Abstract
Purpose
The integration of palliative care (PC) into oncological management is recommended well before the end of life. It improves quality of life and symptom control and reduces the aggressiveness of end-of-life care. However, its appropriate timing is still debated. Entry into an early-phase clinical trial (ECT) represents hopes for the patient when standard treatments have failed. It is an opportune moment to integrate PC to preserve the patient’s general health status. The objective of this study was to evaluate the motives for acceptance or refusal of early PC management in patients included in an ECT.
Methods
Patients eligible to enter an ECT were identified and concomitant PC was proposed. All patients received exploratory interviews conducted by a researcher. Their contents were analyzed in a double-blind thematic analysis with a self-determination model.
Results
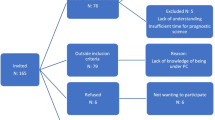
Motives for acceptance (PC acceptors: n = 27) were both intrinsic (e.g., pain relief, psychological support, anticipation of the future) and extrinsic (e.g., trust in the medical profession, for a relative, to support the advance of research). Motives for refusal (PC refusers: n = 3) were solely intrinsic (e.g., PC associated with death, negative representation of psychological support, no need for additional care, claim of independence).
Conclusions
The motives of acceptors and refusers are not internalized in the same way and call for different autonomy needs. Acceptors and refusers are influenced by opposite representations of PC and a different perception of mixed management.
Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Chakiba C, Grellety T, Bellera C, Italiano A (2018) Encouraging trends in modern phase 1 Oncology Trials | NEJM, 378, 2242‑2243. https://doi.org/10.1056/NEJMc1803837
Derbez B (2018) Les paradoxes du care dans les essais cliniques de phase I en oncologie. Scie Sociales et Santé 36(1):5. https://doi.org/10.3917/sss.361.0005
Gota V, Nookala M, Yadav A, Kannan S, Ali R (2015) Quality of informed consent in cancer clinical trials in India: A cross-sectional survey. J Clin Onco 33(15_suppl):e17652–e17652. https://doi.org/10.1200/jco.2015.33.15_suppl.e17652
Lynöe N, Sandlund M, Dahlqvist G, Jacobsson L (1991) Informed consent: study of quality of information given to participants in a clinical trial. BMJ 303(6803):610–613. https://doi.org/10.1136/bmj.303.6803.610
Schaeffer MH, Krantz DS, Wichman A, Masur H, Reed E, Vinicky JK (1996) The impact of disease severity on the informed consent process in clinical research. Am J Med 100(3):261–268. https://doi.org/10.1016/S0002-9343(97)89483-1
Cohen L, de Moor C, Amato RJ (2001) The association between treatment-specific optimism and depressive symptomatology in patients enrolled in a Phase I cancer clinical trial 91(10):1949‑1955. https://doi.org/10.1002/1097-0142(20010515)91:10<1949::aid-cncr1218>3.0.co;2-a
Derbez B (2018) Entre essai clinique et soins palliatifs : travail de démarcation et d’appariement en oncologie. Sociologie du Travail 60(2). https://doi.org/10.4000/sdt.2026
Cassel JB, Del Fabbro E, Arkenau T, Higginson IJ, Hurst S, Jansen LA, Miller FG (2016) Phase I cancer trials and palliative care: antagonism, irrelevance, or synergy? J Pain Symptom Manag 52(3):437–445. https://doi.org/10.1016/j.jpainsymman.2016.02.014
Davis MP, Temel JS, Balboni T, Glare P (2015) A review of the trials which examine early integration of outpatient and home palliative care for patients with serious illnesses. Ann Palliat Med 4(3):99–121. https://doi.org/10.3978/j.issn.2224-5820.2015.04.04
Ferrell B, Chung V, Hughes MT, Koczywas M, Azad NS, Ruel NH, Smith TJ (2021) A Palliative Care Intervention for Patients on Phase 1 Studies. J Palliat Med 24(6):846–856. https://doi.org/10.1089/jpm.2020.0597
Haun MW, Estel S, Rücker G, Friederich H-C, Villalobos M, Thomas M, Hartmann M (2017) Early palliative care for adults with advanced cancer. Cochrane Database Syst Rev (6). https://doi.org/10.1002/14651858.CD011129.pub2
Hui D, Parsons H, Nguyen L, Palla SL, Yennurajalingam S, Kurzrock R, Bruera E (2010) Timing of palliative care referral and symptom burden in phase 1 cancer patients: A retrospective cohort study. Cancer 116(18):4402–4409. https://doi.org/10.1002/cncr.25389
Markman M (2017) Why Should phase 1 cancer trial participation and optimal end-of-life care be incompatible? Oncology 92(1):1–2. https://doi.org/10.1159/000452161
Temel JS, Gallagher ER, Jackson VA, Blinderman CD, Billings JA (2010) Early palliative care for patients with metastatic non–small-cell lung cancer. New England J Med 363(8):733–42. https://doi.org/10.1056/NEJMoa1000678
Bakitas MA, Tosteson TD, Li Z, Lyons KD, Hull JG, Li Z, Ahles TA (2015) Early versus delayed initiation of concurrent palliative oncology care: patient outcomes in the ENABLE III randomized controlled trial. J Clin Oncol 33(13):1438–1445. https://doi.org/10.1200/JCO.2014.58.6362
Zhuang H, Ma Y, Wang L, Zhang H (2018) Effect of early palliative care on quality of life in patients with non-small-cell lung cancer. Curr Oncol (Toronto, Ont.) 25(1):54–58. https://doi.org/10.3747/co.25.3639
INCa (2021) Stratégie décennale de Lutte Contre les Cancers 2021–2030. Retrieved from 2 may 2024 at https://www.e-cancer.fr/Institut-national-du-cancer/Strategie-de-lutte-contre-les-cancers-en-France/La-strategie-decennale-de-lutte-contre-les-cancers-2021-2030
Thienprayoon R, LeBlanc T (2015) Early integration of palliative care into the care of patients with cancer 2015(1):479–83. https://doi.org/10.1182/asheducation-2015.1.479
Ceccaldi J (2017) L’annonce palliative. In Soins palliatifs (Vol. 2ème ed., p. 117–121). Paris: Dunod. https://doi.org/10.3917/dunod.bioyv.2017.01.0117
LeBlanc TW, El-Jawahri A (2015) When and why should patients with hematologic malignancies see a palliative care specialist? Hematology 2015(1):471–478. https://doi.org/10.1182/asheducation-2015.1.471
Jordan RI, Allsop MJ, ElMokhallalati Y, Jackson CE, Edwards HL, Chapman EJ, Deliens L, Bennett MI (2020) Duration of palliative care before death in international routine practice: a systematic review and meta-analysis. BMC Med 18(1):368. https://doi.org/10.1186/s12916-020-01829-x
Castaing M (2015) Entre savoir et incertitude, circulation de la parole : interdisciplinarité en soins palliatifs pédiatriques. Méd Palliat : Soins de Supp - Accompagnement - Éthique 14(5):295–300. https://doi.org/10.1016/j.medpal.2015.09.001
Magerl H, Zeller O (2014) Les valeurs de l’Approche centrée sur la personne en soins palliatifs: Accompagnement des mourants et des personnes endeuillées, travail des intervenants en équipe pluridisciplinaire. Approche Centrée sur la Personne. Pratique et recherche, n° 19(1), 83‑94. https://doi.org/10.3917/acp.019.0083
Bruera E, Yennurajalingam S (2012) Palliative care in advanced cancer patients: how and when? Oncologist 17(2):267–273. https://doi.org/10.1634/theoncologist.2011-0219
Braun V, Clarke V (2006) Using thematic analysis in psychology. Qual Res Psychol 3(2):77–101. https://doi.org/10.1191/1478088706qp063oa
Vaismoradi M, Turunen H, Bondas T (2013) Content analysis and thematic analysis: Implications for conducting a qualitative descriptive study: qualitative descriptive study. Nurs Health Sci 15(3):398–405. https://doi.org/10.1111/nhs.12048
Leech NL, Onwuegbuzie AJ (2011) Beyond constant comparison qualitative data analysis: Using NVivo. Sch Psychol Q 26(1):70–84. https://doi.org/10.1037/a0022711
Ryan RM, Deci EL (2000) Intrinsic and Extrinsic Motivations: Classic Definitions and New Directions 25(1):54‑67. https://doi.org/10.1006/ceps.1999.1020
Bell JAH, Balneaves LG (2015) Cancer patient decision making related to clinical trial participation: an integrative review with implications for patients’ relational autonomy. Supp Care Cancer 23(4):1169–1196. https://doi.org/10.1007/s00520-014-2581-9
Catt S, Langridge C, Fallowfield L, Talbot DC, Jenkins V (2011) Reasons given by patients for participating, or not, in Phase 1 cancer trials - ScienceDirect 47(10):1490‑1497. https://doi.org/10.1016/j.ejca.2011.02.020
Earle CC, Landrum MB, Souza JM, Neville BA, Weeks JC, Ayanian JZ (2008) Aggressiveness of Cancer Care Near the End of Life: Is It a Quality-of-Care Issue? J Clin Oncol 26(23):3860–3866. https://doi.org/10.1200/JCO.2007.15.8253
Jenkins VA, Anderson JL, Fallowfield LJ (2010) Communication and informed consent in phase 1 trials: a review of the literature from January 2005 to July 2009. Supp Care Cancer 18(9):1115–1121. https://doi.org/10.1007/s00520-010-0836-7
Ling J, Rees E, Hardy J (2000) What in¯uences participation in clinical trials in palliative care in a cancer centre? Eur J Cancer 36(5):621–6. https://doi.org/10.1016/s0959-8049(99)00330-5
Pentz RD, White M, Harvey RD, Farmer ZL, Liu Y, Lewis C, Dashevskaya O, Owonikoko T, Khuri FR (2012) Therapeutic misconception, misestimation, and optimism in participants enrolled in phase 1 trials: Phase 1 participants’ understanding. Cancer 118(18):4571–4578. https://doi.org/10.1002/cncr.27397
Kyeremanteng K, Beckerleg W, Wan C, Vanderspank-Wright B, D’Egidio G, Sutherland S, Hartwick M, Gratton V, Sarti AJ (2020) Survey on Barriers to Critical Care and Palliative Care Integration. Am J Hospice and Pallia Med®, 37(2):108‑116. https://doi.org/10.1177/1049909119867658
Goni-Fuste B, Pergolizzi D, Monforte-Royo C, Julià-Torras J, Rodríguez-Prat A, Crespo I (2023) What makes the palliative care initial encounter meaningful? A descriptive study with patients with cancer, family carers and palliative care professionals. Palliat Med 37(8):1252–1265. https://doi.org/10.1177/02692163231183998
Jia Z, Yeh IM, Lee CH, Yeung AS, Tulsky JA, Leiter RE (2022) Barriers and facilitators to advance care planning among chinese patients with advanced cancer and their caregivers. J Palliat Med 25(5):774–782. https://doi.org/10.1089/jpm.2021.0404
Deci EL, Ryan RM (1985) Conceptualizations of intrinsic motivation and self-determination. In: Intrinsic motivation and self-determination in human behavior (Plenum Press., p. 11–40). Retrieved from 2 may 2024 at https://link.springer.com/chapter/10.1007/978-1-4899-2271-7_2
Gauthier L, Sénécal C, Guay F (2007) Construction et validation de l’Échelle de motivation à avoir un enfant (EMAE) 57(2):77‑89. https://doi.org/10.1016/j.erap.2006.09.001
Erel M, Marcus E-L, Dekeyser-Ganz F (2017) Barriers to palliative care for advanced dementia: a scoping review. Ann Palliat Med 6(4):36579–36379. https://doi.org/10.21037/apm.2017.06.13
Seccareccia D, Wentlandt K, Kevork N, Workentin K, Blacker S, Gagliese L, Grossman D, Zimmermann C (2015) Communication and quality of care on palliative care units: a qualitative study. J Palliat Med 18(9):758–764. https://doi.org/10.1089/jpm.2014.0408
Aubry R (2005) Annoncer un diagnostic difficile ou un pronostic péjoratif: vérité et stratégies de communication. - Résultats de votre recherche - Banque de données en santé publique. Retrieved from 2 novembre 2021 at https://bdsp-ehesp.inist.fr/vibad/index.php?action=getRecordDetail&idt=327968
Parajuli J, Tark A, Jao Y-L, Hupcey J (2020) Barriers to palliative and hospice care utilization in older adults with cancer: A systematic review. J Geriatr Oncol 11(1):8–16. https://doi.org/10.1016/j.jgo.2019.09.017
Ahmed N, Bestall JE, Ahmedzai SH, Payne SA, Clark D, Noble B (2004) Systematic review of the problems and issues of accessing specialist palliative care by patients, carers and health and social care professionals. Palliat Med 18(6):525–542. https://doi.org/10.1191/0269216304pm921oa
Meo N, Hwang U, Morrison RS (2011) Resident perceptions of palliative care training in the emergency department. J Palliat Med 14(5):548–555. https://doi.org/10.1089/jpm.2010.0343
Lee Y-Y, Lin JL (2010) Do patient autonomy preferences matter? Linking patient-centered care to patient–physician relationships and health outcomes. Soc Sci Med 71(10):1811–1818. https://doi.org/10.1016/j.socscimed.2010.08.008
Godfroid T (2016) 2. L’autonomie dans les relations soignants-soignés : une approche par le cancer. In: La dynamique sociale des subjectivités en cancérologie (p 41‑54). Toulouse: Érès. https://doi.org/10.3917/eres.derb.2016.01.0041
Sarradon-Eck A, Besle S, Troian J, Capodano G, Mancini J (2019) Understanding the barriers to introducing early palliative care for patients with advanced cancer: a qualitative study. J Palliat Med 22(5), jpm.2018.0338. https://doi.org/10.1089/jpm.2018.0338
Gysels M, Shipman C, Higginson IJ (2008) “I Will Do It If It Will Help Others:” Motivations Among Patients Taking Part in Qualitative Studies in Palliative Care. J Pain Symptom Manage 35(4):347–355. https://doi.org/10.1016/j.jpainsymman.2007.05.012
Acknowledgements
This work was supported by La Ligue Nationale contre le Cancer. We thank Julia Barbat and the Clinical Research Associates—Zohra Akkal, Sakina Benmameche-Medjahed, Carine Chabé—for their help. Finally, we also wish to thank all the patients for their participation.
Funding
This work was supported by La Ligue Nationale contre le Cancer (AK/IP/BC 16651). All authors have received research support from La Ligue contre le Cancer.
Author information
Authors and Affiliations
Contributions
Conception and design: all authors Provision of study patients: Mona Amini-Adle, Mehdi Brahmi, Philippe Cassier, Helene Vanacker, Loïc Verlingue, Armelle Vinceneux, Aurélie Swalduz Collection and assembly of data: Amélie Anota, Valentine Baudry, Johanna Despax, Julien Gautier, Magali Girodet, Mathilde Lochmann Data analysis and interpretation: Mehdi Brahmi, Philippe Cassier, Véronique Christophe, Gisèle Chvetzoff, Johanna Despax, Julien Gautier, Magali Girodet, Mathilde Lochmann, Helene Vanacker, Armelle Vinceneux Writing: Valentine Baudry, Véronique Christophe, Gisèle Chvetzoff, Johanna Despax, Julie Duranti, Julien Gautier, Magali Girodet, Mathilde Lochmann, Helene Vanacker, Armelle Vinceneux Final approval of manuscript: all authors.
Corresponding author
Ethics declarations
Ethics approval
“PALPHA” was a research not involving human participants (NRIPH). It is performed in line with the principles of the Declaration of Helsinki. PALPHA complies with reference methodology MR004 (Ref. R201-004–064) of the Commission Nationale de l'Informatique et des Libertés (CNIL). PALPHA was registered with ClinicalTrials.gov as an observational study (NCT04717440). The protocol received a favorable opinion (advisory assent n°2020–006) from the independent Ethical Review Group of the Léon Bérard Center on December 15th, 2020.
Consent to participate
A patient information sheet was developed for the participants. They did not have to sign an informed consent form. Their non-opposition (i.e., oral declaration made by participants of their non-opposition to the research, applied to non-RIPH projects that do not require a consent form) was notified and traceable in their medical file.
Consent for publication
All personal data has been anonymized during the study and in this publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lochmann, M., Girodet, M., Despax, J. et al. Qualitative evaluation of motives for acceptance or refusal of early palliative care in patients included in early-phase clinical trials in a French comprehensive cancer center: the PALPHA study. Support Care Cancer 32, 353 (2024). https://doi.org/10.1007/s00520-024-08535-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08535-x