Abstract
The common bile duct and the main pancreatic duct open into the duodenum, where they frequently form a common channel. The sphincter of Oddi is located at the distal end of the pancreatic and bile ducts; it regulates the outflow of bile and pancreatic juice. In patients with a pancreaticobiliary maljunction, the action of the sphincter does not functionally affect the junction. Therefore, in these patients, two-way regurgitation (pancreatobiliary and biliopancreatic reflux) occurs. This results in various pathological conditions of the biliary tract and the pancreas. Biliopancreatic reflux could be confirmed by: operative or postoperative T-tube cholangiography; CT combined with drip infusion cholangiography; histological detection of gallbladder cancer cells in the main pancreatic duct; and reflux of bile on the cut surface of the pancreas. Biliopancreatic reflux occurs frequently in patients with a long common channel. Although the true prevalence, degree, and pathophysiology of biliopancreatic reflux remain unclear, biliopancreatic reflux is related to the occurrence of acute pancreatitis. Obstruction of a long common channel easily causes bile flow into the pancreas. Even if no obstruction is present, biliopancreatic reflux can still result in acute pancreatitis in some cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The common bile duct and the main pancreatic duct open into the duodenum where they frequently form a common channel. The sphincter of Oddi is located at the distal end of the pancreatic and bile ducts; it regulates the outflow of bile and pancreatic juice. In patients with pancreaticobiliary maljunction (PBM), the common channel can be so long that the junction of the pancreatic and bile ducts is located outside of the duodenal wall. Since the action of the sphincter does not functionally affect such a junction, two-way regurgitation (pancreatobiliary and biliopancreatic reflux) occurs. This results in various pathological conditions of the biliary tract and the pancreas [1–3].
Given that the hydropressure within the pancreatic duct is usually greater than the hydropressure within the bile duct, pancreatic juice frequently refluxes into the biliary duct (pancreatobiliary reflux) [4–6]. The stagnation of activated pancreatic juice mixed with bile may injure the epithelium of the biliary tract and induce metaplasia or promote cancer progression. The biliary mucosa of PBM patients can be considered to be a premalignant region, given the high incidence of biliary cancer and increased cell proliferation with K-ras mutations in such patients [7]. Once PBM is diagnosed, it is recommended that prophylactic biliary surgery be done before malignant changes occur in the biliary tract [8, 9].
On the other hand, biliopancreatic reflux was previously noted as an etiological factor related to acute pancreatitis [10]. However, the relationship between biliopancreatic reflux and acute pancreatitis is controversial [4–6, 11, 12]. This paper reviews the recent advances in the diagnosis of biliopancreatic reflux, as well as its pathophysiology and clinical implications.
Anatomy of the pancreaticobiliary junction
There are three ways that the main pancreatic duct and common bile duct open into the duodenum: separate openings, one opening without a common channel; and one opening with a common channel [13, 14]. The incidence of common channel formation ranges from 55 [15] to 82% [13, 14]. Dowdy et al. [16] reported that the length of the common channel ranged from 1 to 12 mm, with an average length of 4.4 mm; Sterling [15] reported that the length of the common channel ranged from 1.2 to 8.4 mm, with an average of 4.4 mm. Rienhoff and Pickrell [17] reported that 92 (53%) of 173 cases had a common channel up to 2 mm long, 62 (36%) had a common channel ranging from 3 to 5 mm long, and 19 (11%) had a common channel that was >6 mm long.
The sphincter of Oddi, which is composed of the sphincter choledochus, the sphincter pancreaticus and the sphincter ampullae, is located at the distal end of the pancreatic and bile ducts. It regulates the outflow of bile and pancreatic juice [13, 14]. The common channel can be so long that the junction of the pancreatic and bile ducts is located outside of the duodenal wall; this occurs in PBM. In such patients, the action of the sphincter does not functionally affect the junction, and this results in two-way regurgitation.
Diagnosis of biliopancreatic reflux
Pancreatobiliary reflux is diagnosed based on the presence of an elevated amylase level in bile obtained intraoperatively, through an indwelling T-tube, or during endoscopic retrograde cholangiopancreatography (ERCP). The bile amylase level in PBM patients is usually markedly elevated [1, 18]. On secretin-stimulated dynamic magnetic resonance cholangiopancreatography (MRCP), pancreatobiliary reflux in PBM patients can be visualized as preferential filling of the long common channel and the biliary tract with minimal duodenal filling [19, 20]. Biliopancreatic reflux occurs in many patients who have pancreatobiliary reflux.
Operative or postoperative T-tube cholangiography
The reflux of dye or contrast medium into the pancreatic duct during operative cholangiography in patients with a normal pancreaticobiliary junction has been known for a long time. Several published studies have dealt with this subject [21, 22]. However, they have not examined the effect of variables, such as the rate of injection, the pressure of the injection, the type of anesthesia, and the use of premedicating drugs. Under fluoroscopic observation, a small amount of contrast medium is gradually injected using the least possible pressure. After the lower common bile duct is filled, several spot films of the periampullary regions are taken. Reflux of contrast medium into the pancreatic duct can only occur when the sphincter of Oddi is relaxed when there is a common channel, and, at the same time, the medium flows into the duodenum. Whenever there is increased flow through the common channel and the amupulla, the duodenum will be filled and there will be reflux into the pancreatic duct, provided that the sphincter of Oddi is relaxed and the intraampullary pressure is high. Contraction of the sphincter makes pancreatic reflux more difficult due to the local anatomy (Fig. 1) [23]. However, in patients with PBM with confluence of the pancreatic and bile duct high above the sphincter, pancreatic reflux can occur also when the sphincter contracts.
Schematic illustration showing mechanism of biliopancreatic reflux, usual out-flow, pancreatobiliary reflux, and duodenopancreatic reflux [23]
In general, no stasis of contrast fluid is noted in the pancreatic duct. There is considerable individual variation in the length of reflux down the pancreatic duct. In patients with a normal pancreaticobiliary junction, the incidence of pancreatic reflux has been reported to be 12.5% (15/120) [24], 13.3% (13/98) [25], 15.4% (31/211) [26], 18.7% (115/614) [27], and 27.6% (69/250) [28].
CT combined with drip infusion cholangiography
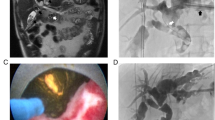
Multidetector row computed tomography (CT) combined with drip infusion cholangiography (DIC) can be used to demonstrate more details of the anatomy of the biliary tract. After intravenous injection, meglumine iotroxate, the contrast medium used in DIC, is absorbed by hepatocytes and excreted into the bile. Bile duct imaging using this method is considered to reflect the dynamic and physiological bile flow better than operative cholangiography. On DIC-CT, reflux of the contrast medium into the main pancreatic duct can be demonstrated in patients with PBM [29]. Sugiyama et al. [30] demonstrated that, on DIC-CT, biliopancreatic reflux in a patient with mild acute pancreatitis associated with PBM was enhanced by gallbladder contraction that occurred after yolk ingestion. Fumino et al. [31] reported that biliopancreatic reflux was observed on DIC-CT in 6 (40%) of 15 patients with PBM. We noted biliopancreatic reflux on DIC-CT in a 17-year-old male with recurrent mild acute pancreatitis and an 8-mm-long common channel; in this patient, the communication of the pancreatic and bile ducts was occluded when the sphincter contracted (Figs. 2 and 3).
Multidetector row CT combined with drip infusion cholangiography of a 17-year-old male with recurrent mild acute pancreatitis, showing reflux of contrast medium into the main pancreatic duct. [8] On ERCP, high confluence of pancreatobiliary ducts with an 8-mm-long common channel was observed in this patient
Post operative T-tube cholangiography of the same patient as Fig. 2. a The pancreatic duct was demonstrated through the common channel (arrow) when the sphincter of Oddi was relaxed. b The communication between the bile and pancreatic ducts was destroyed when the sphincter was contracted
Histopathological detection of gallbladder cancer cells in the main pancreatic duct
Suda and Miyano [32] noted squamous cancer cells in the main pancreatic duct in a patient with adenosquamous cell cancer of the gallbladder who had PBM; they suspected the possibility that bile refluxed into the pancreatic duct.
Reflux of the bile on the cut surface of the pancreas
Dohke et al. [33] reported a pancreatic cancer case in whom reflux of bile was observed spontaneously on the cut surface of the pancreas during distal pancreatectomy; biliopancreatic reflux was detected on operative cholangiography, but the patient had a very short common channel.
Pathophysiology and clinical implications of biliopancreatic reflux
The clinical implications of biliopancreatic reflux in patients with a normal pancreaticobiliary junction remain unclear. Nevertheless, it has been proposed that biliopancreatic reflux is related to acute pancreatitis.
Gallstone migration through the major duodenal papilla sometimes causes acute pancreatitis [34]. However, the mechanism whereby biliary calculi trigger acute pancreatitis is not known. As early as 1901, Opie [10] detailed the pathological findings of two patients who died from hemorrhagic necrosis of the pancreas; both had a gallstone impacted in the major papilla. Opie [10] suggested that the formation of a communication between the bile duct and the pancreatic duct at the papilla was the triggering event; bile could thus flow from the biliary tract into the pancreas and this would result in disease. In addition, experimental studies have documented that bile aspirated from the canine gallbladder and injected into the main pancreatic duct produces severe hemorrhagic necrosis of the pancreas. This hypothesis was referred to as the “common channel theory”, and it became quite popular. However, Opie’s conclusions were challenged based on the following: (1) pancreatic duct pressure is normally higher than choledochal pressure, so that pancreatic juice would be expected to flow into the biliary tract rather than bile to flow into the pancreatic duct [4–6]; (2) many cases have a common channel that is too short to permit bile to flow into the pancreas behind an impacted stone; and (3) injection of bile into the pancreatic duct does not initiate hemorrhagic necrotizing pancreatitis [35].
As reflux of the bile into the pancreatic duct can sometimes take place and yet the incidence of pancreatitis was very low, reflux of the bile into the pancreatic duct under normal anatomical system might be a physiologic process and seldom productive of inflammatory changes within the pancreas. However, Armstrong and Taylor [26] reported a high incidence of biliopancreatic reflux on operative cholangiography of patients with previous acute gallstone pancreatitis (62.3% (33/53) with a history of acute gallbladder pancreatitis vs. 14.6% (82/561) in those with no previous history of acute gallbladder pancreatitis; P < 0.0001). It is also of interest that 72% of the patients with a history of pancreatitis and biliopancreatic reflux were found to have a common channel that was 5 mm or longer, while only 20% of those without pancreatitis had a common channel that was 5 mm or longer. Howel and Bergh [36] found a positive correlation between biliopancreatic reflux of contrast medium and subsequent serum amylase elevations following cholecystectomy and operative cholangiography. They noted that injection of the bile into the pancreatic duct produced marked hyperamylasemia. Based on experiments that mimicked different patterns of stone impaction at the choledochoduodenal junction in rabbits, Arendt et al. [37] reported that biliopancreatic reflux and acute pancreatitis might be due to a combination of stone migration effects, such as biliary hypertension, temporary pancreatic duct obstruction, and bacterial infection of bile. Given these findings, biliopancreatic reflux may be considered a potential pathogenic mechanism that initiates acute biliary pancreatitis in patients who have both a common channel and a bacterial infection of their choledochal secretions.
Acute pancreatitis associated with pancreaticobiliary maljunction
Since fluid pressure is usually higher within the pancreatic duct than within the bile duct, pancreatic juice frequently refluxes into the bile duct [4–6]. However, bile may reflux into the pancreatic duct via PBM under some circumstances, such as during bile stasis in a choledochal cyst or a common channel, as well as in patients with cholangitis [38]. Refluxed bile may activate pancreatic enzymes, particularly phospholipase A2, which may result in cause acute pancreatitis [39, 40].
The frequency of acute pancreatitis in children with a choledochal cyst is reportedly as high as 68% [41] and ranges from 18 [42] to 23% [43] in adults. Among our 107 PBM patients [44–46], four had acute pancreatitis; two of the patients were children, and the other two were elderly. On cholangiography, three of these four patients were found to have a congenital choledochal cyst. Although in three patients the acute pancreatitis was mild and in one patient it was severe, all cases resolved with conservative treatment. Three of the patients who had excision of the extrahepatic bile duct with biliary diversion did not develop further episodes of pancreatitis. In the one patient with acute relapsing pancreatitis, a small radiolucent defect was seen in a dilated long common channel on ERCP during injection of contrast medium. Kaneko et al. [47] reported that radiolucent filling defects composed of protein plugs were detected in a long common channel in 40% of 55 patients with a choledochal cyst; in 82% of cases, the protein plugs disappeared spontaneously or after flushing. During an attack of acute pancreatitis, a transient increase in intraductal pressure in the pancreatic duct may occur. This increased pressure is probably due to the presence of bile reflux into the pancreatic duct that may occur when a long common channel is temporarily obstructed by plugs.
Acute pancreatitis associated with high confluence of pancreaticobiliary ducts
There are cases that have a relatively long common channel that are not classified as having PBM, because their sphincter of Oddi includes the pancreaticobiliary ductal junction. To investigate the clinical significance of a relatively long common channel, high confluence of pancreaticobiliary ducts (HCPBD) was defined as a common channel ≥6 mm, in which the communication was occluded when the sphincter was contracted [48–50]. Among 3,300 patients who had ERCP in our hospital, 62 patients were diagnosed as having HCPBD. Reflux of contrast medium into the pancreatic duct was detected in 86% of HCPBD patients who had postoperative T-tube cholangiography. The bile amylase level in all HCPBD patients examined was markedly elevated, and 10% of these HCPBD patients also had gallbladder cancer. Thus, in HCPBD patients, as in PBM patients, pancreatobiliary, and biliopancreatic reflux appears to occur sometimes.
Among our patients, acute pancreatitis occurred in 17 (27%) of 62 HCPBD patients. Acute pancreatitis was attributed to biliary tract stones in 10 patients and to alcohol abuse in three patients. Given Opie’s common channel theory [10], in which obstruction of outflow below the common bile duct and the pancreatic duct would permit bile flow into the pancreas, acute biliary pancreatitis would seem to occur more readily in HCPBD patients with a relatively long common channel. In the remaining four HCPBD patients with no history of excessive alcohol intake or biliary stones, acute pancreatitis might have been induced in a manner similar to that which occurs in PBM patients, that is due to biliopancreatic reflux via a relatively long common channel (Figs. 2 and 3). The rate of pancreatitis was different in patients with PBM (4%) and HCPBD (28%). PBM was suspected based on the findings of imaging modalities such as magnetic resonance cholangiopancreatography or computed tomography. However, HCPBD could only be diagnosed on ERCP that was done to elucidate certain cholangiopancreatic features. Therefore, the highly selective nature of this HCPBD patient population is likely to have affected our results; the actual risk of acute pancreatitis in HCPBD patients may actually be less than that noted in our patients.
Conclusions
Biliopancreatic reflux can be examined using various methods. Biliopancreatic reflux can occur frequently in PBM or HCPBD patients; it can also occur in some individuals without these conditions. The true prevalence, degree, and precise pathophysiology of biliopancreatic reflux remain unclear. Nevertheless, biliopancreatic reflux is thought to be related to the occurrence of acute pancreatitis. The obstruction of a long common channel can easily result in bile flowing into the pancreas. In some cases, even with no obstruction, biliopancreatic reflux can still induce acute pancreatitis.
References
The Japanese Study Group on Pancreaticobiliary Maljunction. Diagnostic criteria of pancreaticobiliary maljunction. J Hepatobiliary Pancreat Surg. 1994;1:219–21.
Kamisawa T, Egawa N, Nakajima H, Tsuruta K, Okamoto A, Matsukawa M. Origin of the long common channel based on pancreatographic findings in pancreaticobiliary maljunction. Dig Liver Dis. 2005;37:363–7.
Kamisawa T, Tu Y, Nakajima H, Egawa N, Tsuruta K, Okamoto A. The presence of a common channel and associated pancreaticobiliary disease: a prospective ERCP study. Dig Liver Dis. 2007;39:173–9.
Csendes A, Kruse A, Funch-Jensen P, Oster MJ, Omsholt J, Amdrup E. Pressure measurements in the biliary and pancreatic duct system in controls and patients with gallstones, previous cholecystectomy, or common bile duct stones. Gastroenterology. 1979;77:1203–10.
Carr-Locke D, Gregg JA. Endoscopic manometry of pancreatic and biliary sphincter zones in man. Basal results in healthy volunteers. Dig Dis Sci. 1981;26:7–15.
Arendt T, Stoffregen C, Kloehn S, Monig H, Nizze H, Folsch UR. Santorini’s duct-risk factor for acute pancreatitis or protective morphologic variant? Experiments in rabbits. Eur J Gastroenterol Hepatol. 1997;9:569–73.
Tanno S, Obara T, Fujii T, Mizukami Y, Shudo R, Nishino N, et al. Proliferative potential and K-ras mutation in epithelial hyperplasia of the gallbladder in patients with anomalous pancreaticobiliary ductal union. Cancer. 1998;83:267–75.
Kamisawa T, Okamoto A. Biliopancreatic and pancreatobiliary refluxes in cases with and without pancreaticobiliary maljunction: diagnosis and clinical implications. Digestion. 2006;73:228–36.
Seki M, Yanagisawa A, Ninomiya E, Ninomiya Y, Ohta H, Saiyra A, et al. Clinicopathology of pancreaticobiliary maljunction: relationship between alterations in background biliary epithelium and neoplastic development. J Hepatobiliary Pancreat Surg. 2005;12:254–62.
Opie EL. The etiology of acute hemorrhagic pancreatitis. Bull Johns Hopkins Hosp. 1901;12:484–787.
Arendt T. The pathogenesis of acute biliary pancreatitis: a controversial issue. Part I. The concept of biliopancreatic reflux. Gastroenterol J. 1989;49:50–3.
Senninger N. Bile-induced pancreatitis. Eur Surg Res. 1992;24(Suppl 1):68–73.
Suda K, Miyano T, Hashimoto K. The choledocho-pancreatico-ductal junction in infantile obstructive jaundice diseases. Acta Pathol Jpn. 1980;30:187–94.
Suda K, Miyano T, Konuma I, Matsumoto M. An abnormal pancreatico-choledocho-ductal junction in cases of biliary tract carcinoma. Cancer. 1983;52:2086–8.
Sterling JA. The common channel for bile and pancreatic ducts. Surg Gynecol Obstet. 1954;98:420–4.
Dowdy GS, Waldron GW, Brown WG. Surgical anatomy of the pancreaticobiliary ductal system. Arch Surg. 1962;84:229–46.
Rienhoff WF, Pickrell KL. Pancreatitis: an anatomic study of the pancreatic and extrahepatic biliary systems. Arch Surg. 1945;51:205–19.
Davenport M, Stringer MD, Howard ER. Biliary amylase and congenital choledochal dilatation. J Pediatr Surg. 1995;30:474–7.
Matos C, Nicaise N, Deviere J, Cassart M, Metens T, Struyven T, et al. Choledochal cysts: comparison of findings at MR cholangiopancreatography and endoscopic retrograde cholangiopancreatography in eight patients. Radiology. 1998;209:443–8.
Hosoki T, Hasuike Y, Takeda Y, Michita T, Watanabe Y, Sakamori R, et al. Visualization of pancreaticobiliary reflux in anomalous pancreaticobiliary junction by secretin-stimulated dynamic magnetic resonance cholangiopancreatography. Acta Radiol. 2004;45:375–82.
Whitaker PH, Parkinson EG, Hughes JH. Television fluoroscopy for operative cholangiography. An analysis of 150 cases. Clin Radiol. 1968;19:368–78.
Duschieri A, Hughes JH, Cohen M. Manometric choledochography during biliary surgery. Ann R Coll Surg Engl. 1971;48:5.
Yokoyama Y. Cinecholangiographic study of the reflux of bile into the pancreatic duct. Nippon Shokaki Geka Gakkai Zasshi. 1986;19:1009–19. (in Japanese).
Paulino-Netto A, Paulino F. Pancreatic ductal reflux. Am J Dig Dis. 1963;8:666–72.
Fujisaki S, Tomita R, Koshinaga T, Fukuzawa M. Analysis of pancreaticobiliary ductal union based on intraoperative cholangiography in patients undergoing laparoscopic cholecystectomy. Scand J Gastroenterol. 2002;37:956–9.
Armstrong CP, Taylor TV. Pancreatic-duct reflux and acute gallstone pancreatitis. Ann Surg. 1986;204:59–64.
Cuschieri A, Hughes JH. Pancreatic reflux during operative choledochography. Brit J Surg. 1973;60:933–6.
Heloury Y, Leborgne J, Rogez JM, Robert R, Lehur PA, Pannier M, et al. Radiological anatomy of the bile ducts based on intraoperative investigation in 250 cases. Anat Clin. 1985;7:93–102.
Nambu A, Ichikawa T, Katoh K, Araki T. A case of abnormal pancreaticobiliary junction evidenced by 3D drip infusion cholangiography CT. J Comput Assist Tomogr. 2001;25:653–5.
Sugiyama M, Haradome H, Takahara T, Izumisato Y, Abe N, Masaki T, et al. Biliopancreatic reflux via anomalous pancreaticobiliary junction. Surgery. 2004;135:457–9.
Fumino S, Tokiwa K, Katoh T, Ono S, Iwai N. New insight into bile flow dynamics in anomalous arrangement of the pancreaticobiliary duct. Br J Surg. 2002;89:865–9.
Suda K, Miyano T. Bile pancreatitis. Arch Pathol Lab Med. 1985;109:433–6.
Dohke M, Kato H, Motohara T, Okushiba S, Takahashi T, Kodama T. A case of pancreas carcinoma observed reflux of the bile juice on cut surface of the pancreas. Nippon Shokakibyo Gakkai Zasshi. 1995;92:1895–8. (in Japanese with English abstract).
Acosta JM, Ledesma CL. Gallstone migration as a cause of acute pancreatitis. N Engl J Med. 1974;290:484–7.
Thal A, Perry JF Jr, Egner W. A clinical and morphological study of forty-two cases of fatal acute pancreatitis. Surg Gynecol Obstet. 1957;105:191–202.
Howell CW, Bergh GS. Pancreatic duct filling during cholangiography: its effect upon serum amylase levels. Gastroenterology. 1950;6:309–16.
Arendt T, Nizze H, Monig H, Kloehn S, Stuber E, Folsch UR. Biliary pancreatic reflux-induced acute pancreatitis-myth or possibility? Eur J Gastroenterol Hepatol. 1999;11:329–35.
Sugiyama M, Atomi Y, Kuroda A. Pancreatic disorders associated with anomalous pancreaticobiliary junction. Surgery. 1999;126:492–7.
Shimada K, Yanagisawa J, Nakayama F. Increased lysophosphatidylcholine and pancreatic enzyme content in bile of patients with anomalous pancreaticobiliary ductal junction. Hepatology. 1991;13:438–44.
Nakamura T, Okada A, Higaki J, Tojo H, Okamoto M. Pancreaticobiliary maljunction-associated pancreatitis: an experimental study on the activation of pancreatic phospholipase A2. World J Surg. 1996;20:543–50.
Okada A, Higaki J, Nakamura T, Hukui Y, Kamata S. Pancreatitis associated with choledochal cyst and other anomalies in childhood. Br J Surg. 1995;82:829–32.
Stain SC, Guthrie CR, Yellin AE, Donovan AJ. Choledochal cyst in the adult. Ann Surg. 1995;222:128–33.
Lipsett PA, Pitt HA, Colombani PM, Boitnott JK, Cameron JL. Choledochal cyst disease. A Changing pattern of presentation. Ann Surg. 1994;220:644–52.
Kamisawa T, Matsukawa M, Amemiya K, Tu Y, Egawa N, Okamoto A, et al. Pancreatitis associated with pancreaticobiliary maljunction. Hepatogastroenterology. 2003;50:1665–8.
Kamisawa T, Egawa N, Tsuruta K, Okamoto A, Matsukawa M. Pancreatitis associated with congenital abnormalities of the pancreaticobiliary system. Hepatogastroenterology. 2005;52:223–9.
Kamisawa T, Tu Y, Nakajima H, Egawa N, Tsuruta K, Okamoto A, et al. Acute pancreatitis associated with a long common channel. Abdom Imaging. 2007;32:365–9.
Kaneko K, Ando H, Ito T, Watanabe Y, Seo T, Harada T, et al. Protein plugs cause symptoms in patients with choledochal cysts. Am J Gastroenterol. 1997;92:1018–21.
Kamisawa T, Amemita K, Tu Y, Egawa N, Sakaki N, Tsuruta K, et al. Clinical significance of a long common channel. Pancreatology. 2001;2:122–8.
Kamisawa T, Funata N, Hayashi Y, Egawa N, Nakajima H, Tsuruta K, et al. Pathologic changes in the non-carcinomatous epithelium of the gallbladder in patients with a relatively long common channel. Gastrointest Endosc. 2004;60:56–60.
Kamisawa T, Kuwata G, Chen PY, Tu Y, Fujiwara T, Endoh J, et al. Precancerous lesions in the gallbladder of patients with a long common channel. Dig Endosc. 2006;18:192–5.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kamisawa, T., Kurata, M., Honda, G. et al. Biliopancreatic reflux—pathophysiology and clinical implications. J Hepatobiliary Pancreat Surg 16, 19–24 (2009). https://doi.org/10.1007/s00534-008-0010-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00534-008-0010-5