Abstract
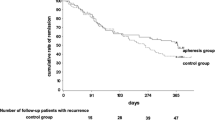
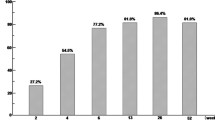
Background. Recently, granulocyte and monocyte adsorption apheresis (GMA) has been shown to be effective for active ulcerative colitis (UC). Its original weekly treatment schedule is effective in about 70% of active UC. However, it takes about 3–4 weeks to achieve remission, and the efficacy of a more frequent treatment schedule has not been elucidated yet. We performed a pilot open-labeled prospective, randomized, controlled study comparing weekly and an intensive treatment schedule with three treatment sessions per week in the first 2 weeks. Methods. Thirty active UC patients with moderate disease activity were prospectively and randomly assigned to receive the original or the intensive treatment schedule for a total of ten sessions. The proportion of the patients achieving remission and the time to achieve remission among them was compared between the two groups. The incidences of adverse effects were also compared between the two groups. Results. The rate of inducing remission in the original and intensive treatment group was 66.7% and 80%, respectively (P = 0.25, NS). The time to achieve remission was 27.2 days in the original group and 10.7 days in the intensive group (P = 0.04). Adverse effects were observed in two patients in each groups (NS). Conclusions. Intensive treatment with GMA is an efficacious and safe treatment for active UC. Because it induces rapid remission, it may be a more ideal treatment regimen than the conventional weekly treatment.
Similar content being viewed by others
References
Podolsky DK. Inflammatory bowel disease. N Engl J Med 2002;347:417–29.
Hibi T, Ogata H. Novel pathophysiological concepts of inflammatory bowel disease. J Gastroenterol 2006;41:10–6.
Hibi T, Sakuraba A. Is there a role for apheresis in gastrointestinal disorders? Nat Clin Pract Gastroenterol Hepatol 2005;2:200–1.
Bicks RO, Groshart KD. The current status of T-lymphocyte apheresis (TLA) treatment of Crohn’s disease. J Clin Gastroenterol 1989;11:136–8.
Shimoyama T, Sawada K, Hiwatashi N, Sawada T, Matsueda K, Munakata A, et al. Safety and efficacy of granulocyte and monocyte adsorption apheresis in patients with active ulcerative colitis: a multicenter study. J Clin Apher 2001;16:1–9.
Suzuki Y, Yoshimura N, Saniabadi AR, Saito Y. Selective granulocyte and monocyte adsorptive apheresis as a first-line treatment for steroid naive patients with active ulcerative colitis: a prospective uncontrolled study. Dig Dis Sci 2004;49:565–71.
Hanai H, Watanabe F, Takeuchi K, Iida T, Yamada M, Iwaoka Y, et al. Leukocyte adsorptive apheresis for the treatment of active ulcerative colitis: a prospective, uncontrolled, pilot study. Clin Gastroenterol Hepatol 2003;1;28–35.
Naganuma M, Funakoshi S, Sakuraba A, Takagi H, Inoue N, Ogata H, et al. Granulocytapheresis is useful as an alternative therapy in patients with steroid-refractory or-dependent ulcerative colitis. Inflamm Bowel Dis 2004;10:251–7.
Fukuda Y, Matsui T, Suzuki Y, Kanke K, Matsumoto T, Takazoe M, et al. Adsorptive granulocyte and monocyte apheresis for refractory Crohn’s disease: an open multicenter prospective study. J Gastroenterol 2004;39:1158–64.
Nagashima M, Yoshino S, Tanaka H, Yoshida N, Kashiwagi N, Saniabadi AR. Granulocyte and monocyte apheresis suppresses symptoms of rheumatoid arthritis: a pilot study. Rheumatol Int 1998;18:113–8.
Garland CF, Lilienfeld AM, Mendeloff AI, Markowitz JA, Terrell KB, Garland FC. Incidence rates of ulcerative colitis and Crohn’s disease in fifteen areas of the United States. Gastroenterology 1981;81:1115–24.
Lichtiger S, Present DH, Kornbluth A, Gelernt I, Bauer J, Galler G, et al. Cyclosporine in severe ulcerative colitis refractory to steroid therapy. N Engl J Med 1994;330:1841–5.
Guyatt G, Mitchell A, Irvine EJ, Singer J, Williams N, Goodacre R, et al. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology 1989;96:804–10.
Baron JH, Connell AM, Lennard-Jones JE. Variation between observers in describing mucosal appearances in proctocolitis. Br Med J 1964;1:89–92.
Mahida YR. The key role of macrophages in the immunopathogenesis of inflammatory bowel disease. Inflamm Bowel Dis 2000;6:21–33.
Lampinen M, Carlson M, Sangfelt P, Taha Y, Thorn M, Loof L, et al. IL-5 and TNF-alpha participate in recruitment of eosinophils to intestinal mucosa in ulcerative colitis. Dig Dis Sci 2001;46:2004–9.
Kashiwagi N, Nakano M, Saniabadi AR, Adachi M, Yoshikawa T. Anti-inflammatory effect of granulocyte and monocyte adsorption apheresis in a rabbit model of immune arthritis. Inflammation 2002;26:199–205.
Kashiwagi N, Sugimura K, Koiwai H, Yamamoto H, Yoshikawa T, Saniabadi AR, et al. Immunomodulatory effects of granulocyte and monocyte adsorption apheresis as a treatment for patients with ulcerative colitis. Dig Dis Sci 2002;47:1334–41.
Sawada K, Kusugami K, Suzuki Y, Bamba T, Munakata A, Hibi T, et al. Leukocytapheresis in ulcerative colitis: results of a multicenter double-blind prospective case-control study with sham apheresis as placebo treatment. Am J Gastroenterol 2005;100:1362–9.
Muratov V, Lundahl J, Ulfgren AK, Elvin K, Fehrman I, Ahlborg N, et al. Down-regulation of interferon-gamma parallels clinical response to selective leukocyte apheresis in patients with inflammatory bowel disease: a 12-month follow-up study. Int J Colorectal Dis 2006;21:493–504.
Domenech E, Hinojosa J, Esteve-Comas M, Gomollon F, Herrera JM, Bastida G, et al. Granulocyte aphaeresis in steroid-dependent inflammatory bowel disease: a prospective, open, pilot study. Aliment Pharmacol Ther 2004;20:1347–52.
Rowe FA, Walker JH, Karp LC, Vasiliauskas EA, Plevy SE, Targan SR. Factors predictive of response to cyclosporin treatment for severe, steroid-resistant ulcerative colitis. Am J Gastroenterol 2000;95:2000–8.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sakuraba, A., Sato, T., Naganuma, M. et al. A pilot open-labeled prospective randomized study between weekly and intensive treatment of granulocyte and monocyte adsorption apheresis for active ulcerative colitis. J Gastroenterol 43, 51–56 (2008). https://doi.org/10.1007/s00535-007-2129-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-007-2129-6