Abstract
Purpose
The objective of this study was to estimate the cost of antiemetic therapy for chemotherapy-induced nausea and vomiting (CINV) in daily practice in Japan.
Methods
This was a retrospective observational study using medical records. Eligible patients were those with bladder or testicular cancer receiving platinum-containing highly emetogenic chemotherapy. The incidence of CINV on days 1–5 in single-day chemotherapy and on days 1–9 in multiple-day chemotherapy, and the costs of antiemetic therapy directly associated with the administration of antiemetics were estimated. The analysis of costs was performed from a hospital perspective.
Results
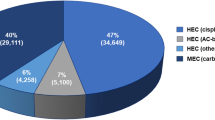
A total of 54 patients or 169 chemotherapy courses were included. In all chemotherapy courses 5-HT3 receptor antagonists were used on the day(s) that platinum-containing agents were administered and frequently used on subsequent days. In contrast, the use of corticosteroids was infrequent. Acute CINV in single-day chemotherapy was well controlled, but the incidences of delayed CINV in single-day chemotherapy and CINV in multiple-day chemotherapy were relatively high. The costs for antiemetic therapy were $484.65 in courses with CINV and $318.56 in courses without CINV, and the difference was approximately $170 per chemotherapy course, which was considered to be mainly imputable to the prevalence of CINV.
Conclusions
The cost of antiemetic therapy for CINV is substantial in Japan as well as in other countries, and it is suggested that the onset of CINV is a possible cost driver. The improvements in antiemetic therapy may contribute not only to improved patient well-being but also to a reduction of economic burden.
Similar content being viewed by others
Abbreviations
- CINV:
-
Chemotherapy-induced nausea and vomiting
References
de Boer-Dennert M, de Wit R, Schmitz PI, Djontono J, van Beurden V, Stoter G, Verweij J (1997) Patient perceptions of the side-effects of chemotherapy: the influence of 5HT3 antagonists. Br J Cancer 76:1055–1061
Hickok JT, Roscoe JA, Morrow GR, King DK, Atkins JN, Fitch TR (2003) Nausea and emesis remain significant problems of chemotherapy despite prophylaxis with 5-hydroxytryptamine-3 antiemetics: a University of Rochester James P. Wilmot Cancer Center Community Clinical Oncology Program Study of 360 cancer patients treated in the community. Cancer 97:2880–2886
Sun CC, Bodurka DC, Weaver CB, Rasu R, Wolf JK, Bevers MW, Smith JA, Wharton JT, Rubenstein EB (2005) Rankings and symptom assessments of side effects from chemotherapy: insights from experienced patients with ovarian cancer. Support Care Cancer 13:219–227
Hesketh PJ (2008) Chemotherapy-induced nausea and vomiting. N Engl J Med 358:2482–2494
Roila F, Hesketh PJ, Herrstedt J, Antiemetic Subcommittee of the Multinational Association of Supportive Care in Cancer (2006) Prevention of chemotherapy- and radiotherapy-induced emesis: results of the 2004 Perugia International Antiemetic Consensus Conference. Ann Oncol 17:20–28
Ihbe-Heffinger A, Ehlken B, Bernard R, Berger K, Peschel C, Eichler HG, Deuson R, Thödtmann J, Lordick F (2004) The impact of delayed chemotherapy-induced nausea and vomiting on patients, health resource utilization and costs in German cancer centers. Ann Oncol 15:526–536
Cohen L, de Moor CA, Eisenberg P, Ming EE, Hu H (2007) Chemotherapy-induced nausea and vomiting: incidence and impact on patient quality of life at community oncology settings. Support Care Cancer 15:497–503
Navari RM (2007) Prevention of emesis from multiple-day and high-dose chemotherapy regimens. J Natl Compr Canc Netw 5:51–59
Lachaine J, Yelle L, Kaizer L, Dufour A, Hopkins S, Deuson R (2005) Chemotherapy-induced emesis: quality of life and economic impact in the context of current practice in Canada. Support Cancer Ther 2:181–187
Ballatori E, Roila F, Ruggeri B, Porrozzi S, Iannopollo M, Soru G, Cruciani G, Daniele B, Locatelli MC, Pellissier J, Deuson R (2007) The cost of chemotherapy-induced nausea and vomiting in Italy. Support Care Cancer 15:31–38
Tina Shih YC, Xu Y, Elting LS (2007) Costs of uncontrolled chemotherapy-induced nausea and vomiting among working-age cancer patients receiving highly or moderately emetogenic chemotherapy. Cancer 110:678–685
Ishimaru H, Takayama S, Shiokawa M, Inoue T (2008) Cost-effectiveness analysis of 5-HT3 receptor antagonist drugs in cancer chemotherapy. Gan To Kagaku Ryoho 35:619–623 (in Japanese)
Sato J, Terui K, Souma A, Fujita S, Hayakari M (2007) Survey on antiemetic therapy in ambulatory cancer chemotherapy and medical economics for standardization. Gan To Kagaku Ryoho 34:1637–1642 (in Japanese)
Tarn YH, Hu S, Kamae I, Yang BM, Li SC, Tangcharoensathien V, Teerawattananon Y, Limwattananon S, Hameed A, Aljunid SM, Bapna JS (2008) Health-care systems and pharmacoeconomic research in Asia-Pacific region. Value Health 11:S137–S155
Pectasides D, Pectasides M, Economopoulos T (2006) Systemic chemotherapy in locally advanced and/or metastatic bladder cancer. Cancer Treat Rev 32:456–470
Kondagunta GV, Motzer RJ (2006) Chemotherapy for advanced germ cell tumors. J Clin Oncol 24:5493–5502
Hesketh PJ, Kris MG, Grunberg SM, Beck T, Hainsworth JD, Harker G, Aapro MS, Gandara D, Lindley CM (1997) Proposal for classifying the acute emetogenicity of cancer chemotherapy. J Clin Oncol 15:103–109
SASInstitute (2000) JMP statistics and graphic guide. SAS Institute, Cary
Antiemetic Subcommittee of the Multinational Association of Supportive Care in Cancer (MASCC) (1998) Prevention of chemotherapy- and radiotherapy-induced emesis: results of Perugia Consensus Conference. Ann Oncol 9:811–819
Einhorn LH, Rapoport B, Koeller L, Grunberg SM, Feyer P, Rittenberg C, Aapro M (2005) Antiemetic therapy for multiple-day chemotherapy and high-dose chemotherapy with stem cell transplant: review and consensus statement. Support Care Cancer 13:112–116
Japan Society of Clinical Oncology (2010) Seitoyaku tekiseishiyou guideline. Kanehara Shuppan, Tokyo, in Japanese
Geling O, Eichler HG (2005) Should 5-hydroxytryptamine-3 receptor antagonists be administered beyond 24 hours after chemotherapy to prevent delayed emesis? Systematic re-evaluation of clinical evidence and drug cost implications. J Clin Oncol 23:1289–1294
Oo TH, Hesketh PJ (2005) Drug insight: new antiemetics in the management of chemotherapy-induced nausea and vomiting. Nat Clin Pract Oncol 2:196–201
Gralla R, Lichinitser M, Van Der Vegt S, Sleeboom H, Mezger J, Peschel C, Tonini G, Labianca R, Macciocchi A, Aapro M (2003) Palonosetron improves prevention of chemotherapy-induced nausea and vomiting following moderately emetogenic chemotherapy: results of a double-blind randomized phase III trial comparing single doses of palonosetron with ondansetron. Ann Oncol 14:1570–1577
Eisenberg P, Figueroa-Vadillo J, Zamora R, Charu V, Hajdenberg J, Cartmell A, Macciocchi A, Grunberg S, 99–04 Palonosetron Study Group (2003) Improved prevention of moderately emetogenic chemotherapy-induced nausea and vomiting with palonosetron, a phaemacologically novel 5-HT3 receptor antagonist: results of a phase III, single-dose trial versus dolasetron. Cancer 98:2473–2482
Saito M, Aogi K, Sekine I, Yoshizawa H, Yanagita Y, Sakai H, Inoue K, Kitagawa C, Ogura T, Mitsuhashi S (2009) Palonosetron plus dexamethasone versus granisetron plus dexamethasone for prevention of nausea and vomiting during chemotherapy: a double-blind, double-dummy, randomized, comparative phase III trial. Lancet Oncol 10:115–124
Acknowledgments
This study was partly supported by a grant from Pfizer Health Research Foundation (2009).
Conflict of interest
No conflict of interest is declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamada, S., Hinotsu, S., Hori, K. et al. The cost of antiemetic therapy for chemotherapy-induced nausea and vomiting in patients receiving platinum-containing regimens in daily practice in Japan: a retrospective study. Support Care Cancer 20, 813–820 (2012). https://doi.org/10.1007/s00520-011-1155-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-011-1155-3