Abstract
Aims
Diabetic microvascular complications of retinopathy, nephropathy and neuropathy may occur at hemoglobin A1c levels (HbA1c) below the 6.5% (48 mmol/mol) diagnostic threshold. Our objective was to assess the validity of the HbA1c diagnostic cutpoint of 6.5% based upon published evidence of the prevalence of retinopathy, nephropathy and neuropathy as markers of diabetes.
Methods
Data Sources PubMed, Embase, Cochrane, Scopus and CINAHL from 1990-March 2019, grey literature sources. Study Selection All studies reported after 1990 (to ensure standardized HbA1c values) where HbA1c levels were presented in relation to prevalence of retinopathy, nephropathy or neuropathy in subjects not known to have diabetes. Data Extraction Studies were screened independently, data abstracted, and risk of bias appraised. Data Synthesis Data were synthesized using HbA1c categories of < 6.0% (< 42 mmol/mol), 6.0–6.4% (42–47 mmol/mol) and ≥ 6.5% (≥ 48 mmol/mol). Random-effects meta-analyses were conducted for retinopathy, nephropathy and neuropathy prevalence stratified by HbA1c categories. Random-effects multivariable meta-regression was conducted to identify predictors of retinopathy prevalence and sources of between-study heterogeneity.
Results
Pooled mean prevalence was: 4.0%(95% CI: 3.2–5.0%) for retinopathy, 10.5% (95% CI: 4.0–19.5%) for nephropathy, 2.5% (95% CI: 1.1–4.3%) for neuropathy. Mean prevalence when stratified for HbA1c < 6.0%, 6.0–6.4% and ≥ 6.5% was: retinopathy: 3.4% (95% CI: 1.8–5.4%), 2.3% (95% CI: 1.6–3.2%) and 7.8%(95% CI: 5.7–10.3%); nephropathy: 7.1% (95% CI: 1.7–15.9%), 9.6% (95% CI: 0.8–26.4%) and 17.1% (95% CI: 1.0–46.9%); neuropathy: 2.1% (95% CI: 0.0–6.8%), 3.4% (95% CI: 0.0–11.6%) and 2.8% (95% CI: 0.0–12.8%). Multivariable meta-regression showed HbA1c ≥ 6.5% (OR: 4.05; 95% CI: 1.92–8.57%), age > 55 (OR: 3.23; 95% CI 1.81–5.77), and African-American race (OR: 10.73; 95% CI: 4.34–26.55), to be associated with higher retinopathy prevalence. Marked heterogeneity in prevalence estimates was found across all meta-analyses (Cochran’s Q-statistic p < 0.0001).
Conclusions
The prevalence of nephropathy and moderate retinopathy was increased in subjects with HbA1c values ≥ 6.5% confirming the high specificity of this value for diagnosing T2DM; however, at HbA1c < 6.5% retinopathy increased at age > 55 years and, most strikingly, in African-Americans, suggesting there may be excess microvascular complication prevalence (particularly nephropathy) in individuals below the diabetes diagnostic threshold.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of diabetes has reached epidemic proportions globally, with 424.9 million affected adults (20–79 y), representing 8.8% of the global adult population. Current projections indicate that this figure will rise to 628.6 million by the year 2045, affecting almost 10% of the worldwide adult population [1]. Type 2 diabetes (T2DM) accounts for the vast majority (90–95%) of diabetes cases and is commonly characterized by the inability of pancreatic beta cells to meet the demand for insulin secretion due to a relative deficit of functional beta cells in a setting of peripheral insulin resistance. There has been much debate over the years as to how T2DM should be diagnosed; what should be measured, and the diagnostic targets have changed, being refined as our understanding of the disease has improved along with improvement in analytical methods. The diagnostic criteria for T2DM are established [2], but it is clear that a continuum in blood glucose level exists from normoglycemia to frank diabetes. As such, in 1997 and 2003, the Expert Committee on Diagnosis and Classification of diabetes mellitus recognized a cohort of subjects whose glucose levels did not meet the criteria for diabetes but were too high to be considered as normal [3, 4]; this “prediabetic” group exhibited impaired fasting glucose (IFG) [fasting plasma glucose (FPG) levels of 100–125 mg/dL (5.6–6.9 mmol/L) and/or impaired glucose tolerance (IGT)] defined as a 2-h plasma glucose following a 75 g oral glucose tolerance test (OGTT) of 140–199 mg/dL (7.8–11.0 mmol/L)] and represents individuals at high risk for development of T2DM.
Hemoglobin A1c (HbA1c) is considered key for assessing glycemic control in patients known to have diabetes, and several prospective studies using HbA1c have shown a strong, continuous association between HbA1c and the development of diabetes and complications [5,6,7]. An International Expert Committee (IEC) recommended an HbA1c level of 6.5% (48 mmol/mol) as the diagnostic threshold for T2DM diagnosis [8], purporting that individuals with HbA1c levels above this cutoff have a much higher probability of having retinopathy than those below. Both the American Diabetes Association (ADA) and, subsequently, the World Health Organization (WHO) endorsed this opinion, which was entirely based upon the risk of observing diabetic retinopathy, without consideration for other diabetic microvascular complications; however, the subject has to have had diabetes for a period of time for microvascular complications to develop that may occur at a lower HbA1c than 6.5%.
Key studies contributing to the IEC/ADA/WHO diagnostic threshold decision were the results of the cross-sectional Evaluation of Screening and Early Detection Strategies for Type 2 Diabetes and Impaired Glucose Tolerance (DETECT-2) study [9] and three epidemiological studies undertaken in the 1990s on Pima Indians, Egyptians and US subjects enrolled in the National Health and Nutrition Examination Survey (NHANES) study [3, 10, 11].
The current HbA1c threshold was based on data available at the time of these groups’ reports and, even at the time, there was debate as to whether a level of 6.5% (48 mmol/mol) may be too high since, in many studies, it identified fewer patients as having diabetes than the traditional blood glucose criteria [12]. Since then, a number of studies have been performed with the aim of better characterizing the HbA1c threshold for prevalent or incident retinopathy. The studies prior to 2013 were summarized in a publication by Kowall and Rathmann who looked at retinopathy, nephropathy and neuropathy [13]; since this time, there have been a number of relevant publications which can now be considered.
The key aims of this study were to perform a contemporary systematic review and meta-analysis to verify the HbA1c cutpoint of 6.5% (48 mmol/mol) for the diagnosis of T2DM using currently available retinopathy data and, secondly, to extend the analysis to establish the prevalence of nephropathy and neuropathy at differing levels of HbA1c.
Methods
Data sources and search strategy
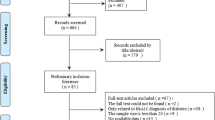
This systematic review was guided by the Cochrane Collaboration Handbook [14], and followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines [15]. The PRISMA checklist is shown in Supplementary Table 1. A comprehensive systematic search for literature was conducted in the academic databases PubMed, Embase, Cochrane, Scopus, CINAHL and in sources for grey literature in October 2019 (Supplementary Table 2).
Pre-searches to identify relevant search terms, search strategies and information sources were performed in May–October 2019. PubMed’s MeSH was used to systematically identify search terms that encompass the variations in terminology for glycated hemoglobin, including thresholds or cutpoints, as well as terms that describe diabetic microvascular complications (Tables 1, 2, 3). The search strategy developed in PubMed was replicated in all databases with eventual search terms or technical variations documented (Supplementary Table 2). The PRESS peer review of electronic search strategies: 2015 guideline statement was used to peer review the search string [16].
All selected search terms were searched in a combination of “Abstract” and “Article Title” (alternatively “Topic” or “Title, Abstract and Keyword”) and in MeSH/Subject Headings/Thesaurus when available. A publication year filter to include studies from 1990 up to the search date was applied to account for the predominant lack of standardized/harmonized HbA1c measurement before this time. In order to ensure literature saturation and inclusion of pre-indexed materials, no additional filters or limitations were included.
In addition to the search in academic databases, Open Grey, Clinical Trials.gov, The New York Academy of Medicine-Grey Literature Report and ProQuest Dissertation and Theses were searched for grey literature. Hand screening of the references lists of all studies selected to be included in the review was also conducted.
A full search log including search technical details, results and notes about search term variations/translations for all databases can be found in Supplementary Table 2.
A review protocol for this study was registered in the Prospero international prospective register of systematic reviews: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=99410PROSPERO2018CRD42018099410.
Study selection
Search results were imported into the Covidence systematic review tool where duplicate publications were identified and duplicates excluded. Study eligibility was independently determined by two investigators (AEB and SLA). Where there was a discrepancy, both investigators revisited the publication in question, discussed the results with the group and re-entered their decision into the database.
The criterion for inclusion was for a publication to report the prevalence of microvascular complications by levels of HbA1c. More specifically, studies were only considered if they provided a prevalence measure for a microvascular complication stratified by HbA1c of < 6% (< 42 mmol/mol), 6–6.4% (42–47 mmol/mol) and ≥ 6.5% (48 mmol/mol) in the same study. The reason for that is to ensure rigor in the association with microvascular complications. Exclusion criteria were studies that did not report microvascular complications by different HbA1c level and those published before 1990.
For terminology, a “publication” is a document containing a relevant outcome measure, while a “study” indicates all details pertaining to a specific outcome measure—one publication may contribute multiple studies. A study, such as “prevalence of retinopathy,” could include multiple stratified “measures,” such as prevalence by age group.
Data extraction and quality assessment
Following screening of records for eligibility, eligible studies had full-text screening (Supplementary Figure 1). Extracted variables included: author(s), publication title, year(s) of data collection, publication year, country of origin, country of survey, study design, study sampling procedure, study population and its characteristics (e.g., sex, age and ethnicity), sample size, HbA1c measurement method, HbA1c stratification outcome measures, complications, retinal photography method, number of eyes photographed, determination of albumin creatinine ratio, measure of peripheral neuropathy. Two investigators (AEB and SLA) independently assessed the full-text articles and determined the eligibility of studies for inclusion in the systematic review.
Quality assessment
Risk of bias (ROB) and precision assessments were performed for all studies included in the review. Guided by the Cochrane approach [17], studies were classified as having “low” vs. “high” ROB on two quality domains assessing (1) consistency in HbA1c diagnostic measurement across all study participants (consistent vs. not consistent) and (2) rigor of sampling methodology (probability-based vs. non-probability-based). Studies with unavailable information for any given domain were classified as having “unclear” ROB for that domain. Studies including at least 100 participants were considered as having higher precision. For a prevalence of microvascular complications of 1% (see prevalence by HbA1c in Table 1) and a sample size of 100, the 95% confidence interval (95% CI) is 0–5%, an acceptable CI for the prevalence of microvascular complications (a lower sample size is needed to detect a higher prevalence). Results of the quality assessment are shown in Supplementary Tables 3 and 4.
Data synthesis and analysis
Statistical analysis
Meta-analysis methodology
Meta-analyses were conducted to estimate the pooled mean of diabetes complications in relation to the stratified HbA1c values (extracted overall outcome measures for a specific HbA1c category, such as 6.0–6.4, were substituted with stratified measures such as by sub-levels of HbA1c, that is 6.0, 6.1, 6.2…, or population characteristics). Forest plots were constructed to visualize prevalence measures and 95% confidence intervals (CIs) associated with each of the three outcomes of interest (retinopathy, nephropathy and neuropathy). Estimates for the pooled mean prevalence and 95% CIs were then calculated using random-effects meta-analyses. Here, variances of prevalence measures were first stabilized using a Freeman–Tukey-type arcsine square-root transformation [18, 19]. Inverse variance weighting [19, 20] was subsequently applied prior to pooling estimates using a DerSimonian–Laird random-effects model [21]. This model assumes a normal distribution for true effect sizes (prevalence) across studies and thus factors in sampling variation and true heterogeneity in effect size [22].
A heterogeneity assessment was further conducted using Cochran’s Q statistic to confirm existence of heterogeneity across studies and I2 to quantify magnitude of between-study variation that is due to true differences in effect size rather than chance [22, 23].
Meta-analyses were implemented in R version 3.4.2 [24].
Meta-regression methodology
Associations with prevalence and sources of between-study heterogeneity were identified using random-effects univariable and multivariable meta-regression analyses. Predictors considered a priori included: HbA1c levels, age and race. Factors associated with prevalence at p value ≤ 0.10 in univariable analysis were eligible for inclusion in the multivariable model. In the latter, a p value ≤ 0.10 but > 0.05 was considered as showing “good evidence” for an association with prevalence, while a p value ≤ 0.05 indicated strong evidence for an association with prevalence.
The magnitude of the association between these predictors and prevalence was determined by calculating, in the univariable analysis, odds ratios (ORs) and their associated 95% CIs, and in the multivariable analysis, by calculating adjusted odds ratios (AORs) and their associated 95% CIs.
Meta-regressions were conducted in Stata/SE version 13 [25] using the metareg package [26].
Results
Literature search
A total of 12,845 references were identified through the literature search and uploaded to the systematic review software Covidence for de-duplication and screening. 9370 references (9324 from the database search and 46 from the grey literature search) qualified for title and abstract screening after de-duplication. The screening process in Covidence software was blinded. Of the 9370 references, 9206 records were excluded. 164 full-text articles were then assessed for eligibility; of those 135 were excluded because of insufficient details of the relationship of the microvascular complication to the HbA1c values less than 6.5% (48 mmol/mol). Twenty nine publications remained eligible for inclusion in the systematic review, 22 relating to retinopathy (Table 1) [6, 9, 11, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46]; 4 relating to nephropathy (Table 2) [28, 38, 47, 48] and 3 relating to neuropathy (Table 3) [38, 49, 50]. No additional relevant references were identified in the hand searching of the reference lists of the 29 included studies. The retinopathy studies contributed 74 stratified measures for the quantitative meta-analysis according to HbA1c as detailed above.
Study characteristics
The definitions of retinopathy, nephropathy and neuropathy used in each study are shown in Tables 1, 2 and 3, respectively.
Diabetic retinopathy, defined as diabetes-related damage to the retina, is classified into early stage non-proliferative diabetic retinopathy (NPDR, comprising microaneurysms and macular edema) and second-stage proliferative diabetic retinopathy (PDR, comprising neovascularization and vitreous hemorrhages) and fundal photography is required for diagnosis [51]. HbA1c was stratified into the following categories in each study: < 6% (< 42 mmol/mol), = 6–6.4% (42–47 mmol/mol) and ≥ 6.5% (≥ 48 mmol/mol).
The forest plot for retinopathy is shown in Fig. 1. The median prevalence of retinopathy was overall 4.0% (range: 0–48.3%), but varied according to HbA1c levels (Table 4). It was 2.7% (range: 0–15.9%) for HbA1c < 6.0% (< 42 mmol/mol), 1.2% (range: 0.2–14.8%) for HbA1c 6.0–6.4% 42–47 mmol/mol) and 5.6% (range: 0.4–48.3%) for HbA1c ≥ 6.5% (≥ 48 mmol/mol).
The forest plot for nephropathy is shown in Fig. 2. The median prevalence of nephropathy was assessed at 11.2% (range: 0.9–32.6%). However, there was also variability based on HbA1c value, with a median of 7.3% (range: 1.0–19.7%) for HbA1c < 6.0% (< 42 mmol/mol), 12.4% (range: 0.9–18.5%) for HbA1c 6.0–6.4% (42–47 mmol/mol) and 26.0% (range: 1.9–32.6%) for HbA1c ≥ 6.5% (≥ 48 mmol/mol) (Table 4).
The forest plot for neuropathy is shown in Fig. 3. The median prevalence for neuropathy was 3.9% (range: 0.2–8.5%). It was 2.8% (range: 0.2–5.0%) for HbA1c < 6.0% (< 42 mmol/mol), 5.6% (range: 0.2–8.5%) for HbA1c 6.0–6.4% (42–47 mmol/mol) and 3.8% (range: 0.2–8.5%) for HbA1c ≥ 6.5% (≥ 48 mmol/mol) (Table 4).
Quality assessment
Supplementary Tables 3 and 4 show the results of the summarized and study-specific quality assessments. In sum, 72.7% (n = 16 out of 22) of retinopathy studies, 50.0% (n = 2 out of 4) of nephropathy studies and 66.7% (n = 2 out of 3) of neuropathy studies included at least 100 participants and therefore were considered as having higher precision.
Low risk of bias, assessed as consistency in measuring HbA1c across study participants, was found in 81.8% of studies assessing retinopathy, all studies assessing nephropathy, and 66.7% of studies assessing neuropathy. The majority of studies assessing retinopathy (95.5%) and all studies assessing nephropathy and neuropathy used probability-based sampling and hence were also classified as having low ROB on that quality domain.
Overall, studies reporting the prevalence of microvascular complications of T2DM were of acceptable quality: 81.8% of retinopathy studies, all nephropathy studies and 66.7% of neuropathy studies had low ROB on both quality domains. High ROB on both domains was found in only 4.5% of retinopathy studies and none of nephropathy or neuropathy studies.
Meta-analysis results
The pooled mean prevalence was estimated at 4.1% (95% CI: 3.2–5.0%) for retinopathy, 10.5% (95% CI: 4.4–18.8%) for nephropathy and 2.5% (95% CI: 1.1–4.3%) for neuropathy (Table 4 and Figs. 1, 2, 3).
There was evidence for heterogeneity in prevalence estimates across all meta-analyses (Table 4). P value for Cochran’s Q statistic was almost always < 0.0001. I2 was > 90% indicating that most variability is due to true differences in prevalence across studies rather than chance.
HbA1c Level and risk of retinopathy
For retinopathy, when the prevalence of complications was stratified according to HbA1c, using categories of < 6.0% (< 42 mmol/mol), 6.0–6.4% (42–46 mmol/mol) and ≥ 6.5% (≥ 48 mmol/mol), there was a distinct increase in retinopathy prevalence from a pooled mean of 3.41% (95% CI: 1.84–5.42) in the < 6.0% (< 42 mmol/mol) stratum and a pooled mean of 2.40% (95% CI: 1.65–3.29) in the 6–6.4% 42–47 mmol/mol) stratum to a pooled mean of 7.97% (95% CI: 5.70–10.57) in the ≥ 6.5% (≥ 48 mmol/mol) stratum. The J-shaped curve describing this association can be found in Fig. 4. The prevalence of retinopathy stratified in 0.1% increments is shown in Fig. 5. The latter analysis showed no trend of increasing retinopathy for HbA1c ranging from 6.0 to 6.4%, but a marked increase in prevalence at HbA1c of 6.5% and greater.
Pooled mean prevalence and adjusted odds ratio of retinopathy according to HbA1c. a Prevalence of retinopathy (%) stratified by HbA1c levels of < 6.0%, 6.1–6.4% and 6.5% or greater, showing marked increase in retinopathy prevalence in the 6.5% or greater group. b Adjusted odds ratio for retinopathy prevalence (for age, sex and ethnicity) stratified by HbA1c levels of < 6.0%, 6.1–6.4% and 6.5% or greater, showing marked increase in the odds ratio in the 6.5% or greater group
HbA1c Level and risk of nephropathy
The albumin/creatinine ratio (expressed as mg albumin: mmol creatinine; ACR) is a sensitive indicator of kidney disease, and patients are classified according to the 2012 guidelines developed by the Kidney Disease: Improving Global Outcomes organization (KDIGO) [52] as: normal < 3 mg/mmol (A1), moderate 3–30 mg/mmol (A2), severe > 30 mg/mmol (A3; nephropathy). Notably, for nephropathy, the prevalence of this complication overall tended to be higher than that for retinopathy. Again, there was a distinct increase in nephropathy prevalence from a pooled mean of 7.12% (95% CI: 1.68–15.85) in the < 6.0% (< 42 mmol/mol) stratum and a pooled mean of 9.56% (95% CI: 0.75–26.41) in the 6–6.4% (42–47 mmol/mol) stratum to a pooled mean of 17.10% (95% CI: 0.96–46.85) in the ≥ 6.5% (≥ 48 mmol/mol) stratum.
HbA1c Level and risk of neuropathy
Diabetic neuropathy, defined as the signs and symptoms of neuropathy wherein diabetes is the underlying cause, most frequently manifests as a distal, symmetric deficit [53]. For neuropathy, no trend in the pooled mean was seen when stratified according to HbA1c: < 6.0% (< 42 mmol/mol) (pooled mean of 2.06%; 95% CI: 0.02–6.76), 6–6.4% (42–47 mmol/mol) (pooled mean 3.39%; 95% CI: 0.00–11.55) and ≥ 6.5% (≥ 48 mmol/mol) (pooled mean 2.81%; 95% CI: 0.00–12.77). This may, however, be due to the paucity of studies available.
Subgroup Analysis
As a higher prevalence of complications may be anticipated with increasing age, the meta-analysis data of studies reporting retinopathy were stratified by age using both two strata (age ≤ 55 years and > 55 years) and three strata (age ≤ 55 years, age 55–59 years and > 60 years) with levels of HbA1c (Table 5). As anticipated, the pooled means did tend to increase with age, though this trend did not differ substantially from the original analysis.
Certain ethnic populations are also known to develop diabetes complications more frequently, notably African-Americans [46]. A sensitivity analysis was therefore performed on the results of the meta-analysis for studies reporting retinopathy separately for African-Americans and non-Black populations (Table 6). The results showed the same upward trend for retinopathy with increasing levels of HbA1c in both the Black and non-Black populations, though the African-Americans populations had a notably higher prevalence of retinopathy at every level of HbA1c; the prevalence of retinopathy in non-African-Americans in those studies did not differ to the overall non-African American prevalence.
There were insufficient studies for both nephropathy and neuropathy to perform subgroup analyses.
Meta-regression results
Only retinopathy prevalence had a sufficient number of studies to warrant conduct of meta-regression analysis (Table 7).
The univariable analysis showed an association with prevalence for HbA1c levels, age and race; these were therefore included in the multivariable model. Here, HbA1c stratification showed a marked and significant increase of retinopathy prevalence at ≥ 6.5% with an AOR of 4.0 (95% CI: 1.9–8.5; p < 0.001) after controlling for the confounding effect of age and race. The model also showed that individuals > 55 years of age had threefold higher odds for retinopathy than younger individuals (AOR: 3.3; 95% CI: 1.8–6.0; p < 0.001). African-Americans also had significantly higher odds for retinopathy prevalence than those of other race (AOR: 11.0; 95% CI: 4.4–27.5; p < 0.001). This model explained 44.8% of the variation in retinopathy prevalence.
Discussion
The meta-analysis relating stratified HbA1c to the prevalence of retinopathy, nephropathy and neuropathy showed clearly that for moderate retinopathy, the recent data are in accord with a HbA1c diagnostic cutoff of 6.5% (48 mmol/mol and above) for T2DM, with the inflection point for the increase in diabetic retinopathy prevalence being at 6.5% (48 mmol/mol), as also shown by the stratified analysis using the J-shaped curve. Pooled mean retinopathy prevalence by 0.1% HbA1c changes showed no obvious signal of a rising retinopathy prevalence for HbA1c 6.0 to 6.4%, with the inflection point being at 6.5% or greater. This being said, there were too few studies assessing retinopathy prevalence at HbA1c increments of 6.3% and 6.4% for evidence to be conclusive. The latter analysis also showed considerable retinopathy prevalence at below 6.0% (42 mmol/mol) HbA1c. It should be also emphasized that the stratification of HbA1c and retinopathy was based on the detection of moderate retinopathy and it is unknown whether the diagnostic cutpoint for T2DM may be altered by prevalence studies on minimal diabetic retinopathy. These results differ from that reported in a previous meta-analysis that concluded that HbA1c thresholds could not be identified from the studies on microvascular complications, though the threshold of HbA1c of 6.5% was strong for severe retinopathy [13].
The heterogeneity in the retinopathy data was controlled for in the meta-regression and showed, in both the univariate and multivariate analyses, the increased odds ratio of 3.2 and 4.05, respectively, with an HbA1c of 6.5% or greater; however, as noted the stratified HbA1c data revealed a J-shaped curve showing accountable retinopathy prevalence at below 6.0%. The meta-regression also revealed the increase in retinopathy prevalence with those aged over 55 years, with an OR of 3.23 and a striking increase in retinopathy prevalence for African-Americans with an OR of 10.73 on multivariate analysis. While this is in accord with the previous data [3, 9,10,11], retinopathy clearly occurs earlier than the cutoff of 6.5%. The magnitude of the association of retinopathy with race found here may not be representative as only 8 studies were available among African-Americans, though it raises the question whether a different diagnostic cutoff for T2DM for African-Americans aged greater than 55 years may be appropriate, as they would appear to be the group at greatest risk [54]. It should also be emphasized that the stratification of HbA1c and retinopathy was based on the detection of moderate retinopathy and it is unknown whether the diagnostic cutpoint for T2DM may be altered by prevalence studies on minimal diabetic retinopathy. Thus, the T2DM diagnostic cutpoint for HbA1c may not be a universal “one size fits all,” but may need to be stratified according to age and race.
The main limitations of the 22 studies of retinopathy included in the meta-analysis were that retinal photography was not standardized and that the degree of retinopathy was often reported as moderate or poorly specified [27, 38]. As different studies have measured the complications in different ways, this may have affected the outcome and we may therefore have underestimated the prevalence of the diabetic complications. There were few studies focusing specifically on the onset of minimal diabetes retinopathy. In addition, minimal retinopathy may be difficult to differentiate between diabetes and non-diabetes-related retinopathy and therefore only moderate diabetes changes were identified in all studies analyzed. This could suggest that with a standardized procedure specifically identifying minimal diabetic retinopathy, that the inflection point for the onset of retinopathy would be a HbA1c lower than 6.5% (48 mmol/mol), but this is clearly speculative as there is insufficient evidence to support this at present.
Four studies were evaluated which considered the onset of nephropathy, the data showing that the HbA1c cutoff of 6.5% (48 mmol/mol) was appropriate with a significant increase in the urine albumin/creatinine ratio. Again, it is evident that there were significant numbers of subjects with albuminuria in the range of “prediabetes” between 6.0–6.4% (42–47 mmol/mol); however, the main limitations were the small number of studies, heterogeneity of the population and lack of division of the HbA1c level into 0.1% ranges that prevented a more stringent determination. In addition, all of the studies were cross sectional with a need for longitudinal studies to be performed.
There were only three studies with sufficient data that could be evaluated for the onset of neuropathy, and these were inconclusive with no inflection point seen for the HbA1c cutoff, with a prevalence of neuropathy at an HbA1c of 6.0–6.4 (42–46 mmol/mol) being the same as that of 6.5% (48 mmol/mol) and above. The main limitations here include the limited amount of data in too few studies, heterogeneity of the population and poor division of the HbA1c range into 0.1% ranges; however, given the limited data, it is unlikely that division of HbA1c into 0.1% ranges would have allowed a more stringent determination.
Conclusions
In conclusion, based on the strongest data of moderate retinopathy, this systematic review and meta-analysis is in accord that the HbA1c diagnostic cutpoint of 6.5% (48 mmol/mol) is highly specific for diagnosing T2DM, though the increased prevalence of retinopathy in those aged 55 years or greater and in African-Americans may suggest a lower threshold is appropriate for those groups. However, this analysis highlights that diabetic microvascular complications commonly occur at lower HbA1c values, and the sensitivity of diagnosing T2DM may improve with a lower diagnostic cutpoint. There is a need for studies that standardize the definition of minimal diabetic retinopathy with more sensitive measures of nephropathy and neuropathy to determine if a lower threshold is appropriate.
Availability of data and materials
All the data for this study will be made available upon reasonable request to the corresponding author.
References
DF Diabetes Atlas (2017); 8th:ttp://www.diabetesatlas.org
Targets G (2019) Standards of medical care in diabetes-2019. Diabetes Care 42(Suppl 1):S61–s70
Report of the expert committee on the diagnosis and classification of diabetes mellitus (1997) Diabetes Care, vol 20(7), pp1183–1197
Genuth S, Alberti KG, Bennett P et al (2003) Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 26(11):3160–3167
Zhang X, Gregg EW, Williamson DF et al (2010) A1C level and future risk of diabetes: a systematic review. Diabetes Care 33(7):1665–1673
Selvin E, Ning Y, Steffes MW et al (2011) Glycated hemoglobin and the risk of kidney disease and retinopathy in adults with and without diabetes. Diabetes 60(1):298–305
Ackermann RT, Cheng YJ, Williamson DF, Gregg EW (2011) Identifying adults at high risk for diabetes and cardiovascular disease using hemoglobin A1c national health and nutrition examination survey 2005–2006. Am J Prev Med 40(1):11–17
Gillett MJ (2009) International expert committee report on the role of the A1c assay in the diagnosis of diabetes: diabetes care 2009; 32(7): 1327–1334. Clin Biochem Rev 30(4):197–200
Colagiuri S, Lee CM, Wong TY, Balkau B, Shaw JE, Borch-Johnsen K (2011) Glycemic thresholds for diabetes-specific retinopathy: implications for diagnostic criteria for diabetes. Diabetes Care 34(1):145–150
McCance DR, Hanson RL, Charles MA et al (1994) Comparison of tests for glycated haemoglobin and fasting and two hour plasma glucose concentrations as diagnostic methods for diabetes. BMJ (Clin Res Ed) 308(6940):1323–1328
Engelgau MM, Thompson TJ, Herman WH et al (1997) Comparison of fasting and 2-hour glucose and HbA1c levels for diagnosing diabetes. Diagnostic criteria and performance revisited. Diabetes Care 20(5):785–791
Cowie CC, Rust KF, Byrd-Holt DD et al (2010) Prevalence of diabetes and high risk for diabetes using A1C criteria in the US population in 1988–2006. Diabetes Care 33(3):562–568
Kowall B, Rathmann W (2013) HbA1c for diagnosis of type 2 diabetes. Is there an optimal cut point to assess high risk of diabetes complications, and how well does the 6.5% cutoff perform? Diabetes, Metab Syndr Obes Targets Ther 6:477–491
Higgins JP, Green S (2011) Cochrane handbook for systematic reviews of interventions. Wiley, New Jersey
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62(10):1006–1012
McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C (2016) PRESS peer review of electronic search strategies: 2015 guideline statement. J Clin Epidemiol 75:40–46
Higgins JPT, Green S (2008) Cochrane Collaboration Cochrane handbook for systematic reviews of interventions. Wiley-Blackwell, NJ
Freeman MF, Tukey JW (1950) Transformations related to the angular and the square root. Ann Math Stat 21:607–611
Miller JJ (1978) The inverse of the freeman—Tukey double arcsine transformation. Am Stat 32(4):138–138
Barendregt JJ, Doi SA, Lee YY, Norman RE, Vos T (2013) Meta-analysis of prevalence. J Epidemiol Commun Health 67(11):974–978
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7(3):177–188
Borenstein M (2009) Introduction to meta-analysis. John Wiley & Sons, Chichester
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558
R core team (2017) R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing
StataCorp (2015) Stata Statistical Software: Release 14. College Station, TX: StataCorp LP. 2015
Harbord RM, Higgins JPT (2008) Meta-regression in Stata. Stata J 8(4):493–519
Ito C, Maeda R, Ishida S, Harada H, Inoue N, Sasaki H (2000) Importance of OGTT for diagnosing diabetes mellitus based on prevalence and incidence of retinopathy. Diabetes Res Clin Pract 49(2–3):181–186
Tapp RJ, Zimmet PZ, Harper CA et al (2006) Diagnostic thresholds for diabetes: the association of retinopathy and albuminuria with glycaemia. Diabetes Res Clin Pract 73(3):315–321
Samadi Aidenloo N, Mehdizadeh A, Valizadeh N, Abbaszadeh M, Qarequran S, Khalkhali H (2016) Optimal glycemic and hemoglobin A1c thresholds for diagnosing diabetes based on prevalence of retinopathy in an Iranian population. Irani Red Crescent Medl J 18(8):e31254
Almdal TP, Handlos LN, Valerius M et al (2014) Glycaemic threshold for diabetes-specific retinopathy among individuals from Saudi Arabia, Algeria and Portugal. Diabetes Res Clin Pract 103(3):e44–46
Bertelsen G, Peto T, Lindekleiv H et al (2014) Sex differences in risk factors for retinopathy in non-diabetic men and women: the tromso eye study. Acta Ophthalmol 92(4):316–322
Bower JK, Brancati FL, Selvin E (2013) No ethnic differences in the association of glycated hemoglobin with retinopathy: the national health and nutrition examination survey 2005–2008. Diabetes Care 36(3):569–573
Cheng YJ, Gregg EW, Geiss LS et al (2009) Association of A1c and fasting plasma glucose levels with diabetic retinopathy prevalence in the US population: implications for diabetes diagnostic thresholds. Diabetes Care 32(11):2027–2032
Cho NH, Kim TH, Woo SJ et al (2013) Optimal HbA1c cutoff for detecting diabetic retinopathy. Acta Diabetol 50(6):837–842
Fukushima S, Nakagami T, Suto C, Hirose A, Uchigata Y (2013) Prevalence of retinopathy and its risk factors in a Japanese population. J Diabetes Invest 4(4):349–354
Park YM, Ko SH, Lee JM et al (2014) Glycaemic and haemoglobin A1c thresholds for detecting diabetic retinopathy: the fifth Korea national health and nutrition examination survey (2011). Diabetes Res Clin Pract 104(3):435–442
Lamparter J, Raum P, Pfeiffer N et al (2014) Prevalence and associations of diabetic retinopathy in a large cohort of prediabetic subjects: the Gutenberg health study. J Diabetes Complicat 28(4):482–487
Metcalf PA, Kyle C, Kenealy T, Jackson RT (2017) HbA1c in relation to incident diabetes and diabetes-related complications in non-diabetic adults at baseline. J Diabetes Complicat 31(5):814–823
Nakagami T, Takahashi K, Suto C et al (2017) Diabetes diagnostic thresholds of the glycated hemoglobin A1c and fasting plasma glucose levels considering the 5-year incidence of retinopathy. Diabetes Res Clin Pract 124:20–29
Okosun IS, Turbow S, McJenkin K, Monique Davis-Smith Y, Seale JP (2016) Diagnostic performance of glycated hemoglobin for diabetic retinopathy in non-diabetic older overweight/obese African-Americans. Diabetes Res Clin Pract 120:124–131
Pang C, Jia L, Jiang S et al (2012) Determination of diabetic retinopathy prevalence and associated risk factors in Chinese diabetic and pre-diabetic subjects: Shanghai diabetic complications study. Diabetes/Metabol Res Rev 28(3):276–283
Sabanayagam C, Khoo EY, Lye WK et al (2015) Diagnosis of diabetes mellitus using HbA1c in Asians: relationship between HbA1c and retinopathy in a multiethnic Asian population. J Clin Endocrinol Metabol 100(2):689–696
Sabanayagam C, Liew G, Tai ES et al (2009) Relationship between glycated haemoglobin and microvascular complications: is there a natural cut-off point for the diagnosis of diabetes? Diabetologia 52(7):1279–1289
Tsugawa Y, Takahashi O, Meigs JB et al (2012) New diabetes diagnostic threshold of hemoglobin A(1c) and the 3-year incidence of retinopathy. Diabetes 61(12):3280–3284
Massin P, Lange C, Tichet J et al (2011) Hemoglobin A1c and fasting plasma glucose levels as predictors of retinopathy at 10 years: the French DESIR study. Arch Ophthalmol 129(2):188–195
Tsugawa Y, Mukamal KJ, Davis RB, Taylor WC, Wee CC (2012) Should the hemoglobin A1c diagnostic cutoff differ between blacks and whites? A cross-sectional study. Ann Intern Med 157(3):153–159
Toulis KA, Jiang CQ, Hemming K et al (2018) Glycated hemoglobin, albuminuria and surrogate markers of macrovascular disease in adults without diabetes: the Guangzhou Biobank cohort study, cardiovascular disease subcohort. Can J Diabetes 42(3):245–250.e241
Xing FY, Neeland IJ, Gore MO et al (2014) Association of prediabetes by fasting glucose and/or haemoglobin A1c levels with subclinical atherosclerosis and impaired renal function: observations from the Dallas Heart Study. Diabetes Vasc Dis Res 11(1):11–18
Tapp RJ, Shaw JE, de Courten MP, Dunstan DW, Welborn TA, Zimmet PZ (2003) Foot complications in Type 2 diabetes: an Australian population-based study. Diabetic Med 20(2):105–113
Kurisu SO KI, Sasaki H, Tanaka H, Yamaneki M, Nakanishi I, Furuta H, et al. (2016) Polyneuropathy or neuropathic pain did not increase at prediabetic stage in Japanese population. J Diabetes Invest
Solomon SD, Chew E, Duh EJ et al (2017) Diabetic retinopathy: a position statement by the American diabetes association. Diabetes Care 40(3):412–418
Stevens PE, Levin A (2013) Kidney disease: improving global outcomes chronic kidney disease guideline development Work group M. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomesclinical practice guideline. Ann Intern Med 158(11):825–830
Bansal V, Kalita J, Misra UK (2006) Diabetic neuropathy. Postgrad Med J 82(964):95–100
Chatterjee R, Maruthur NM, Edelman D (2015) Novel risk factors for type 2 diabetes in African–Americans. Curr DiabRep 15(12):103
Acknowledgements
The authors acknowledge the services provided by the Biostatistics, Epidemiology, and Biomathematics Research Core at the Weill Cornell Medicine-Qatar. SLA is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
Open Access funding provided by the Qatar National Library.
Author information
Authors and Affiliations
Contributions
AEB and SLA performed the systematic analysis, interpreted the data and wrote the manuscript. WGJ, EE, EK and KGMMA conceived of the study and contributed to the writing of the manuscript. HC and LJAR analyzed and interpreted the data and contributed to the writing of the manuscript. LO performed the search and assisted with writing the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No authors have any conflict of interest or competing interests to declare.
Consent to participate
Not applicable.
Consent for publication
All authors gave their consent for publication.
Ethics approval
Not applicable.
Informed consent
Since the studies included in this systematic review and meta-analysis are already published work and therefore in the public domain with no individual identifying information, no informed consent was required to undertake this study.
Human and Animal Rights
No animals were used in the performance of this systematic review and meta-analysis.
Additional information
Managed by Antonio Secchi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Stephen L Atkin and W. Garry John are Joint senior authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
592_2020_1606_MOESM4_ESM.docx
Summary of precision and risk of bias (ROB) assessment. Summary of precision and risk of bias (ROB) assessment for studies reporting the prevalence of microvascular complications associated with type 2 diabetes mellitus (DOCX 17 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Butler, A.E., English, E., Kilpatrick, E.S. et al. Diagnosing type 2 diabetes using Hemoglobin A1c: a systematic review and meta-analysis of the diagnostic cutpoint based on microvascular complications. Acta Diabetol 58, 279–300 (2021). https://doi.org/10.1007/s00592-020-01606-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01606-5