Abstract
Purpose
Patient-reported outcome measures (PROM) are used to measure treatment efficacy in clinical trials. The impact of the choice of a PROM and the cut-off values for ‘meaningful important differences’ (MID) on the study results in patients with lumbar spinal stenosis (LSS) is unclear.
Objective
The objective is to study the consequences of applying different PROMs and values for MID for pain and disability on the proportions of patients with improvement.
Design
Prospective multi-center cohort study.
Methods
Proportions of patients with improvement using established MID cut-off values were calculated and compared for PROMs for pain and disability.
Results
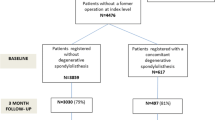
466 patients with LSS completed a baseline and 6-month follow-up assessment and were analyzed. Treatment modalities included surgery (65 %), epidural steroid injections (15 %), or conservative care (20 %). The prevalence of patients fulfilling the criteria for MID ranged from 40 to 70 % across all outcome measures and cut-offs. The agreement of the spinal stenosis outcome measure (SSM) symptom subscale with other pain scales, and the SSM function subscale with other function scales was fair to moderate (Cohen’s κ value between 0.24 and 0.5). Disagreement in the assessment of MID (MID reported by patients in one scale but not the other) was found in at least one-third of the patients.
Conclusion
The MID in outcome scores for this population varied from 40 to 70 %, depending on the measure or cut-off score used. Further, the disagreement between domain specific measures indicates that differences between studies may be also related to the choice of an outcome measures. An international consensus on the use and reporting of outcome measures in studies on lumbar spinal stenosis is needed.


Similar content being viewed by others
References
Barrett B, Brown D, Mundt M, Brown R (2005) Sufficiently important difference: expanding the framework of clinical significance. Med Decis Making 25:250–261. doi:10.1177/0272989X05276863
Stucki G, Daltroy L, Liang MH, Lipson SJ, Fossel AH, Katz JN (1996) Measurement properties of a self-administered outcome measure in lumbar spinal stenosis. Spine (Phila Pa 1976) 21:796–803
Cleland J, Whitman J, Houser J, Wainner R, Childs J (2012) Psychometric properties of selected tests in patients with lumbar spinal stenosis. The spine journal 12:921–931
Pratt RK, Fairbank JC, Virr A (2002) The reliability of the Shuttle Walking Test, the Swiss Spinal Stenosis Questionnaire, the Oxford Spinal Stenosis Score, and the Oswestry Disability Index in the assessment of patients with lumbar spinal stenosis. Spine (Phila Pa 1976) 27:84–91
Sekiguchi M, Wakita T, Otani K, Onishi Y, Fukuhara S, Kikuchi S, Konno S (2012) Development and validation of a symptom scale for lumbar spinal stenosis. Spine (Phila Pa 1976) 37:232–239. doi:10.1097/BRS.0b013e318216afb4
EuroQol Group (1990) EuroQol—a new facility for the measurement of health-related quality of life. The EuroQol Group. Health Policy 16:199–208
Steurer J, Nydegger A, Held U, Brunner F, Hodler J, Porchet F, Min K, Mannion AF, Michel B (2010) LumbSten: the lumbar spinal stenosis outcome study. BMC Musculoskelet Disord 11:254. doi:10.1186/1471-2474-11-254
World Medical Assembly (WMA) (2006) WMA Declaration of Helsinki-Ethical principles for medical research involving human subjects. Adopted by the 18th WMA general assembly, Helsinki, Finland, June 1964 and amended by the 64th WMA General Assembly, Fortaleza, Brazil, October 2013. http://www.wma.net/en/30publications/30ethicsmanual/index.html
Fokter SK, Yerby SA (2006) Patient-based outcomes for the operative treatment of degenerative lumbar spinal stenosis. Eur Spine J 15:1661–1669. doi:10.1007/s00586-005-0033-4
Thornes E, Grotle M (2008) Cross-cultural adaptation of the Norwegian version of the spinal stenosis measure. Eur Spine J 17:456–462. doi:10.1007/s00586-007-0576-7
Wertli MM, Steurer J, Wildi LM, Held U (2014) Cross-cultural adaptation of the German version of the spinal stenosis measure. Eur Spine J 23:1309–1319. doi:10.1007/s00586-014-3245-7
Downie WW, Leatham PA, Rhind VM, Wright V, Branco JA, Anderson JA (1978) Studies with pain rating scales. Ann Rheum Dis 37:378–381. doi:10.1136/ard.37.4.378
Kremer E, Atkinson JH, Ignelzi RJ (1981) Measurement of pain: patient preference does not confound pain measurement. Pain 10:241–248
Choinière M, Amsel R (1996) A visual analogue thermometer for measuring pain intensity. J Pain Symptom Manag 11:299–311
Roland M, Morris R (1983) A study of the natural history of back pain. Part I: development of a reliable and sensitive measure of disability in low-back pain. Spine (Philadelphia, Pa 1976) 8:141–144
Exner V, Keel P (2000) Measuring disability of patients with low-back pain—validation of a German version of the Roland and Morris disability questionnaire. Schmerz 14:392–400
Ostelo RW, Deyo RA, Stratford P, Waddell G, Croft P, Von Korff M, Bouter LM, de Vet HC (2008) Interpreting change scores for pain and functional status in low back pain: towards international consensus regarding minimal important change. Spine (Phila Pa 1976) 33:90–94. doi:10.1097/BRS.0b013e31815e3a10
Friedly JL, Comstock BA, Turner JA, Heagerty PJ, Deyo RA, Sullivan SD, Bauer Z, Bresnahan BW, Avins AL, Nedeljkovic SS, Nerenz DR, Standaert C, Kessler L, Akuthota V, Annaswamy T, Chen A, Diehn F, Firtch W, Gerges FJ, Gilligan C, Goldberg H, Kennedy DJ, Mandel S, Tyburski M, Sanders W, Sibell D, Smuck M, Wasan A, Won L, Jarvik JG (2014) A randomized trial of epidural glucocorticoid injections for spinal stenosis. N Engl J Med 371:11–21. doi:10.1056/NEJMoa1313265
Minamide A, Yoshida M, Yamada H, Nakagawa Y, Hashizume H, Iwasaki H, Tsutsui S (2015) Clinical outcomes after microendoscopic laminotomy for lumbar spinal stenosis: a 5-year follow-up study. Eur Spine J 24:396–403. doi:10.1007/s00586-014-3599-x
Roland M, Fairbank J (2000) The Roland–Morris disability questionnaire and the Oswestry disability questionnaire. Spine (Phila Pa 1976) 25:3115–3124
Viera AJ, Garrett JM (2005) Understanding interobserver agreement: the kappa statistic. Fam Med 37:360–363
Sim J, Wright CC (2005) The kappa statistic in reliability studies: use, interpretation, and sample size requirements. Phys Ther 85:257–268
Vach W (2005) The dependence of Cohen’s kappa on the prevalence does not matter. J Clin Epidemiol 58:655–661. doi:10.1016/j.jclinepi.2004.02.021
Core Team R (2013) R: A language and environment for statistical computing. In: Core Team R (ed) R Foundation for statistical computing. R Core Team, Vienna
Parker SL, Adogwa O, Mendenhall SK, Shau DN, Anderson WN, Cheng JS, Devin CJ, McGirt MJ (2012) Determination of minimum clinically important difference (MCID) in pain, disability, and quality of life after revision fusion for symptomatic pseudoarthrosis. Spine J 12:1122–1128. doi:10.1016/j.spinee.2012.10.006
Munting E, Roder C, Sobottke R, Dietrich D, Aghayev E, Spine Tango C (2015) Patient outcomes after laminotomy, hemilaminectomy, laminectomy and laminectomy with instrumented fusion for spinal canal stenosis: a propensity score-based study from the Spine Tango registry. Eur Spine J 24:358–368. doi:10.1007/s00586-014-3349-0
Mannion AF, Fekete TF, Wertli MM, Mattle M, Nauer S, Kleinstuck FS, Jeszenszky D, Haschtmann D, Becker HJ, Porchet F, Lumbar Spinal Stenosis Outcome Study Group (2015) Could less be more when assessing patient-rated outcome in spinal stenosis? Spine (Phila Pa 1976) 40:710–718. doi:10.1097/BRS.0000000000000751
Machado GC, Ferreira PH, Harris IA, Pinheiro MB, Koes BW, van Tulder M, Rzewuska M, Maher CG, Ferreira ML (2015) Effectiveness of surgery for lumbar spinal stenosis: a systematic review and meta-analysis. PLoS ONE 10:e0122800. doi:10.1371/journal.pone.0122800
Pincus T, Santos R, Breen A, Burton AK, Underwood M, Multinational Musculoskeletal Inception Cohort Study Collaboration (2008) A review and proposal for a core set of factors for prospective cohorts in low back pain: a consensus statement. Arthritis Rheum 59:14–24. doi:10.1002/art.23251
Burgstaller JM, Porchet F, Steurer J, Wertli MM (2015) Arguments for the choice of surgical treatments in patients with lumbar spinal stenosis—a systematic appraisal of randomized controlled trials. BMC Musculoskelet Disord 16:96. doi:10.1186/s12891-015-0548-8
Gum JL, Glassman SD, Carreon LY (2013) Clinically important deterioration in patients undergoing lumbar spine surgery: a choice of evaluation methods using the Oswestry Disability Index, 36-Item Short Form Health Survey, and pain scales: clinical article. J Neurosurg Spine 19:564–568. doi:10.3171/2013.8.spine12804
Lurie JD, Spratt KF, Blood EA, Tosteson TD, Tosteson AN, Weinstein JN (2011) Effects of viewing an evidence-based video decision aid on patients’ treatment preferences for spine surgery. Spine (Phila Pa 1976) 36:1501–1504. doi:10.1097/BRS.0b013e3182055c1e
Stacey D, Legare F, Col NF, Bennett CL, Barry MJ, Eden KB, Holmes-Rovner M, Llewellyn-Thomas H, Lyddiatt A, Thomson R, Trevena L, Wu JH (2014) Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev 1:Cd001431. doi:10.1002/14651858.CD001431.pub4
Acknowledgments
We thank our research staff at all studies sites for their continuous meticulous work to include and follow-up with patients. In particular, we thank MM and GP for their efforts to assure high quality data intake and database handling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources and role of sponsors
The lumbar stenosis outcome study (LSOS) was funded by the Helmut Horten Foundation, OPO-Foundations, Symphasis Foundation, Baugarten Foundation, and the Pfizer-Foundation for geriatrics and research in geriatrics. The funders had no influence on the study design, on the data collection, data management, statistical analysis, the interpretation of the data, the content of the manuscript, and the decision to submit the paper for publication.
Conflict of interest
The authors declare no financial interests or affiliations with institutions, organizations, or companies relevant to the manuscript. All authors had full access to the data, interpreted the analysis and commented on the final manuscript.
Ethical statement
This cohort study was conducted in compliance with all international laws and regulations as well as any applicable guidelines. The study was approved by the independent Ethics Committee of the Canton Zurich (KEK-ZH-NR: 2010-0395/0). A study protocol was published: Steurer et al. [7].
Rights and permissions
About this article
Cite this article
Wertli, M.M., Buletti, F.C., Held, U. et al. A comparison between different outcome measures based on “meaningful important differences” in patients with lumbar spinal stenosis. Eur Spine J 26, 450–461 (2017). https://doi.org/10.1007/s00586-016-4587-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4587-0