Abstract
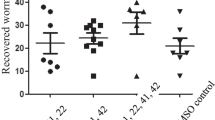
This study investigated the effective dose of methanolic Azadirachta indica leaf extracts, MAILE, combined with diminazene diaceturate, DDA, in the treatment of experimental Trypanosoma brucei brucei infection in rats. Acute toxicity study of the drug and extract combinations was carried in non-infected rats. Eleven different groups of ten rats each were used. Ten out of the eleven groups were infected with T. brucei brucei and used to determine the effective dose of MAILE and DDA combination to be used in the treatment of the infection. All the infected rats were treated, viz, 7.0 mg/kg body weight (bw) DDA plus 500 mg/kg bw MAILE (group 1); 7.0 mg/kg body bw DDA plus 250 mg/kg bw MAILE (group 2); 7.0 mg/kg body bw DDA plus 125 mg/kg bw MAILE (group 3); 3.5 mg/kg body bw DDA plus 500 mg/kg bw MAILE (group 4); 3.5 mg/kg body bw DDA plus 250 mg/kg bw MAILE (group 5); 3.5 mg/kg body bw DDA plus 125 mg/kg bw MAILE (group 6); 1.8 mg/kg bw DDA plus 500 mg/kg bw MAILE (group 7); 1.8 mg/kg bw DDA plus 250 mg/kg bw MAILE (group 8); 1.8 mg/kg body bw DDA plus 125 mg/kg bw MAILE (group 9). Two other groups, infected untreated (group 10) and uninfected untreated (group 11), served as negative and positive control, respectively. The parameters assessed to determine the effective dose combination of the two were onset of parasitaemia (OP), level of parasitaemia (LOP), clearance of parasites post-treatment (COPPT), relapse of infection period (RIP), erythrocyte counts (EC), packed cell volume (PCV) and total leucocyte counts (TLC). There was no significant difference (p < 0.05) in OP between the groups. A day following treatment, the LOP of groups 1, 2, 3 and 4 was found to be significantly lower (p < 0.05) than that of groups 5 and 6 (p < 0.05) which in turn was lower (p < 0.05) than that of groups 7, 8 and 9, respectively. The mean COPPT of groups 5 and 6 was significantly (p < 0.05) longer than that of groups 1, 2, 3 and 4. There was no significant difference (p < 0.05) in the mean COPPT among groups 1, 2, 3 and 4. There was no clearance of parasites in groups 7, 8 and 9. The mean RIP of group 5 and 6 was significantly shorter (p < 0.05) than in group 4. There was no relapse of infection in group 1, 2 and 3 rats. Rats in groups 1, 2, 3 and 4 had significantly higher (p < 0.05) PCV, EC and TLC 10 days post-treatment, and that trend continued throughout the experimental period when compared to other infected groups. It was concluded that dose combination of 125 mg/kg bw extract plus 7 mg/kg bw DDA was the best dose combination judging from the parameters assessed.

Similar content being viewed by others
References
Coles EH (1986) Determination of packed cell volume. In: Coles EH (ed) Veterinary clinical pathology. Saunders, Philadelphia, pp 17–19
Egbe-Nwiyi TN, Igbokwe IO, Onyeyili PA (2006) Relapse of infection in single and mixed trypanosome infections in rats after diminazene aceturate treatment. Veterinarski Arhiv 76(3):255–262
Geerts S, Holmes PH (1998) Drug management and parasite clearance in bovine trypanosomiasis in Africa. The programme against African trypanosomiasis (PAAT), Technical and Scientific series 1. FAO, Rome, pp 5–31
Herbert WJ, Lumsden WHR (1976) Trypanosoma brucei: a rapid “matching method” for estimating the host parasitaemia. Exp Parasitol 40:427–431
Holmes PH, Eisler MC, Geerts S (2004) Current chemotherapy of animal trypanosomiasis. In: Maudlin I, Holmes PH, Miles MA (eds) The trypanosomiasis. CAB International, Wallingford, pp 431–444
Katzung BG (1998) Drugs used in gastrointestinal diseases. In: Basic and clinical pharmacology. Appleton and Lange, Norwalk
Ketkar CM (1976) Utilization of neem and its by-products. Final technical report. Khadi and Village Industries Commission, Bombay
Nok AJ, Eseivo KAN, Longdet I et al (1993) Trypanocidal potentials of Azadirachta indica: in vivo activity of leaf extract against Trypanosoma brucei. J Clin BiochemNutrit 15:113–118
Omoja VU, Anaga AO, Obidike IR, Ihedioha TE, Umeakuana PU, Mhomga LI, Asuzu IU, Anika SM (2011a) The effects of combination of methanolic leaf extract of Azadirachta indica and diminazene diaceturate in the treatment of experimental Trypanosoma brucei brucei infection in rats. Asian Pac J Trop Med 4(5):337–341
Omoja VU, Anaga OA, Obidike IK, Ihedioha TE, Umeakuana PU, Mhomga LI, Asuzu IU, Anika SM (2011b) Erythrocytic profile of rats infected with T. brucei brucei and treated with a combination of Azadirachta indica leaf extract and diminazene diaceturate. Comp Clin Pathol. doi:10.1007/s00580-011-1297-1
SaiRam M, Iiavazhagan G, Sharma SK (2000) Animicribial activity of a new vaginal contraceptive: NIM-76 from neem oil. J Ethnopharm 71:377–382
Samdi SM, Abenga JN, Attahir A, Wayo BM, Sumayin HM, Haruna MK et al (2010) Constraints in the control of African trypanosomiasis: the prevailing factor in Kurmin Kaduna, Northern, Nigeria. Int J Anim Vet Adv 2(1):31–36
Samdi SM, Fajinmi AO, Kalejaye JO, Wayo B, Haruna MK, Yamap JE et al (2011) Prevalence of trypanosomosis in cattle at slaughter in Kaduna central abattoir. Asian J Anim Sci 5(2):162–165
Schalm OW, Jain NC, Carroll EJ (1975) Veterinary haematology, 3rd edn. Lea & Febiger, Philadelphia, pp 17–25, pp. 129–125 and 268–269
Schmutterer H (1990) Properties and potential of natural pesticides from the neem (Azadirachta indica). Ann Rev Entomol 35:271–297
Shetty SN, Anika SM (1982) Dose response relationship. In: Laboratory manual of pharmacology and toxicology. Fourth Dimension Publishers, Enugu, pp 30–33
Venugopalan A (1994) Essentials of veterinary surgery, 7th edn. Oxford and IBH Publishing Co. PVT. LTD, New Delhi, pp 423–424
WHO (2001) PAN pesticides database. Pesticide Action Network, North America, San Francisco
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Omoja, V.U., Obidike, I.R. Determination of effective dose combination of Azadirachta indica leaf extracts and diminazene diaceturate in the treatment of experimental Trypanosoma brucei brucei infection in rats. Comp Clin Pathol 22, 747–754 (2013). https://doi.org/10.1007/s00580-012-1477-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-012-1477-7