective phase III North Central Cancer Treatment Group clinical trial. J Clin Oncol. 2007;25(10):1255–9.
Lorenzi B, Brading AF, Martellucci J, Cetta F, Mortensen NJ. Short-term effects of neoadjuvant chemoradiotherapy on internal anal sphincter function: a human in vitro study. Dis Colon Rectum. 2012;55(4):465–72.
Lundby L, Krogh K, Jensen VJ, Gandrup P, Qvist N, Overgaard J, et al. Long-term anorectal dysfunction after postoperative radiotherapy for rectal cancer. Dis Colon Rectum. 2005;48(7):1343–9 (discussion 1349-1352; author reply 1352).
Croese AD, Lonie JM, Trollope AF, Vangaveti VN, Ho YH. A meta-analysis of the prevalence of low anterior resection syndrome and systematic review of risk factors. Int J Surg. 2018;56:234–41.
Bolton WS, Chapman SJ, Corrigan N, Croft J, Collinson F, Brown JM, et al. The incidence of low anterior resection syndrome as assessed in an international randomized controlled trial (MRC/NIHR ROLARR). Ann Surg. 2021;274(6):e1223–9.
Battersby NJ, Juul T, Christensen P, Janjua AZ, Branagan G, Emmertsen KJ, et al. Predicting the risk of bowel-related quality-of-life impairment after restorative resection for rectal cancer: A Multicenter Cross-Sectional Study. Dis Colon Rectum. 2016;59(4):270–80.
Chen TY, Emmertsen KJ, Laurberg S. What Are the Best Questionnaires to capture anorectal function after surgery in rectal cancer? Curr Colorectal Cancer Rep. 2015;11:37–43.
Akizuki E, Okita K, Noda A, Tsuruma T, Nishimori H, Sasaki K, et al. Clinical utility and characteristics of the LARS Score compared to the CCIS. World J Surg. 2022;46(4):925–32.
Emmertsen KJ, Laurberg S. Rectal Cancer Function Study G: Impact of bowel dysfunction on quality of life after sphincter-preserving resection for rectal cancer. Br J Surg. 2013;100(10):1377–87.
Pieniowski EHA, Nordenvall C, Palmer G, Johar A, Tumlin Ekelund S, Lagergren P, et al. Prevalence of low anterior resection syndrome and impact on quality of life after rectal cancer surgery: population-based study. BJS Open. 2020;4(5):935–42.
Acknowledgements
We thank Jane Charbonneau, DVM, from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest or ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kiyozumi, Y., Nagasaki, T., Matsui, S. et al. The evaluation of postoperative bowel dysfunction in Japanese patients with rectal cancer. Surg Today 53, 596–604 (2023). https://doi.org/10.1007/s00595-022-02598-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02598-1