Abstract
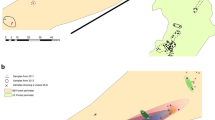
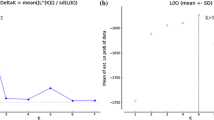
Tuber species are ectomycorrhizal ascomycetes establishing relationships with different host trees and forming hypogeous fruiting bodies known as truffles. Among Tuber species, Tuber aestivum Vittad. has a wide distributional range being found naturally all over Europe. Here, we performed large-scale population genetic analyses in T. aestivum to (i) investigate its genetic diversity at the European scale, (ii) characterize its genetic structure and test for the presence of ecotypes and (iii) shed light into its demographic history. To reach these goals, 230 ascocarps from different populations were genotyped using 15 polymorphic simple sequence repeat markers. We identified 181 multilocus genotypes and four genetic groups which did not show a clear geographical separation; although, one of them was present exclusively in Southeast France, Italy and Spain. Fixation index values between pairs of genetic groups were generally high and ranged from 0.29 to 0.45. A significant deficit of heterozygosity indicated a population expansion instead of a recent population bottleneck, suggesting that T. aestivum is not endangered in Europe, not even in Mediterranean regions. Our study based on a large-scale population genetic analysis suggests that genetically distinct populations and likely ecotypes within T. aestivum are present. In turn, this study paves the way to future investigations aimed at addressing the biological and/or ecological factors that have concurred in shaping the population genetic structure of this species. Present results should also have implications for the truffle market since defining genetic markers are now possible at least for some specific T. aestivum genetic groups.



Similar content being viewed by others
References
Adriaensen K, Vrålstad T, Noben JP, Vangronsveld J, Colpaert JV (2005) Copper-adapted Suillus luteus, a symbiotic solution for pines colonizing Cu mine spoils. Appl Environ Microbiol 71:7279–7284
Agapow PM, Burt A (2001) Indices of multilocus linkage disequilibrium. Mol Ecol Notes 1:101–102
Belfiori B, Riccioni C, Paolocci F, Rubini A (2013) Mating type locus of Chinese black truffles reveals heterothallism and the presence of cryptic species within the T. indicum species complex. PLoS One 8, e82353
Bergemann SE, Douhan GW, Garbelotto M, Miller SL (2006) No evidence of population structure across three isolated subpopulations of Russula brevipes in an oak/pine woodland. New Phytol 170:177–184
Bonito GM, Gryganskyi AP, Trappe JM, Vilgalys R (2010) A global meta-analysis of Tuber ITS rDNA sequences: species diversity, host associations and long-distance dispersal. Mol Ecol 19:4994–5008
Bonito G, Smith ME, Nowak M, Healy RA, Guevara G, Cázares E et al (2013) Historical biogeography and diversification of truffles in the Tuberaceae and their newly identified southern hemisphere sister lineage. PLoS One 8, e52765
Büntgen U, Egli S, Camarero JJ, Fischer EM, Stobbe U, Kauserud H, Tegel W, Sproll L, Stenseth NC (2012) Drought-induced decline in Mediterranean truffle harvest. Nat Clim Chang 2:827–829
Callot G (1999) La Truffe, La Terre, la Vie. INRA, Paris, 210p
Carriconde F, Gardes M, Jargeat P, Heilmann-Clausen J, Mouhamadou B, Gryta H (2008) Population evidence of cryptic species and geographical structure in the cosmopolitan ectomycorrhizal fungus, Tricholoma scalpturatum. Microb Ecol 56:513–524
Ceruti A, Fontana A, Nosenzo C (2003) Le specie Europee del genere Tuber, una revisione storica. Museo Regionale di Scienze Naturali, Monographie XXXVII, Regione Piemonte, Torino
Chatin A (1887) Une nouvelle espèce de truffe. C R Acad Sci 104:1132–1135
Chevalier G, Frochot H (2002) La truffe de Bourgogne. Editions Pétrarque, Levallois-Perret, France
Chevalier G, Desmas C, Frochot H, Riousset L (1979) L’ espèce Tuber aestivum Vitt. : I. Définition. Mushroom Science X, (Part 1):957–975
Colpaert JV, Vandenkoornhuyse P, Adriaensen K, Vangronsveld J (2000) Genetic variation and heavy metal tolerance in the ectomycorrhizal basidiomycete Suillus luteus. New Phytol 147:367–379
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Cornuet JM, Piry S, Luikart G, Estoup A, Solignac M (1999) New methods employing multilocus genotypes to select or exclude populations as origins of individuals. Genetics 153:1989–2000
Di Rienzo A, Peterson AC, Garza JC, Valdes AM, Slatkin M, Freimer NB (1994) Mutational processes of simple-sequence repeat loci in human populations. Proc Natl Acad Sci U S A 91:3166–3170
Douhan GW, Vincenot L, Gryta H, Selosse MA (2011) Population genetics of ectomycorrhizal fungi: from current knowledge to emerging directions. Fungal Biol 115:569–597
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20
Dupré C, Chevalier G, Branlard G (1985) Caractérisation des Tuber par électrophorèse de leurs protéines. In CR 1er Colloque national sur les technologies de purification des protéines. DPIC-INPL, Paris, pp 465–467
Gandeboeuf D, Dupré C, Chevalier G (1994) Use of isoenzyme analysis to differentiate truffles from Europe. Acta Bot Gallica 141:455–463
García-Cunchillos I, Sánchez S, Barriuso JJ, Pérez-Collazos E (2014) Population genetics of the westernmost distribution of the glaciations-surviving black truffle Tuber melanosporum. Mycorrhiza. doi:10.1007/s00572-013-0540-9
Grubisha LC, Bergemann SE, Bruns TD (2007) Host islands within the California Northern Channel Islands create fine-scale genetic structure in two sympatric species of the symbiotic ectomycorrhizal fungus Rhizopogon. Mol Ecol 16:1811–1822
Guillot G (2008) Inference of structure in subdivided populations at low levels of genetic differentiation—the correlated allele frequencies model revisited. Bioinformatics 24:2222–2228
Guillot G, Mortier F, Estoup A (2005a) GENELAND: a computer package for landscape genetics. Mol Ecol Notes 5:712–715
Guillot G, Estoup A, Mortier F, Cosson JF (2005b) A spatial statistical model for landscape genetics. Genetics 170:1261–1280
Hardy OJ, Vekemans X (2002) SPAGeDi: a versatile computer program to analyse spatial genetic structure at the individual or population levels. Mol Ecol Notes 2:618–620
Hitchcock CJ, Chambers SM, Cairney JWG (2011) Genetic population structure of the ectomycorrhizal fungus Pisolithus microcarpus suggests high gene flow in south-eastern Australia. Mycorrhiza 21:131–137
Hortal S, Trocha LK, Murat C, Chybicki IJ, Buée M, Trojankiewicz M et al (2012) Beech roots are simultaneously colonized by multiple genets of the ectomycorrhizal fungus Laccaria amethystina clustered in two genetic groups. Mol Ecol 21:2116–2129
IPCC (2013) Fifth Assessment Report Climate Change 2013: The Physical Science Basis—Summary for Policymakers, Working Group I Contribution to the IPCC, 2013
Jargeat P, Martos F, Carriconde F, Gryta H, Moreau PA, Gardes M (2010) Phylogenetic species delimitation in ectomycorrhizal fungi and implications for barcoding: the case of the Tricholoma scalpturatum complex (Basidiomycota). Mol Ecol 19:5216–5230
Jeandroz S, Murat C, Wang YJ, Bonfante P, Le Tacon F (2008) Molecular phylogeny and historical biogeography of the genus Tuber, the ‘true truffles’. J Biogeogr 35:815–829
Johnson D, Martin F, Cairney JWG, Anderson IC (2012) The importance of individuals: intraspecific diversity on mycorrhizal plants and fungi in ecosystems. New Phytol 194:614–628
Jourand P, Ducousso M, Reid R, Majorel C, Richert C, Riss J, Lebrun M (2010) Nickel-tolerant ectomycorrhizal Pisolithus albus ultramafic ecotype isolated from nickel mines in New Caledonia strongly enhance growth of a host plant at toxic nickel concentrations. Tree Physiol 30:1311–1319
Le Tacon F (2011) Point de vue sur Tuber aestivum Vittad. Le Trufficulteur 74:13–17
Le Tacon F, Marçais B, Courvoisier M, Murat C, Becker M (2014) Climatic variations explain annual fluctuations in French Périgord black truffle wholesale markets but does not explain the decrease in black truffle production over the last 48 years. Mycorrhiza 24(1 supplement):115–125
Luikart G, Cornuet JM (1998) Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv Biol 12:228–237
Luikart G, Allendorf FW, Cornuet JM, Sherwin WB (1998a) Distortion of allele frequency distributions provides a test for recent population bottlenecks. J Hered 89:238–247
Luikart G, Sherwin WB, Steele B, Allendorf FW (1998b) Usefulness of molecular markers for detecting population bottlenecks and monitoring genetic change. Mol Ecol 7:963–974
Mello A, Cantisani A, Vizzini A, Bonfante P (2002) Genetic variability of Tuber uncinatum and its relatedness to other black truffles. Environ Microbiol 4:584–594
Merényi Z, Varga T, Geml J, Orczán Ákos K, Chevalier G, Bratek Z (2014) Phylogeny and phylogeography of Tuber brumale aggr. Mycorrhiza. doi:10.1007/s00572-014-0566-7
Molinier V, Murat C, Morin E, Gollotte A, Wipf D, Martin F (2013a) First identification of polymorphic microsatellite markers in the Burgundy truffle, Tuber aestivum (Tuberaceae). App Plant Sci 1:1200220
Molinier V, van Tuinen D, Chevalier G, Gollotte A, Wipf D, Redecker D (2013b) A multigene phylogeny demonstrates that Tuber aestivum and Tuber uncinatum are conspecific. Org Divers Evol 13:503–512
Molinier V, Murat C, Frochot H, Wipf D, Splivallo R (2015) Fine-scale spatial genetic structure analysis of the black truffle Tuber aestivum and its link to aroma variability. Environ Microbiol. (accepted) http://onlinelibrary.wiley.com/doi/10.1111/1462-2920.12910/abstract
Montecchi A, Sarasini M (2000) Funghi Ipogei d’Europa. AMB, Brescia Italy
Mouches C, Duthil P, Poitou N, Delmas J, Bove J (1981) Caractérisation des espèces truffières par analyse de leurs protéines en gels de polyacrylamide et application de ces techniques à la taxonomie des champignons. Mushroom Science 11:819–831
Murat C (2015) Forty years of inoculating seedlings with truffle fungi: past and future perspectives. Mycorrhiza 25:77–81
Murat C, Diez J, Luis P, Delaruelle C, Dupre C, Chevalier G et al (2004) Polymorphism at the ribosomal DNA ITS and its relation to postglacial re-colonization routes of the Perigord truffle Tuber melanosporum. New Phytol 164:401–411
Murat C, Riccioni C, Belfiori B, Cichocki N, Labbe J, Morin E et al (2011) Distribution and localization of microsatellites in the Perigord black truffle genome and identification of new molecular markers. Fungal Genet Biol 48:592–601
Murat C, Rubini A, Riccioni C, De la Varga H, Akroume E, Belfiori B et al (2013) Fine scale spatial genetic structure of the black truffle (Tuber melanosporum) investigated with neutral microsatellites and functional mating type genes. New Phytol 199:176–187
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci U S A 70:3321–3323
Okuda Y, Shimomura N, Funato C, Nagasawa E, Matsumoto T (2013) Genetic variation among natural isolates of the ectomycorrhizal hypogenous fungus, Rhizopogon roseolus from Japanese pine forests inferred using AFLP markers. Mycoscience 54:13–18
Pacioni G, Pomponi G (1991) Genotypic patterns of some Italian populations of the Tuber aestivum-Tuber mesentericum complex. Mycotaxon 42:171–179
Pacioni G, Frizzi G, Miranda M, Visca C (1993) Genetics of a Tuber aestivum population (Ascomycotina, Tubérales). Mycotaxon 47:93–100
Paolocci F, Rubini A, Riccioni C, Topini F, Arcioni S (2004) Tuber aestivum and Tuber uncinatum: two morphotypes or two species? FEMS Microbiol Lett 235:109–115
Paolocci F, Rubini A, Riccioni C, Arcioni S (2006) Reevaluation of the life cycle of Tuber magnatum. Appl Environ Microbiol 72:2390–2393
Payen T, Murat C, Bonito G (2014) Truffle phylogenomics: new insights into truffle evolution and truffle life cycle. In Francis M. Martin editor Adv Bot Res Vol. 70, Oxford: United Kingdom, 211–234
Payen T, Murat C, Gigant A, Morin E, De Mita S, Martin F (2015) A survey of genome-wide single nucleotide polymorphisms through genome re-sequencing in the Périgord black truffle (Tuber melanosporum Vittad.). Mol Ecol Resources (in press)
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Piry S, Luikart G, Cornuet JM (1999) Computer note. BOTTLENECK: a computer program for detecting recent reductions in the effective size using allele frequency data. J Hered 90:502–503
Pomarico M, Figliuolo G, Rana GL (2007) Tuber spp. biodiversity in one of the southernmost European distribution areas. Biodivers Conserv 16:3447–3461
Riccioni C, Belfiori B, Rubini A, Passeri V, Arcioni S, Paolocci F (2008) Tuber melanosporum outcrosses: analysis of the genetic diversity within and among its natural populations under this new scenario. New Phytol 180:466–478
Riousset L, Riousset G, Chevalier G, Bardet MC (2001) Truffes d’Europe et de chine. INRA Editions, Paris, France
Rivera Y, Burchhardt KM, Kretzer AM (2014) Little to no genetic structure in the ectomycorrhizal basidiomycete Suillus spraguei (Syn. S. pictus) across parts of the northeastern USA. Mycorrhiza 24:227–232
Rubini A, Topini F, Riccioni C, Paolocci F, Arcioni S (2004) Isolation and characterization of polymorphic microsatellite loci in white truffle (Tuber magnatum). Mol Ecol Notes 4:116–118
Rubini A, Paolocci F, Riccioni C, Vendramin GG, Arcioni S (2005) Genetic and phylogeographic structures of the symbiotic fungus Tuber magnatum. Appl Environ Microbiol 71:6584–6589
Rubini A, Belfiori B, Riccioni C, Tisserant E, Arcioni S, Martin F, Paolocci F (2011) Isolation and characterization of MAT genes in the symbiotic ascomycete Tuber melanosporum. New Phytol 189:710–722
Seager R, Ting M, Held I, Kushnir Y, Lu J, Vecchi G, Huang HP, Harnik N, Lau NC, Li C et al (2007) Model projections on an imminent transition to a more arid climate in southwestern North America. Science 316:1181–1184
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Academic, London
Splivallo R, Valdez N, Kirchhoff N, Ona MC, Schmidt JP, Feussner I, Karlovsky P (2012) Intraspecific genotypic variability determines concentrations of key truffle volatiles. New Phytol 194:823–835
Stenberg P, Lundmark M, Saura A (2003) MLGsim: a program for detecting clones using a simulation approach. Mol Ecol Notes 3:329–331
Sterl A, Severijns C, Dijkstra H, Hazeleger W, Oldenborgh G, Broeke M, Burgers G, Hurk B, Leeuwen P, Velthoven P (2008) When can we expect extremely high surface temperatures? Geophys Res Lett. doi:10.1029/2008GL034071
Stobbe U, Büntgen U, Sproll L, Tegel W, Egli S, Fink S (2012) Spatial distribution and ecological variation of re-discovered German truffle habitats. Fungal Ecol 5:591–599
Stobbe U, Egli S, Tegel W, Peter M, Sproll L, Büntgen U (2013) Potential and limitations of Burgundy truffle cultivation. Appl Microbiol Biotechnol 97:5215–5224. doi:10.1007/s00253-013-4956-0
Taylor JW, Jacobson DJ, Kroken S, Kasuga T, Geiser DM, Hibbett DS, Fisher MC (2000) Phylogenetic species recognition and species concepts in fungi. Fungal Genet Biol 31:21–32
Thioulouse J, Dray S (2007) Interactive multivariate data analysis in R with the ade4 and ade4TkGUI packages. J Stat Softw 22:1–14
Thioulouse J, Chessel D, Doledec S, Olivier JM (1997) ADE-4: a multivariate analysis and graphical display software. Stat Comput 7:75–83
Turesson G (1922) The genotypical response of the plant species to the habitat. Hereditas 3:211–350
Urbanelli S, Sallicandro P, De Vito E, Bullini L, Biocca E (1998) Biochemical systematics of some species in the genus Tuber. Mycologia 90:537–546
Vellinga EC, Wolfe BE, Pringle A (2009) Global patterns of ectomycorrhizal introductions. New Phytol 181:960–973
Vincenot L, Nara K, Sthultz C, Labbe J, Dubois MP, Tedersoo L et al (2012) Extensive gene flow over Europe and possible speciation over Eurasia in the ectomycorrhizal basidiomycete Laccaria amethystina complex. Mol Ecol 21:281–299
Vittadini C (1831) Monographia tuberacearum. France
Wadud MA, Nara K, Lian C, Ishida TA, Hogetsu T (2014) Genet dynamics and ecological functions of the pioneer ectomycorrhizal fungi Laccaria amethystina and Laccaria laccata in a volcanic desert on Mount Fuji. Mycorrhiza 24:551–563
Weden C, Danell E, Tibell L (2005) Species recognition in the truffle genus Tuber—the synonyms Tuber aestivum and Tuber uncinatum. Environ Microbiol 7:1535–1546
Acknowledgments
The work presented was supported by the ‘Conseil Régional de Bourgogne (Programme Jeune Chercheur Entrepreneur; Grant 20100112095254682–1)’, the ‘Ernst Göhner Stiftung’ and the Swiss State Secretariat for Education, Research and Innovation (SERI; COST Action FP1203). The UMR1136 is supported by a grant overseen by the French National Research Agency (ANR) as part of the ‘Investissements d'Avenir’ programme (ANR-11-LABX-0002-01, Lab of Excellence ARBRE). The authors thank Denise Stalder, Gérard Chevalier, Henri Frochot, Jean-Claude Ferrand, Mr. Déquéant, Mr. Besson, Willy Tegel and all other truffle growers for providing samples. We are grateful to Falbien Halkett for the help in the population genetic analysis, the choice of the tests and their interpretation. We thank Marie-Lara Bouffaud, François Le Tacon, Francis Martin and Stéphane De Mita for providing constructive advice and helpful discussions. We would like to thank Andrea Rubini, Claudia Riccioni and two anonymous reviewers for helpful comments on the manuscript and Aimée Orsini for English language editing. The authors also thank the strategic plateform ‘GENTYANE’ INRA, Ibisa 2009 and its group leader Mr. Poncet.
Author contributions
V.M., C.M. and D.W. designed the experiment. V.M. and C.M. performed experiment and data analyses. B.M. and H.D.V. provided assistance with DNA extraction, SSR amplification and analysis. V.M. and C.M. wrote the manuscript. M.P., A.G., S.E., B.B., F.P. and D.W. contributed to the writing and provided samples. All co-authors approved the final manuscript.
Conflict of interest
The authors declare that they have no competing interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Virginie Molinier and Claude Murat both authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Molinier, V., Murat, C., Peter, M. et al. SSR-based identification of genetic groups within European populations of Tuber aestivum Vittad. Mycorrhiza 26, 99–110 (2016). https://doi.org/10.1007/s00572-015-0649-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-015-0649-0