Abstract
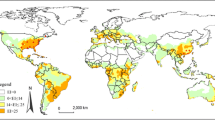
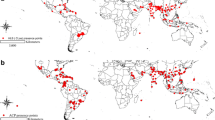
Chrysodeixis includens is a polyphagous pest restricted to the American continent. The occurrence of C. includens is allied, among other factors, by favorable conditions such as temperature, humidity, presence of hosts, and migratory behavior. In this work, we built spatiotemporal species distribution models at continental and global levels for the distribution of C. includens using CLIMEX to determine times and regions favorable for year-round survival and migration of this species and in case of invasion on other continents to apply timely and right phytosanitary measures. Our models estimated high climate suitability for C. includens in Central and large proportions of South America throughout the year. Moreover, there is suitability for C. includens growth in all months of the year in Central and northern part of South America. In the northern hemisphere, these conditions range from April to October, while in mid-southern parts of South America, favorable periods comprise October through June. The countries with the highest suitability for C. includens outside the American continent are located on the African and Asian continents. Our results show variable climate suitability for C. includens during the year that help to understand likely migration pattern in North America. This information would direct efforts for appropriate C. includens management during warm and moist periods of the year. Furthermore, our models notify the need for the development of strategies for the inspection and interception of C. includens especially in central Africa, India, South and Southeast Asia, and Northeast Australia.





Similar content being viewed by others
Data Availability
Electronic Supplementary Material (occurrence data and spatiotemporal model video) available.
References
Addo-Bediako A, Chown SL, Gaston KJ (2000) Thermal tolerance, climatic variability and latitude. Proc R Soc B 267:739–745. https://doi.org/10.1098/rspb.2000.1065
Andrade K, Bueno ADF, Da Silva DM, Stecca CDS, Pasini A, De Oliveira MCN (2016) Bioecological characteristics of Chrysodeixis includens (Lepidoptera: Noctuidae) fed on different hosts. Austral Entomol 55:449–454. https://doi.org/10.1111/aen.12208
Baldin ELL, Lourenção AL, Schlick-Souza EC (2014) Outbreaks of Chrysodeixis includens. Bragantia 73:458–465. https://doi.org/10.1590/1678-4499.0277
Bellard C, Leroy B, Thuiller W, Rysman JF, Courchamp F (2016) Major drivers of invasion risks throughout the world. Ecosphere 7:e01241–e01241. https://doi.org/10.1002/ecs2.1241
Benedito ADS, Fernandes OA, Ferreira CP (2021) Thermal Requirements and Population Viability of Chrysodeixis includens (Lepidoptera: Noctuidae). Neotropical Entomology 50 (2):186–196. https://doi.org/10.1007/s13744-020-00825-3
Bestete LR, Torres JB, Silva RBB, Silva-Torres CSA (2016) Water stress and kaolin spray affect herbivorous insects’ success on cotton. Arthropod-Plant Interactions 10 (5):445–453. https://doi.org/10.1007/s11829-016-9454-8
Bueno ADF, Bortolotto OC, Bueno R, Hamada E, Favetti BM, Silva GV (2017) Efeitos do aquecimento global sobre pragas de oleaginosas. In: Bettiol W, Hamada E, Angelotti F, Auad AM, Ghini R (eds) Aquecimento global e problemas fitossanitários. Embrapa, Brasília, DF, pp 68–68
CABI (2020) Invasive Species Compendium. CAB International. Www.cabi.org/isc. Accessed 12/14/2019
Chamuene A, Araújo TA, Silva G, Costa TL, Berger PG, Picanço MC (2018) Performance of the natural mortality factors of Aphis gossypii (Hemiptera: Aphididae) as a function of cotton plant variety and phenology. Environ Entomol 47:440–447. https://doi.org/10.1093/ee/nvx205
Chown SL, Terblanche JS (2006) Physiological diversity in insects: ecological and evolutionary contexts. Adv Insect Physiol 33:50–152. https://doi.org/10.1016/S0065-2806(06)33002-0
Clark BR, Faeth SH (1998) The evolution of egg clustering in butterflies: a test of the egg desiccation hypothesis. Evol Ecol 12:543–552. https://doi.org/10.1023/A:1006504725592
Colinet H, Hance T (2010) Interspecific variation in the response to low temperature storage in different aphid parasitoids. Ann Appl Biol 156:147–156. https://doi.org/10.1111/j.1744-7348.2009.00374.x
Colinet H, Sinclair BJ, Vernon P, Renault D (2015) Insects in fluctuating thermal environments. Annu Rev Entomol 60:123–140. https://doi.org/10.1146/annurev-ento-010814-021017
Collins FL, Johnson SJ (1985) Reproductive response of caged adult velvetbean caterpillar and soybean looper to the presence of weeds Agric. Ecosyst Environ 14:139–149. https://doi.org/10.1016/0167-8809(85)90091-X
Dynesius M, Jansson R, Huey RB, Sheldon KS, Ghalambor CK, Haak DC, Martin PR (2000) Evolutionary consequences of changes in species' geographical distributions driven by Milankovitch climate oscillations. Proc Natl Acad Sci USA 97:9115–9120. https://doi.org/10.1073/pnas.97.16.9115
Early R, Bradley BA, Dukes JS et al (2016) Global threats from invasive alien species in the twenty-first century and national response capacities. Nat Commun 7:12485–12485. https://doi.org/10.1038/ncomms12485
Eichlin TD, Cunningham HB (1978) The Plusiinae (Lepidoptera: Noctuidae) of America North of Mexico, emphasizing genitalic and larval morphology. United States Department of Agriculture (USDA), Agricultural Research Service. https://doi.org/10.22004/ag.econ.158098
EPPO (2020) EPPO GD Desktop - database on quarantine pests
Estay SA, Lima M, Bozinovic F (2014) The role of temperature variability on insect performance and population dynamics in a warming world. Oikos 123:131–140. https://doi.org/10.1111/j.1600-0706.2013.00607.x
Finch EA, Beale T, Chellappan M, Goergen G, Gadratagi BG, Khan MAM, Rehman A, Rwomushana I, Sarma AK, Wyckhuys KAG, Kriticos DJ (2021) The potential global distribution of the papaya mealybug, a polyphagous pest. Pest Manag Sci 77 (3):1361–1370. https://doi.org/10.1002/ps.6151
Franklin J, Miller JA (2010) Mapping species distributions: spatial inference and prediction. Cambridge University Press. https://doi.org/10.1017/CBO9780511810602
Gasparri NI, Kuemmerle T, Meyfroidt P, le Polain de Waroux Y, Kreft H (2016) The emerging soybean production frontier in southern Africa: conservation challenges and the role of south-south telecouplings. Conserv Lett 9:21–31. https://doi.org/10.1111/conl.12173
GBIF (2020) Occurrence Download https://www.gbif.org/. Accessed 06/29/2020
Goergen G, Kumar PL, Sankung SB, Togola A, Tamò M (2016) First report of outbreaks of the fall armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in west and central Africa. PLoS One 11:e0165632. https://doi.org/10.1371/journal.pone.0165632
Hereford J, Schmitt J, Ackerly DD (2017) The seasonal climate niche predicts phenology and distribution of an ephemeral annual plant. Mollugo verticillata J Ecol 105:1323–1334. https://doi.org/10.1111/1365-2745.12739
Herzog DC (1980) Sampling soybean looper on soybean. In: Kogan M, D.C. H (eds) Sampling methods in soybean entomology. Springer, New York, NY, pp 141–168
Holloway P, Miller JA (2017) A quantitative synthesis of the movement concepts used within species distribution modelling. Ecol Model 356:91–103. https://doi.org/10.1016/j.ecolmodel.2017.04.005
Ireland KB, Kriticos DJ (2019) Why are plant pathogens under-represented in eco-climatic niche modelling? Int J Pest Manage 65:207–216. https://doi.org/10.1080/09670874.2018.1543910
Jarnevich CS, Stohlgren TJ, Kumar S, Morisette JT, Holcombe TR (2015) Caveats for correlative species distribution modeling. Ecol Inform 29:6–15. https://doi.org/10.1016/j.ecoinf.2015.06.007
Jensen RL, Newsom LD, Gibbens J (1974) The soybean looper: effects of adult nutrition on oviposition, mating frequency, and longevity. J Econ Entomol 67:467–470. https://doi.org/10.1093/jee/67.4.467
Jost DJ, Pitre HN (2002) Soybean looper (Lepidoptera: Noctuidae) oviposition on cotton and soybean of different growth stages: influence of olfactory stimuli. J Econ Entomol 95:286–293. https://doi.org/10.1603/0022-0493-95.2.286
Kistner EJ, Hatfield JL (2018) Potential Geographic Distribution of Palmer Amaranth under Current and Future Climates. Agric Environ Lett 3:170044. https://doi.org/10.2134/ael2017.12.0044
Kriticos DJ, Maywald GF, Yonow T, Zurcher EJ, Herrmann NI, Sutherst RW (2015a) CLIMEX version 4: exploring the effects of climate on plants, animals and diseases. CSIRO, Canberra
Kriticos DJ, Ota N, Hutchison WD, Beddow J, Walsh T, Tay WT, Borchert DM, Paula-Moreas SV, Czepak C, Zalucki MP (2015b) The potential distribution of invading Helicoverpa armigera in north America: is it just a matter of time? PLoS One 10:e0119618. https://doi.org/10.1371/journal.pone.0119618
Kriticos DJ, De Barro PJ, Yonow T, Ota N, Sutherst RW (2020) The potential geographical distribution and phenology of Bemisia tabaci Middle East/Asia Minor 1, considering irrigation and glasshouse production. Bull Entomol Res 110:1–10. https://doi.org/10.1017/S0007485320000061
Li X-J, Wu M-F, Ma J et al (2020) Prediction of migratory routes of the invasive fall armyworm in eastern China using a trajectory analytical approach. Pest Manag Sci 76:454–463. https://doi.org/10.1002/ps.5530
Lobo JM (2016) The use of occurrence data to predict the effects of climate change on insects. Curr Opin Insect Sci 17:62–68. https://doi.org/10.1016/j.cois.2016.07.003
Mason LJ, Mack TP (1984) Influence of temperature on oviposition and adult female longevity for the soybean looper, Pseudoplusia includens (Walker) (Lepidoptera: Noctuidae). Environ Entomol 13:379–383. https://doi.org/10.1093/ee/13.2.379
Mitchell ER (1967) Life history of Pseudoplusia includens (Walker) (Lepidoptera: Noctuidae). J Georgia Entomogical Society 2:53–57
Mitchell ER, Chalfant RB, Greene GL, Creighton CS (1975) Soybean looper: populations in Florida, Georgia, and South Carolina, as determined with pheromone-baited BL traps. J Econ Entomol 68:747–750. https://doi.org/10.1093/jee/68.6.747
Moscardi F, Bueno ADF, Sosa-Gómez DR et al (2012) Artrópodes que atacam as folhas da soja. In: Hoffmann-Campo CB, Côrrea-Ferreira BS, Moscardi F (eds) Soja: manejo integrado de insetos e outros artrópodes-praga, 1st edn. Embrapa, Brasília, DF, pp 213–334
Murillo H, Hunt DWA, Vanlaerhoven SL (2013) First records of Chrysodeixis chalcites (Lepidoptera: Noctuidae: Plusiinae) for east-central Canada. Can Entomol 145:338–342. https://doi.org/10.4039/tce.2012.106
Musser FR, Catchot ALJ, Conley SP et al (2019) 2018 soybean insect losses in the United States vol 12. Mississipi State University
Paini DR, Sheppard AW, Cook DC, De Barro PJ, Worner SP (2016) Thomas MB. Global threat to agriculture from invasive species Proc Natl Acad Sci USA 113:7575–7579. https://doi.org/10.1073/pnas.1602205113
Palma J, Maebe K, Jerson Guedes VC, Smagghe G, Guedes RNC (2015) Molecular Variability and Genetic Structure of Chrysodeixis includens (Lepidoptera: Noctuidae), an Important Soybean Defoliator in Brazil. PLOS ONE 10 (3):e0121260. https://doi.org/10.1371/journal.pone.0121260
Ramos RS, Kumar L, Shabani F, Da Silva RS, De Araújo TA, Picanço MC (2019) Climate model for seasonal variation in Bemisia tabaci using CLIMEX in tomato crops. Int J Biometeorol 63:281–291. https://doi.org/10.1007/s00484-018-01661-2
Robinet C, Roques A (2010) Direct impacts of recent climate warming on insect populations. Integr Zool 5:132–142. https://doi.org/10.1111/j.1749-4877.2010.00196.x
Santana PA, Kumar L, Da Silva RS, Picanço MC, Silva RSD, Picanço MC (2019) Global geographic distribution of Tuta absoluta as affected by climate change. J Pest Sci 92:1373–1385. https://doi.org/10.1007/s10340-018-1057-y
Santos SRD, Specht A, Carneiro E et al (2017) Interseasonal variation of Chrysodeixis includens (Walker, [1858]) (Lepidoptera: Noctuidae) populations in the Brazilian savanna. Rev Bras Entomol 61:294–299. https://doi.org/10.1016/j.rbe.2017.06.006
Sinclair TR, Marrou H, Soltani A, Vadez V, Chandolu KC (2014) Soybean production potential in Africa. Glob Food Sec 3:31–40. https://doi.org/10.1016/j.gfs.2013.12.001
Singh AK, Rembold H (1989) Oviposition behaviour of Heliothis armigera (Lepidoptera: Noctuidae) in relation to the day-night cycle. Insect Sci Appl 10:393–400. https://doi.org/10.1017/S1742758400003659
Sosa-Gómez DR, Delpin KE, Moscardi F, Nozaki MDH (2003) The impact of fungicides on Nomuraea rileyi (Farlow) Samson epizootics and on populations of Anticarsia gemmatalis Hübner (Lepidoptera: Noctuidae), on soybean. Neotrop Entomol 32:287–291. https://doi.org/10.1590/S1519-566X2003000200014
Specht A, De Paula-Moraes SV, Sosa-Gómez DR (2015) Host plants of Chrysodeixis includens (Walker) (Lepidoptera, Noctuidae, Plusiinae). Rev Bras Entomol 59:343–345. https://doi.org/10.1016/j.rbe.2015.09.002
Specht A, Sosa-Gómez DR, Roque-Specht VF, Valduga E, Gonzatti F, Schuh SM, Carneiro E (2019) Biotic potential and life tables of Chrysodeixis includens (Lepidoptera: Noctuidae), Rachiplusia nu, and Trichoplusia ni on soybean and forage turnip. J Insect Sci 19. https://doi.org/10.1093/jisesa/iez072
Stoeckli S, Felber R, Haye T (2020) Current distribution and voltinism of the brown marmorated stink bug, Halyomorpha halys, in Switzerland and its response to climate change using a high-resolution CLIMEX model. Int J Biometeorol 64 (12):2019–2032. https://doi.org/10.1007/s00484-020-01992-z
Sutherst RW (2014) Pest species distribution modelling: origins and lessons from history. Biol Invasions 16:239–256. https://doi.org/10.1007/s10530-013-0523-y
Szyniszewska AM, Leppla NC, Manoukis NC, Collier TC, Hastings JM, Kriticos DJ, Bigsby KM (2020) CLIMEX and MED-FOES models for predicting the variability in growth potential and persistence of mediterranean fruit fly (Diptera: Tephritidae) populations. Ann Entomol Soc Am 113. https://doi.org/10.1093/aesa/saz065
Tingle FC, Mitchell ER (1977) Seasonal populations of armyworms and loopers at Hastings. Florida The Florida Entomologist 60:115–122. https://doi.org/10.2307/3494389
Wallner WE (1987) Factors affecting insect population dynamics: differences between outbreak and non-outbreak species. Annu Rev Entomol 32:317–340. https://doi.org/10.1146/annurev.en.32.010187.001533
Webber BL, Yates CJ, Le Maitre DC et al (2011) Modelling horses for novel climate courses: insights from projecting potential distributions of native and alien Australian acacias with correlative and mechanistic models. Divers Distrib 17:978–1000. https://doi.org/10.1111/j.1472-4642.2011.00811.x
Wu L-H, Hill MP, Thomson LJ, Hoffmann AA (2018) Assessing the current and future biological control potential of Trichogramma ostriniae on its hosts Ostrinia furnacalis and Ostrinia nubilalis. Pest Manag Sci 74:1513–1523. https://doi.org/10.1002/ps.4841
Yonow T, Ramirez-Villegas J, Abadie C, Darnell RE, Ota N, Kriticos DJ (2019) Black Sigatoka in bananas: ecoclimatic suitability and disease pressure assessments. PLoS One 14:e0220601. https://doi.org/10.1371/journal.pone.0220601
Zulin D, Ávila CJ, Schlick-Souza EC (2018) Population fluctuation and vertical distribution of the soybean looper (Chrysodeixis includens) in soybean culture. Am J Plant Sci 09:1544–1556. https://doi.org/10.4236/ajps.2018.97113
Acknowledgements
We would like to thank the Instituto Mato-Grossensse de Algodão (IMA) for providing the moth seasonality data and Dr. Darren Kriticos for providing the CRU climate time series dataset.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001 and was supported by the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. JRSS, RSR, and RSS performed Chrysodeixis includens occurrence data collection, model parameter setting, and analysis. JRSS wrote the first draft, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Soares, J.R.S., da Silva, R.S., Ramos, R.S. et al. Distribution and invasion risk assessments of Chrysodeixis includens (Walker, [1858]) (Lepidoptera: Noctuidae) using CLIMEX. Int J Biometeorol 65, 1137–1149 (2021). https://doi.org/10.1007/s00484-021-02094-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-021-02094-0