Abstract
Aims
The current method to diagnose impaired glucose tolerance (IGT) is based on the 2-h plasma glucose (2-hPG) value during a 75-g oral glucose tolerance test (OGTT). Robust evidence demonstrates that the 1-h post-load plasma glucose (1-hPG) ≥ 8.6 mmol/L in those with normal glucose tolerance is highly predictive of type 2 diabetes (T2D), micro and macrovascular complications and mortality. The aim of this study was to conduct a health economic analysis to estimate long-term cost-effectiveness of using the 1-hPG compared to the 2-hPG for screening and assessing the risk of diabetes over 35 years. The main outcome was cost per quality-adjusted life year (QALY) gained.
Methods
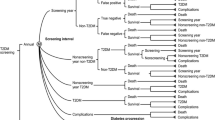
A Monte Carlo–based Markov simulation model was developed to forecast long-term effects of two screening strategies with regards to clinical and cost-effectiveness outcomes. The base case model included 20,000 simulated patients over 35-years follow-up. Transition probabilities on disease progression, mortality, effects on preventive treatments and complications were retrieved from landmark diabetes studies. Direct medical costs were sourced from published literature and inflated to 2019 Euros.
Results
In the lifetime analysis, the 1-hPG was projected to increase the number of years free from disease (2 years per patient); to delay the onset of T2D (1 year per patient); to reduce the incidence of T2D complications (0·6 RR-Relative Risk per patient) and to increase the QALY gained (0·58 per patient). Even if the 1-hPG diagnostic method resulted in higher initial costs associated with preventive treatment, long-term diabetes-related costs as well as complications costs were reduced leading to a lifetime saving of − 31225719.82€. The incremental cost-effectiveness ratio was − 8214.7€ per each QALY gained for the overall population.
Conclusions
Screening prediabetes with the 1-hPG is feasible and cost-effective resulting in reduced costs per QALY. Notwithstanding, the higher initial costs of testing with the 1-hPG compared to the 2-hPG due to incremental preventive intervention, long-term diabetes and complications costs were reduced projecting an overall cost saving of − 8214.7€ per each QALY gained.



Similar content being viewed by others
References
Pedicelli S, Fintini D, Ravà L, Inzaghi E, Deodati A, Spreghini MR et al (2022) Prevalence of prediabetes in children and adolescents by class of obesity. Pediatr Obes. https://doi.org/10.1111/ijpo.12900
Pareek M, Bhatt DL, Nielsen ML, Jagannathan R, Eriksson K-F, Nilsson PM et al (2018) Enhanced predictive capability of a 1-hour oral glucose tolerance test: a prospective population-based cohort study. Diabetes Care 41(1):171–177
Bergman M, Manco M, Sesti G, Dankner R, Pareek M, Jagannathan R et al (2018) Petition to replace current OGTT criteria for diagnosing prediabetes with the 1-hour post-load plasma glucose≥ 155 mg/dl (8.6 mmol/L). Diabetes Res Clin Pract 146:18–33
Fiorentino TV, Marini MA, Succurro E, Andreozzi F, Perticone M, Hribal ML et al (2018) One-hour postload hyperglycemia: implications for prediction and prevention of type 2 diabetes. J Clin Endocrinol Metab 103(9):3131–3143
Fiorentino TV, Succurro E, Andreozzi F, Sciacqua A, Perticone F, Sesti G (2019) One-hour post-load hyperglycemia combined with HbA1c identifies individuals with higher risk of cardiovascular diseases: cross-sectional data from the CATAMERI study. Diabetes Metab Res Rev 35(2):e3096
Jagannathan R, Fiorentino TV, Marini MA, Sesti G, Bergman M (2022) One-hour post-load glucose is associated with severity of hepatic fibrosis. Diabetes Res Clin Pract 189:109977
Bonaventura A, Montecucco F (2019) The STOP DIABETES study: when prevention works. Acta Diabetol 56(5):501–504
Armato JP, DeFronzo RA, Abdul-Ghani M, Ruby RJ (2018) Successful treatment of prediabetes in clinical practice using physiological assessment (STOP DIABETES). Lancet Diabetes Endocrinol 6(10):781–789
Shaw JE, Zimmet PZ, De Courten M, Dowse GK, Chitson P, Ha G et al (1999) Impaired fasting glucose or impaired glucose tolerance. What best predicts future diabetes in Mauritius? Diabetes Care 22(3):399–402
Gabir MM, Hanson RL, Dabelea D, Imperatore G, Roumain J, Bennett PH et al (2000) The 1997 American diabetes association and 1999 world health organization criteria for hyperglycemia in the diagnosis and prediction of diabetes. Diabetes Care 23(8):1108–1112
Stern MP, Williams K, Haffner SM (2002) Identification of persons at high risk for type 2 diabetes mellitus: do we need the oral glucose tolerance test? Ann Intern Med 136(8):575–581
Kahn S (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 46(1):3–19
Molitch ME, Fujimoto W, Hamman RF, Knowler WC (2003) The diabetes prevention program and its global implications. J Am Soc Nephrol 14(suppl 2):S103–S107
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA et al (2002) Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New England j med 346(6):393–403
Group DPPR (2015) Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the diabetes prevention program outcomes study. Lancet Diabetes Endocrinol 3(11):866–875
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW (2008) 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 359(15):1577–1589
Perreault L, Kahn SE, Christophi CA, Knowler WC, Hamman RF, Group DPPR (2009) Regression from pre-diabetes to normal glucose regulation in the diabetes prevention program. Diabetes Care 32(9):1583–1588
Group DPPR (2003) Costs associated with the primary prevention of type 2 diabetes mellitus in the diabetes prevention program. Diabetes Care 26(1):36–47
Bonora E, Bruno G. Osservatorio Arno-Diabete. Il profilo assistenziale della popolazione con diabete. Bologna: Centauro editore, Edizioni Scientifiche 2011.
Berto P, Inzitari D, Scrutinio D, Cimminiello C, Lopatriello S, Rudelli G et al (2010) Clopidogrel idrogenosolfato versus aspirina nella riduzione degli eventi aterotrombotici nelle popolazioni a elevato rischio cardiovascolare: un’analisi italiana di costo-efficacia basata sul trial CAPRIE. PharmacoEconomics Italian Res Articles 12(1):33–50
Roggeri DP, Roggeri A, Brancaccio D, Mazzaferro S, Messa P, Paoletti E et al (2016) End-stage renal disease costs for patients new to hemodialysis in Italy the FARO-2 study. B J Med Med Res 12(5):1
Manco M, Panunzi S, Macfarlane DP, Golay A, Melander O, Konrad T et al (2010) One-hour plasma glucose identifies insulin resistance and β-cell dysfunction in individuals with normal glucose tolerance: cross-sectional data from the relationship between insulin sensitivity and cardiovascular risk (RISC) study. Diabetes Care 33(9):2090–2097
Perreault L, Pan Q, Mather KJ, Watson KE, Hamman RF, Kahn SE et al (2012) Effect of regression from prediabetes to normal glucose regulation on long-term reduction in diabetes risk: results from the diabetes prevention program outcomes study. Lancet 379(9833):2243–2251
Clarke P, Gray A, Holman R (2002) Estimating utility values for health states of type 2 diabetic patients using the EQ-5D (UKPDS 62). Med Decis Making 22(4):340–349
Herman WH, Hoerger TJ, Brandle M, Hicks K, Sorensen S, Zhang P et al (2005) The cost-effectiveness of lifestyle modification or metformin in preventing type 2 diabetes in adults with impaired glucose tolerance. Ann Intern Med 142(5):323–332
Neumann A, Schoffer O, Norström F, Norberg M, Klug SJ, Lindholm L (2014) Health-related quality of life for pre-diabetic states and type 2 diabetes mellitus: a cross-sectional study in Västerbotten Sweden. Health Qual Life Outcomes 12(1):1–10
Alyass A, Almgren P, Akerlund M, Dushoff J, Isomaa B, Nilsson P et al (2015) Modelling of OGTT curve identifies 1 h plasma glucose level as a strong predictor of incident type 2 diabetes: results from two prospective cohorts. Diabetologia 58(1):87–97
Postmus D, de Graaf G, Hillege HL, Steyerberg EW, Buskens E (2012) A method for the early health technology assessment of novel biomarker measurement in primary prevention programs. Stat Med 31(23):2733–2744
Author information
Authors and Affiliations
Contributions
MM and MB: conceptualization. MA, MR: methodology, formal analysis. MTE, AET: data source search and data curation. MA, MM; writing—original draft. All the authors: writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interests.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Additional information
Managed By Massimo Federici .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andellini, M., Manco, M., Esposito, M.T. et al. A simulation model estimates lifetime health and economic outcomes of screening prediabetes using the 1-h plasma glucose. Acta Diabetol 60, 9–17 (2023). https://doi.org/10.1007/s00592-022-01963-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-022-01963-3