Abstract
Background
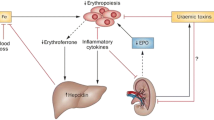
Anemia is a common complication of chronic kidney disease (CKD) and oral iron is recommended as initial therapy. However, response to iron therapy in children with non-dialysis CKD has not been formally assessed.
Methods
We reviewed medical records of pediatric patients with stages II–IV CKD followed in two New York metropolitan area medical centers between 2010 and 2020 and identified subjects who received oral iron therapy. Response to therapy at follow-up visits was assessed by improvement of hemoglobin, resolution of anemia by the 2012 KDIGO definition, and changes in iron status. Potential predictors of response were examined using regression analyses (adjusted for age, sex, eGFR, and center).
Results
Study criteria were met by 65 children (median age 12 years, 35 males) with a median time between visits of 81 days. Median eGFR was 44 mL/min/1.73 m2, and 40.7% had glomerular CKD etiology. Following iron therapy, hemoglobin improved from 10.2 to 10.8 g/dL (p < 0.001), hematocrit from 31.3 to 32.8% (p < 0.001), serum iron from 49 to 66 mcg/dL (p < 0.001), and transferrin saturation from 16 to 21.4% (p < 0.001). There was no significant change in serum ferritin (55.0 to 44.9 ng/mL). Anemia (defined according to KDIGO) resolved in 29.3% of children. No improvement in hemoglobin/hematocrit was seen in 35% of children, and no transferrin saturation improvement in 26.9%. There was no correlation between changes in hemoglobin and changes in transferrin saturation/serum iron, but there was an inverse correlation between changes in hemoglobin and changes in ferritin. The severity of anemia and alkaline phosphatase at baseline inversely correlated with treatment response.
Conclusions
Anemia was resistant to 3 months of oral iron therapy in ~ 30% of children with CKD. Children with more severe anemia at baseline had better treatment response, calling for additional studies to refine approaches to iron therapy in children with anemia of CKD and to identify additional predictors of treatment response.



Similar content being viewed by others
Data Availability
Relevant data will be made available upon reasonable request, assuring confidentiality of study participants.
References
Fadrowski JJ, Pierce CB, Cole SR, Moxey-Mims M, Warady BA, Furth SL (2008) Hemoglobin decline in children with chronic kidney disease: baseline results from the chronic kidney disease in children prospective cohort study. Clin J Am Soc Nephrol 3:457–462
Koshy SM, Geary DF (2008) Anemia in children with chronic kidney disease. Pediatr Nephrol 23:209–219
Akchurin O, Molino AR, Schneider MF, Atkinson MA, Warady BA, Furth SL (2023) Longitudinal relationship between anemia and statural growth impairment in children and adolescents with nonglomerular CKD: findings from the Chronic Kidney Disease in Children (CKiD) study. Am J Kidney Dis 81:457-465.e1
Ganz T, Nemeth E (2016) Iron balance and the role of hepcidin in chronic kidney disease. Semin Nephrol 36:87–93
Atkinson MA, Kim JY, Roy CN, Warady BA, White CT, Furth SL (2015) Hepcidin and risk of anemia in CKD: a cross-sectional and longitudinal analysis in the CKiD cohort. Pediatr Nephrol 30:635–643
McMurray J, Parfrey P, Adamson JW, Aljama P, Berns JS, Bohlius J, Drüeke TB, Finkelstein FO, Fishbane S, Ganz T (2012) Kidney disease: improving global outcomes (KDIGO) anemia work group. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl :279–335
Patino E, Akchurin O (2022) Erythropoiesis-independent effects of iron in chronic kidney disease. Pediatr Nephrol 37:777–788
Akchurin OM, Schneider MF, Mulqueen L, Brooks ER, Langman CB, Greenbaum LA, Furth SL, Moxey-Mims M, Warady BA, Kaskel FJ, Skversky AL (2014) Medication adherence and growth in children with CKD. Clin J Am Soc Nephrol 9:1519–1525
Babitt JL, Eisenga MF, Haase VH, Kshirsagar AV, Levin A, Locatelli F, Małyszko J, Swinkels DW, Tarng D-C, Cheung M (2021) Controversies in optimal anemia management: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int 99:1280–1295
Hollowell JG, van Assendelft OW, Gunter EW, Lewis BG, Najjar M, Pfeiffer C (2005) Hematological and iron-related analytes–reference data for persons aged 1 year and over: United States, 1988–94. Vital Health Stat 11:1–156
Schwartz GJ, Munoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637
Palmer SC, Navaneethan SD, Craig JC, Johnson DW, Tonelli M, Garg AX, Pellegrini F, Ravani P, Jardine M, Perkovic V (2010) Meta-analysis: erythropoiesis-stimulating agents in patients with chronic kidney disease. Ann Intern Med 153:23–33
Rheault MN, Molony JT, Nevins T, Herzog CA, Chavers BM (2017) Hemoglobin of 12 g/dl and above is not associated with increased cardiovascular morbidity in children on hemodialysis. Kidney Int 91:177–182
Akchurin OM (2019) Chronic kidney disease and dietary measures to improve outcomes. Pediatr Clin North Am 66:247–267
Akchurin O, Sureshbabu A, Doty SB, Zhu YS, Patino E, Cunningham-Rundles S, Choi ME, Boskey A, Rivella S (2016) Lack of hepcidin ameliorates anemia and improves growth in an adenine-induced mouse model of chronic kidney disease. Am J Physiol Renal Physiol 311:F877–F889
Wheby MS (1980) Effect of iron therapy on serum ferritin levels in iron-deficiency anemia. Blood 56:138–140
Prentice AM, Doherty CP, Abrams SA, Cox SE, Atkinson SH, Verhoef H, Armitage AE, Drakesmith H (2012) Hepcidin is the major predictor of erythrocyte iron incorporation in anemic African children. Blood 119:1922–1928
Moretti D, Goede JS, Zeder C, Jiskra M, Chatzinakou V, Tjalsma H, Melse-Boonstra A, Brittenham G, Swinkels DW, Zimmermann MB (2015) Oral iron supplements increase hepcidin and decrease iron absorption from daily or twice-daily doses in iron-depleted young women. Blood 126:1981–1989
Patino E, Bhatia D, Vance SZ, Antypiuk A, Uni R, Campbell C, Castillo CG, Jaouni S, Vinchi F, Choi ME, Akchurin O (2022) Iron therapy mitigates chronic kidney disease progression by regulating intracellular iron status of kidney macrophages. JCI insight 8:e159235
Okam MM, Koch TA, Tran M-H (2017) Iron supplementation, response in iron-deficiency anemia: analysis of five trials. Am J Med 130:991-e991
Van Wyck DB, Roppolo M, Martinez CO, Mazey RM, McMurray S (2005) A randomized, controlled trial comparing IV iron sucrose to oral iron in anemic patients with nondialysis-dependent CKD. Kidney Int 68:2846–2856
Macdougall IC, Bock AH, Carrera F, Eckardt K-U, Gaillard C, Van Wyck D, Roubert B, Nolen JG, Roger SD (2014) FIND-CKD: a randomized trial of intravenous ferric carboxymaltose versus oral iron in patients with chronic kidney disease and iron deficiency anaemia. Nephrol Dial Transplant 29:2075–2084
Spinowitz BS, Kausz AT, Baptista J, Noble SD, Sothinathan R, Bernardo MV, Brenner L, Pereira BJG (2008) Ferumoxytol for treating iron deficiency anemia in CKD. J Am Soc Nephrol 19:1599–1605
Powers JM, Buchanan GR, Adix L, Zhang S, Gao A, McCavit TL (2017) Effect of low-dose ferrous sulfate vs iron polysaccharide complex on hemoglobin concentration in young children with nutritional iron-deficiency anemia: a randomized clinical trial. JAMA 317:2297–2304
Guedes M, Muenz DG, Zee J, Bieber B, Stengel B, Massy ZA, Mansencal N, Wong MMY, Charytan DM, Reichel H (2021) Serum biomarkers of iron stores are associated with increased risk of all-cause mortality and cardiovascular events in nondialysis CKD patients, with or without anemia. J Am Soc Nephrol 32:2020–2030
Patino E, Doty SB, Bhatia D, Meza K, Zhu YS, Rivella S, Choi ME, Akchurin O (2020) Carbonyl iron and iron dextran therapies cause adverse effects on bone health in juveniles with chronic kidney disease. Kidney Int 98:1210–1224
Akchurin O, Patino E, Dalal V, Meza K, Bhatia D, Brovender S, Zhu YS, Cunningham-Rundles S, Perelstein E, Kumar J, Rivella S, Choi ME (2019) Interleukin-6 contributes to the development of anemia in juvenile CKD. Kidney Int Rep 4:470–483
Kalra PA, Bhandari S, Saxena S, Agarwal D, Wirtz G, Kletzmayr J, Thomsen LL, Coyne DW (2016) A randomized trial of iron isomaltoside 1000 versus oral iron in non-dialysis-dependent chronic kidney disease patients with anaemia. Nephrol Dial Transplant 31:646–655
Womack R, Berru F, Panwar B, Gutiérrez OM (2020) Effect of ferric citrate versus ferrous sulfate on iron and phosphate parameters in patients with iron deficiency and CKD: a randomized trial. Clin J Am Soc Nephrol 15:1251–1258
Warady BA, Kausz A, Lerner G, Brewer ED, Chadha V, Brugnara C, Dahl NV, Watkins SL (2004) Iron therapy in the pediatric hemodialysis population. Pediatr Nephrol 19:655–661
Atkinson MA, Pierce CB, Fadrowski JJ, Benador NM, White CT, Turman MA, Pan CG, Abraham AG, Warady BA, Furth SL (2012) Association between common iron store markers and hemoglobin in children with chronic kidney disease. Pediatr Nephrol 27:2275–2283
Prats M, Font R, García C, Muñoz-Cortés M, Cabré C, Jariod M, Romeu M, Giralt M, Martinez-Vea A (2014) Oxidative stress markers in predicting response to treatment with ferric carboxymaltose in nondialysis chronic kidney disease patients. Clin Nephrol 81:419–426
Macdougall IC, Bock AH, Carrera F, Eckardt K-U, Gaillard C, Van Wyck D, Meier Y, Larroque S, Perrin A, Roger SD (2017) Erythropoietic response to oral iron in patients with nondialysis-dependent chronic kidney disease in the FIND-CKD trial. Clin Nephrol 88:301
Gaillard CA, Bock AH, Carrera F, Eckardt K-U, Van Wyck DB, Bansal SS, Cronin M, Meier Y, Larroque S, Roger SD (2016) Hepcidin response to iron therapy in patients with non-dialysis dependent CKD: an analysis of the FIND-CKD trial. PLoS One 11:e0157063
Stancu S, Bârsan L, Stanciu A, Mircescu G (2010) Can the response to iron therapy be predicted in anemic nondialysis patients with chronic kidney disease? Clin J Am Soc Nephrol 5:409–416
Takasawa K, Takaeda C, Wada T, Ueda N (2018) Optimal serum ferritin levels for iron deficiency anemia during oral iron therapy (OIT) in Japanese hemodialysis patients with minor inflammation and benefit of intravenous iron therapy for OIT-nonresponders. Nutrients 10:428
Haarhaus M, Brandenburg V, Kalantar-Zadeh K, Stenvinkel P, Magnusson P (2017) Alkaline phosphatase: a novel treatment target for cardiovascular disease in CKD. Nat Rev Nephrol 13:429–442
Tanaka M, Yoshida K, Fukuma S, Ito K, Matsushita K, Fukagawa M, Fukuhara S, Akizawa T (2016) Effects of secondary hyperparathyroidism treatment on improvement in anemia: results from the MBD-5D study. PLoS One 11:e0164865
Van Vuren AJ, Gaillard CAJM, Eisenga MF, Van Wijk R, Van Beers EJ (2019) The EPO-FGF23 signaling pathway in erythroid progenitor cells: opening a new area of research. Front Physiol 10:304
Coe LM, Madathil SV, Casu C, Lanske B, Rivella S, Sitara D (2014) FGF-23 is a negative regulator of prenatal and postnatal erythropoiesis. J Biol Chem 289:9795–9810
Kalantar-Zadeh K, Lee GH, Miller JE, Streja E, Jing J, Robertson JA, Kovesdy CP (2009) Predictors of hyporesponsiveness to erythropoiesis-stimulating agents in hemodialysis patients. Am J Kidney Dis 53:823–834
Badve SV, Zhang L, Coombes JS, Pascoe EM, Cass A, Clarke P, Ferrari P, McDonald SP, Morrish AT, Pedagogos E (2015) Association between serum alkaline phosphatase and primary resistance to erythropoiesis stimulating agents in chronic kidney disease: a secondary analysis of the HERO trial. Can J Kidney Health Dis 2:66
Pacara MF (2022) Safety and effectiveness of the liposomal iron in the treatment of iron deficiency anemia in pediatric patients with chronic kidney disease on hemodialysis. Kidney Int Rep 7:S104
Funding
Regulatory and data management support for this study was partially provided by the Weill Cornell Medicine Clinical and Translational Science Center (funded by the NIH NCATS UL1TR002384). O.A. is supported by the R03 DK135897 and K08 DK114558 from the NIH NIDDK, the Hartwell Foundation, the Sy Syms, and the Bass Foundations. The content does not necessary represent the official views of any of the supporting entities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The results of this study were partially presented at the 54th Annual Scientific Meeting of the European Society of Pediatric Nephrology (ESPN) in Ljubljana, Slovenia (June 2022), and at the 19th International Pediatric Nephrology Association (IPNA) Congress in Calgary, Canada (September 2022).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meza, K., Biswas, S., Talmor, C. et al. Response to oral iron therapy in children with anemia of chronic kidney disease. Pediatr Nephrol 39, 233–242 (2024). https://doi.org/10.1007/s00467-023-06048-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-06048-z