Abstract
Background
Hyperactivity of the alternative complement pathway is the principle defect in C3 glomerulopathies (C3G). Eculizumab, a monoclonal antibody that binds C5 to prevent formation of the membrane attack complex, has been shown to be beneficial in some patients with this disease.
Methods
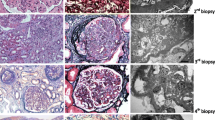
In this open-label, proof-of-concept efficacy-and-safety study, a patient with the initial diagnosis of dense deposit disease (DDD) and allograft recurrence of C3 glomerulonephritis (C3GN) was treated with eculizumab every other week for 1 year. The patient had pathological evidence of C3GN and proteinuria >1 g/day at enrollment. He underwent graft biopsy before enrollment and repeat biopsy at 6 and 12 months.
Results
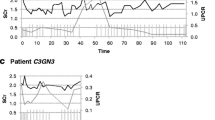
Although no mutations were identified in complement genes, functional studies were positive for C3 nephritic factors and elevated levels of soluble membrane attack complex (sMAC). On therapy, sMAC levels normalized and although proteinuria initially decreased, it increased reaching pre-treatment levels at 12 months. Although serum creatinine remained stable, repeat allograft biopsies showed progression of disease.
Conclusions
Clinical and histopathologic data suggest a partial response to eculizumab in this patient. While eculizumab blocked activation of the terminal complement cascade, persistent dysregulation of the alternative pathway remained, indicating eculizumab alone cannot control disease in this patient. Additional research is required to identify effective anticomplement therapy for this group of C3G patients.



Similar content being viewed by others
References
Habib R, Kleinknecht C, Gubler MC, Maiz HB (1973) Idiopathic membranoproliferative glomerulonephritis. Morphology and natural history. Perspect Nephrol Hypertens 1:491–514
Strife CF, McEnery PT, McAdams AJ, West CD (1977) Membranoproliferative glomerulonephritis with disruption of the glomerular basement membrane. Clin Nephrol 7:65–72
Levy M, Gubler MC, Sich M, Beziau A, Habib R (1978) Immunopathology of membranoproliferative glomerulonephritis with subendothelial deposits (type I MPGN). Clin Immunol Immunopathol 10:477–492
Fakhouri F, Fremeaux-Bacchi V, Noel LH, Cook HT, Pickering MC (2010) C3 glomerulopathy: a new classification. Nat Rev Nephrol 6:494–499
Servais A, Fremeaux-Bacchi V, Lequintrec M, Salomon R, Blouin J, Knebelmann B, Grunfeld JP, Lesavre P, Noel LH, Fakhouri F (2007) Primary glomerulonephritis with isolated C3 deposits: a new entity which shares common genetic risk factors with haemolytic uraemic syndrome. J Med Genet 44:193–199
Sethi S, Fervenza FC, Zhang Y, Nasr SH, Leung N, Vrana J, Cramer C, Nester CM, Smith RJ (2011) Proliferative glomerulonephritis secondary to dysfunction of the alternative pathway of complement. Clin J Am Soc Nephrol 6:1009–1017
Nasr SH, Valeri AM, Appel GB, Sherwinter J, Stokes MB, Said SM, Markowitz GS, D'Agati VD (2009) Dense deposit disease: clinicopathologic study of 32 pediatric and adult patients. Clin J Am Soc Nephrol 4:22–32
Vivarelli M, Pasini A, Emma F (2012) Eculizumab for the treatment of dense deposit disease. N Engl J Med 366:1163–1165
Bomback AS, Smith RJ, Barile GR, Zhang Y, Heher EC, Herlitz L, Stokes MB, Markowitz GS, D'Agati VD, Canetta PA, Radhakrishnan J, Appel GB (2012) Eculizumab for dense deposit disease and C3 glomerulonephritis. Clin J Am Soc Nephrol 7:748–756
Daina E, Noris M, Remuzzi G (2012) Eculizumab in a patient with dense deposit disease. N Engl J Med 366:1161–1163
McCaughan JA, O'Rourke DM, Courtney AE (2012) Recurrent dense deposit disease after renal transplantation: an emerging role for complementary therapies. Am J Transplant 12:1046–1051
Abrera-Abeleda MA, Nishimura C, Smith JL, Sethi S, McRae JL, Murphy BF, Silvestri G, Skerka C, Jozsi M, Zipfel PF, Hageman GS, Smith RJ (2006) Variations in the complement regulatory genes factor H (CFH) and factor H related 5 (CFHR5) are associated with membranoproliferative glomerulonephritis type II (dense deposit disease). J Med Genet 43:582–589
Zhang Y, Meyer NC, Wang K, Nishimura C, Frees K, Jones M, Katz LM, Sethi S, Smith RJ (2012) Causes of alternative pathway dysregulation in dense deposit disease. Clin J Am Soc Nephrol 7:265–274
Maga TK, Nishimura CJ, Weaver AE, Frees KL, Smith RJ (2010) Mutations in alternative pathway complement proteins in American patients with atypical hemolytic uremic syndrome. Hum Mutat 31:E1445–E1460
Provan D, Butler T, Evangelista ML, Amadori S, Newland AC, Stasi R (2007) Activity and safety profile of low-dose rituximab for the treatment of autoimmune cytopenias in adults. Haematologica 92:1695–1698
Zaja F, Vianelli N, Volpetti S, Battista ML, Defina M, Palmieri S, Bocchia M, Medeot M, De Luca S, Ferrara F, Isola M, Baccarani M, Fanin R (2010) Low-dose rituximab in adult patients with primary immune thrombocytopenia. Eur J Haematol 85:329–334
Sethi S, Nester CM, Smith RJ (2012) Membranoproliferative glomerulonephritis and C3 glomerulopathy: resolving the confusion. Kidney Int 81:434–441
Martinez-Barricarte R, Heurich M, Valdes-Canedo F, Vazquez-Martul E, Torreira E, Montes T, Tortajada A, Pinto S, Lopez-Trascasa M, Morgan BP, Llorca O, Harris CL, Rodriguez de Cordoba S (2010) Human C3 mutation reveals a mechanism of dense deposit disease pathogenesis and provides insights into complement activation and regulation. J Clin Invest 120:3702–3712
Gale DP, de Jorge EG, Cook HT, Martinez-Barricarte R, Hadjisavvas A, McLean AG, Pusey CD, Pierides A, Kyriacou K, Athanasiou Y, Voskarides K, Deltas C, Palmer A, Fremeaux-Bacchi V, de Cordoba SR, Maxwell PH, Pickering MC (2010) Identification of a mutation in complement factor H-related protein 5 in patients of Cypriot origin with glomerulonephritis. Lancet 376:794–801
Habbig S, Mihatsch MJ, Heinen S, Beck B, Emmel M, Skerka C, Kirschfink M, Hoppe B, Zipfel PF, Licht C (2009) C3 deposition glomerulopathy due to a functional factor H defect. Kidney Int 75:1230–1234
Smith RJ, Harris CL, Pickering MC (2011) Dense deposit disease. Mol Immunol 48:1604–1610
Cameron JS, Turner DR, Heaton J, Williams DG, Ogg CS, Chantler C, Haycock GB, Hicks J (1983) Idiopathic mesangiocapillary glomerulonephritis. Comparison of types I and II in children and adults and long-term prognosis. Am J Med 74:175–192
Schena FP, Pertosa G, Stanziale P, Vox E, Pecoraro C, Andreucci VE (1982) Biological significance of the C3 nephritic factor in membranoproliferative glomerulonephritis. Clin Nephrol 18:240–246
Schwertz R, Rother U, Anders D, Gretz N, Scharer K, Kirschfink M (2001) Complement analysis in children with idiopathic membranoproliferative glomerulonephritis: a long-term follow-up. Pediatr Allergy Immunol 12:166–172
Nester C, Stewart Z, Myers D, Jetton J, Nair R, Reed A, Thomas C, Smith R, Brophy P (2011) Pre-emptive eculizumab and plasmapheresis for renal transplant in atypical hemolytic uremic syndrome. Clin J Am Soc Nephrol 6:1488–1494
Parker C (2009) Eculizumab for paroxysmal nocturnal haemoglobinuria. Lancet 373:759–767
Kose O, Zimmerhackl LB, Jungraithmayr T, Mache C, Nurnberger J (2010) New treatment options for atypical hemolytic uremic syndrome with the complement inhibitor eculizumab. Semin Thromb Hemost 36:669–672
Acknowledgments
We are grateful to Helen Mundy and Cheryle Ludwig for assistance with the conduct of this study at the Division of Pediatric Nephrology at UMDNJ. This research was supported, in part, by the National Institutes of Health Grant DK074409 (to R.J.S.).
Disclosures
The investigators were responsible for study design, implementation, data analysis, and manuscript preparation.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gurkan, S., Fyfe, B., Weiss, L. et al. Eculizumab and recurrent C3 glomerulonephritis. Pediatr Nephrol 28, 1975–1981 (2013). https://doi.org/10.1007/s00467-013-2503-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2503-y