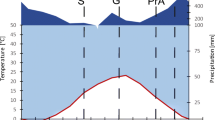
Abstract
The distribution of nitrogen (N) and carbon (C) pools in 100-year-old sessile oaks was investigated in situ at four key periods of tree phenology. Leaves, twigs, trunk and roots were sampled, and total non-structural nitrogen compounds (TNNC) and total non-structural carbohydrates (TNC) were quantified. TNC concentrations decreased more than 50 % between February and May, especially in the above-ground parts. During the same period, TNNC concentrations drastically decreased especially in twigs (more than 55 % below the winter baseline). This indicates high sink strength of new organs at bud break. TNC concentrations increased in summer in all tree compartments highlighting the TNC storage. TNNC deposition for storage constitution began in autumn during leaf senescence. The first organs acting as a N store were coarse roots followed by twigs; whereas all oak tree compartments were potential C store organs except phloem tissue. Arginine and asparagine were the two prevailing amino acids involved in both storage and transport. Besides identifying the compounds involved in the storage and transportation of N, our results highlight that oak seasonal cycles of C and N are not in phase: after intensive use of C and N stores in spring, the replenishment of N stores lags behind that of C stores.


Similar content being viewed by others
References
Barbaroux C, Bréda N (2002) Contrasting distribution and seasonal dynamics of carbohydrate reserves in stem wood of adult ring-porous sessile oak and diffuse-porous beech trees. Tree Physiol 22(17):1201–1210. doi:10.1093/treephys/22.17.1201
Barbaroux C, Bréda N, Dufrêne E (2003) Distribution of above-ground and below-ground carbohydrate reserves in adult trees of two contrasting broad-leaved species (Quercus petraea and Fagus sylvatica). New Phytol 157(3):605–615
Black BL, Fuchigami LH, Coleman GD (2002) Partitioning of nitrate assimilation among leaves, stems and roots of poplar. Tree Physiol 22(10):717–724
Bollmark L, Sennerby-Forsse L, Ericsson T (1999) Seasonal dynamics and effects of nitrogen supply rate on nitrogen and carbohydrate reserves in cutting-derived Salix viminalis plants. Can J For Res 29(1):85–94. doi:10.1139/cjfr-29-1-85
Bradford M (1976) A Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Canton FR, Suarez MF, Canovas FM (2005) Molecular aspects of nitrogen mobilization and recycling in trees. Photosynth Res 83(2):265–278
Cerasoli S, Maillard P, Scartazza A, Brugnoli E, Chaves MM, Pereira JS (2004) Carbon and nitrogen winter storage and remobilisation during seasonal flush growth in two-year-old cork oak (Quercus suber L.) saplings. Ann For Sci 61(7):721–729. doi:10.1051/forest:2004058 ISSN 1286-4560
Coleman GD, Chen THH (1993) Sequence of a poplar bark storage protein gene. Plant Physiol 102(4):1347–1348
Cooke JEK, Weih M (2005) Nitrogen storage and seasonal nitrogen cycling in Populus: bridging molecular physiology and ecophysiology. New Phytol 167(1):19–30
Courty P-E, Buée M, Diedhiou AG, Frey-Klett P, Le Tacon F, Rineau F, Turpault M-P, Uroz S, Garbaye J (2010) The role of ectomycorrhizal communities in forest ecosystem processes: new perspectives and emerging concepts. Soil Biol Biochem 42(5):679–698
Damesin C, Lelarge C (2003) Carbon isotope composition of current-year shoots from Fagus sylvatica in relation to growth, respiration and use of reserves. Plant Cell Environ 26(2):207–219
Dickson RE (1989) Carbon and nitrogen allocation in trees. Ann Sci For 46:S631–S647. doi:10.1051/forest:198905ART0142
Dong SF, Cheng LL, Fuchigami LH New root growth in relation to nitrogen reserves of young Gala/M26 apple trees. In: Neilsen DFBNGPF (ed) 4th International Symposium on Mineral Nutrition of Deciduous Fruit Crops, Penticton, Canada, Aug 13-18 2000. International Society Horticultural Science, Canada, pp 365–369
El Zein R, Maillard P, Breda N, Marchand J, Montpied P, Gerant D (2011) Seasonal changes of C and N non-structural compounds in the stem sapwood of adult sessile oak and beech trees. Tree Physiol 31(8):843–854. doi:10.1093/treephys/tpr074
Frak E, Millard P, Le Roux X, Guillaumie S, Wendler R (2002) Coupling sap flow velocity and amino acid concentrations as an alternative method to N-15 labeling for quantifying nitrogen remobilization by walnut trees. Plant Physiol 130(2):1043–1053. doi:10.1104/pp.002139
Gansert D, Sprick W (1998) Storage and mobilization of nonstructural carbohydrates and biomass development of beech seedlings (Fagus sylvatica L.) under different light regimes. Trees Struct Funct 12((5):247–257. doi:10.1007/pl00009715
Gaufichon L, Reisdorf-Cren Ml, Rothstein SJ, Chardon F, Suzuki A (2010) Biological functions of asparagine synthetase in plants. Plant Sci 179(3):141–153
Gessler A, Schneider S, Weber P, Hanemann U, Rennenberg H (1998) Soluble N compounds in trees exposed to high loads of N: a comparison between the roots of Norway spruce (Picea abies) and beech (Fagus sylvatica) trees grown under field conditions. New Phytol 138(3):385–399
Gojon A, Bussi C, Grignon C, Salsac L (1991) Distribution of NO3− reduction between roots and shoots of peachtree seedlings as affected by NO3− uptake rate. Physiol Plant 82(4):505–512
Grassi G, Millard P, Gioacchini P, Tagliavini M (2003) Recycling of nitrogen in the xylem of Prunus avium trees starts when spring remobilization of internal reserves declines. Tree Physiol 23(15):1061–1068
Guak S, Neilsen D, Millard P, Wendler R, Neilsen GH (2003) Determining the role of N remobilization for growth of apple (Malus domestica Borkh.) trees by measuring xylem-sap N flux. J Exp Botany 54(390):2121–2131. doi:10.1093/jxb/erg228 ISSN 0022-0957
Hoch G, Richter A, Korner C (2003) Non-structural carbon compounds in temperate forest trees. Plant Cell Environ 26(7):1067–1081
Kang SM, Titus JS (1980) Activity profiles of enzymes involved in glutamine and glutamate metabolism in the apple during autumnal senescence. Physiol Plant 50(3):291–297. doi:10.1111/j.1399-3054.1980.tb04465.x
Kramer PJ, Kozlowski T (1979) Physiology of woody plants, vol 811
Langheinrich U, Tischner R (1991) Vegetative storage proteins in Poplar: induction and characterization of a 32- and a 36-kilodalton polypeptide. Plant Physiol 97(3):1017–1025. doi:10.1104/pp.97.3.1017
Larsen TM, Boehlein SK, Schuster SM, Richards NGJ, Thoden JB, Holden HM, Rayment I (2000) Three-dimensional structure of Escherichia coli asparagine synthetase B: a short journey from substrate to product (vol 38, pg 16146, 1999). Biochemistry 39 (24):7330-7330. doi:10.1021/bi005109y
Malaguti D, Millard P, Wendler R, Hepburn A, Tagliavini M (2001) Translocation of amino acids in the xylem of apple (Malus domestica Borkh.) trees in spring as a consequence of both N remobilization and root uptake. J Exp Bot 52(361):1665–1671
Millard P (1996) Ecophysiology of the internal cycling of nitrogen for tree growth. Z Pflanzen Bodenk 159(1):1–10
Millard P, Grelet G-a (2010) Nitrogen storage and remobilization by trees: ecophysiological relevance in a changing world. Tree Physiol 30(9):1083–1095. doi:10.1093/treephys/tpq042
Millard P, Wendler R, Grassi G, Grelet G-A, Tagliavini M (2006) Translocation of nitrogen in the xylem of field-grown cherry and poplar trees during remobilization. Tree Physiol 26(4):527–536. doi:10.1093/treephys/26.4.527
Nuevo M, Auger G, Blanot D, d’Hendecourt L (2008) A detailed study of the amino acids produced from the vacuum UV irradiation of interstellar ice analogs. Orig Life Evol Biosph 38(1):37–56. doi:10.1007/s11084-007-9117-y
Quartieri M, Millard P, Tagliavini M (2002) Storage and remobilisation of nitrogen by pear (Pyrus communis L.) trees as affected by timing of N supply. Eur J Agron 17(2):105–110
Rognes SE (1975) Glutamine-dependent asparagine synthetase from Lupinus luteus. Phytochemistry 14(9):1975–1982. doi:10.1016/0031-9422(75)83108-6
Sagisaka S (1974) Effect of low temperature on amino acid metabolism in wintering poplar: arginine–glutamine relationships. Plant Physiol 53(2):319
Sagisaka S, Araki T (1983) Amino-acid pools in perennial plants at the wintering stage and at the beginning of growth. Plant Cell Physiol 24(3):479–494
Sauter J Jr, van Cleve B (1994) Storage, mobilization and interrelations of starch, sugars, protein and fat in the ray storage tissue of poplar trees. Trees Struct Funct 8(6):297–304
Schmidt S, Stewart GR (1998) Transport, storage and mobilization of nitrogen by trees and shrubs in the wet/dry tropics of northern Australia. Tree Physiol 18(6):403–410
Sievering H, Tomaszewski T, Torizzo J (2007) Canopy uptake of atmospheric N deposition at a conifer forest: part I—canopy N budget, photosynthetic efficiency and net ecosystem exchange. Tellus Ser B-Chem Phys Meteorol 59(3):483–492. doi:10.1111/j.1600-0889.2007.00264.x
Spann TM, Beede RH, DeJong TM (2008) Seasonal carbohydrate storage and mobilization in bearing and non-bearing pistachio (Pistacia vera) trees. Tree Physiol 28(2):207–213. doi:10.1093/treephys/28.2.207
Stassen P, Strydom DK, Stindt HW (1981) Seasonal changes in carbohydrate fractions of young ‘Kakamas’ peach trees. Agroplantae 13:47–53
Staswick PE (1994) Storage proteins of vegetative plant tissue. Annu Rev Plant Physiol Plant Mol Biol 45:303–322
Tagliavini M, Quartieri M, Millard P (1997) Remobilised nitrogen and root uptake of nitrate for spring leaf growth, flowers and developing fruits of pear (Pyrus communis L.) trees. Plant Soil 195(1):137–142
Taylor B (1967) Storage and mobilization of nitrogen in fruit trees: a review. J Aust Inst Agric Sci 33:23–29
Thomas FM, Hilker C (2000) Nitrate reduction in leaves and roots of young pedunculate oaks (Quercus robur) growing on different nitrate concentrations. Environ Exp Bot 43(1):19–32. doi:10.1016/s0098-8472(99)00040-4
Tromp J (1983) Nutrient reserves in roots of fruit trees, in particular carbohydrates and nitrogen. Plant Soil 71(1):401–413
Tromp J, Ovaa JC (1979) Uptake and distribution of nitrogen in young apple trees after application of nitrate or ammonium, with special reference to asparagine and arginine. Physiol Plant 45(1):23–28. doi:10.1111/j.1399-3054.1979.tb01657.x
Valenzuela Núñez L, Gérant D, Maillard P, Bréda N, González Cervantes G, Sánchez Cohen I (2011) Evidence for 26KDa vegetative storage protein in the stem sapwood of mature pedunculate oak. Intersciencia 36(2):142–147
Vitasse Y, Delzon S, Dufrene E, Pontailler JY, Louvet JM, Kremer A, Michalet R (2009) Leaf phenology sensitivity to temperature in European trees: do within-species populations exhibit similar responses? Agric For Meteorol 149(5):735–744. doi:10.1016/j.agrformet.2008.10.019
Wildhagen H, Durr J, Ehlting B, Rennenberg H (2010) Seasonal nitrogen cycling in the bark of field-grown Grey poplar is correlated with meteorological factors and gene expression of bark storage proteins. Tree Physiol 30(9):1096–1110. doi:10.1093/treephys/tpq018
Acknowledgments
Financial support was provided by CNRS through the EC2CO project “Linking models and data to evaluate nitrogen feedbacks on potential future carbon sequestration at local, regional and global scales”. The authors would like to acknowledge Michèle Vieil and Patricia Le Thuaut for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Matyssek.
Rights and permissions
About this article
Cite this article
Bazot, S., Barthes, L., Blanot, D. et al. Distribution of non-structural nitrogen and carbohydrate compounds in mature oak trees in a temperate forest at four key phenological stages. Trees 27, 1023–1034 (2013). https://doi.org/10.1007/s00468-013-0853-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-013-0853-5