Abstract
Background
Xanthogranulomatous cholecystitis (XGC) is an uncommon variant of chronic cholecystitis which can resemble gallbladder adenocarcinoma (GAC) on preoperative imaging and present technical challenges in the performance of cholecystectomy. We examined our experience with each pathology to identify distinguishing characteristics that may guide patient counseling and surgical management.
Methods
A retrospective review of all pathologically confirmed cases of XGC and GAC following cholecystectomy between 2015 and 2021 at a single institution was performed. Clinical, biochemical, radiographic, and intraoperative features were compared.
Results
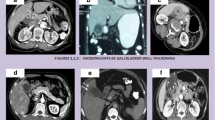
There were 37 cases of XGC and 20 cases of GAC. Patients with GAC were older (mean 70.3 years vs 58.0, p = 0.01) and exclusively female (100% vs 45.9%, p < 0.0001). There were no significant differences in accompanying symptoms between groups (nausea/vomiting, fevers, or jaundice). The mean maximum white blood cell count was elevated for XGC compared to GAC (16.4 vs 8.6 respectively, p = 0.044); however, there were no differences in the remainder of the biochemical profile, including bilirubin, liver transaminases, CEA, and CA 19–9. The presence of an intraluminal mass (61.1% vs 9.1%, p = 0.0001) and lymphadenopathy (18.8%. vs 0.0%, p = 0.045) were associated with malignancy, whereas gallbladder wall thickening as reported on imaging (87.9% vs 38.9%, p = 0.0008) and gallstones (76.5% vs. 50.0%, p = 0.053) were more often present with XGC. Cases of XGC more often had significant adhesions/inflammation (83.8% vs 55.0%, p = 0.03).
Conclusion
Clinical features that may favor benign chronic cholecystitis over gallbladder adenocarcinoma include younger age, male gender, current or prior leukocytosis, and the absence of an intraluminal mass or lymphadenopathy. Laparoscopic cholecystectomy is a safe surgical option for equivocal presentations. Intraoperative frozen section or intentional staging of more extensive procedures based upon final histopathology are valuable surgical strategies.


Similar content being viewed by others
References
Christensen AH, Ishak KG (1970) Benign tumors and pseudotumors of the gallbladder. Report of 180 cases. Arch Pathol 90(5):423–432
Goodman ZD, Ishak KG (1981) Xanthogranulomatous cholecystitis. Am J Surg Pathol 5(7):653–659. https://doi.org/10.1097/00000478-198110000-00007
Deng YL, Cheng NS, Zhang SJ, Ma WJ, Shrestha A, Li FY et al (2015) Xanthogranulomatous cholecystitis mimicking gallbladder carcinoma: an analysis of 42 cases. World J Gastroenterol 21(44):12653–12659
Spinelli A, Schumacher G, Pascher A, Lopez-Hanninen E, Al-Abadi H, Benckert C et al (2006) Extended surgical resection for xanthogranulomatous cholecystitis mimicking advanced gallbladder carcinoma: a case report and review of literature. World J Gastroenterol 12(14):2293–2296
Rahman R, Simoes EJ, Schmaltz C, Jackson CS, Ibdah JA (2017) Trend analysis and survival of primary gallbladder cancer in the United States: a 1973–2009 population-based study. Cancer Med. 6(4):874–880. https://doi.org/10.1002/cam4.1044
Duffy A, Capanu M, Abou-Alfa GK, Huitzil D, Jarnagin W, Fong Y et al (2008) Gallbladder cancer (GBC): 10-year experience at Memorial Sloan-Kettering Cancer Centre (MSKCC). J Surg Oncol 98(7):485–489. https://doi.org/10.1002/jso.21141
Guzmán-Valdivia G (2004) Xanthogranulomatous cholecystitis: 15 years’ experience. World J Surg 28(3):254–257. https://doi.org/10.1007/s00268-003-7161-y
Kwon AH, Matsui Y, Uemura Y (2004) Surgical procedures and histopathologic findings for patients with xanthogranulomatous cholecystitis. J Am Coll Surg 199(2):204–210. https://doi.org/10.1016/j.jamcollsurg.2004.03.018
Agrawal V, Goel A, Krishnani N, Pandey R, Agrawal S, Kapoor VK (2010) p53, carcinoembryonic antigen and carbohydrate antigen 19.9 expression in gall bladder cancer, precursor epithelial lesions and xanthogranulomatous cholecystitis. J Postgrad Med 56(4):262–266. https://doi.org/10.4103/0022-3859.70933
Hale MD, Roberts KJ, Hodson J, Scott N, Sheridan M, Toogood GJ (2014) Xanthogranulomatous cholecystitis: a European and global perspective. HPB (Oxford) 16(5):448–458. https://doi.org/10.1111/hpb.12152
Uchiyama K, Ozawa S, Ueno M, Hayami S, Hirono S, Ina S et al (2009) Xanthogranulomatous cholecystitis: the use of preoperative CT findings to differentiate it from gallbladder carcinoma. J Hepatobiliary Pancreat Surg 16(3):333–338. https://doi.org/10.1007/s00534-009-0067-9
Chang BJ, Kim SH, Park HY, Lim SW, Kim J, Lee KH et al (2010) Distinguishing xanthogranulomatous cholecystitis from the wall-thickening type of early-stage gallbladder cancer. Gut Liver 4(4):518–523. https://doi.org/10.5009/gnl.2010.4.4.518
Rammohan A, Cherukuri SD, Sathyanesan J, Palaniappan R, Govindan M (2014) Xanthogranulomatous cholecystitis masquerading as gallbladder cancer: can it be diagnosed preoperatively? Gastroenterol Res Pract 2014:253645. https://doi.org/10.1155/2014/253645
Rastogi A, Singh DK, Sakhuja P, Gondal R (2010) Florid xanthogranulomatous cholecystitis masquerading as invasive gallbladder cancer leading to extensive surgical resection. Indian J Pathol Microbiol 53(1):144–147. https://doi.org/10.4103/0377-4929.59209
Goshima S, Chang S, Wang JH, Kanematsu M, Bae KT, Federle MP (2010) Xanthogranulomatous cholecystitis: diagnostic performance of CT to differentiate from gallbladder cancer. Eur J Radiol 74(3):e79–e83. https://doi.org/10.1016/j.ejrad.2009.04.017
Parra JA, Acinas O, Bueno J, Güezmes A, Fernández MA, Fariñas MC (2000) Xanthogranulomatous cholecystitis: clinical, sonographic, and CT findings in 26 patients. AJR Am J Roentgenol 174(4):979–983. https://doi.org/10.2214/ajr.174.4.1740979
Lichtman JB, Varma VA (1987) Ultrasound demonstration of xanthogranulomatous cholecystitis. J Clin Ultrasound 15(5):342–345. https://doi.org/10.1002/jcu.1870150509
Casas D, Pérez-Andrés R, Jiménez JA, Mariscal A, Cuadras P, Salas M et al (1996) Xanthogranulomatous cholecystitis: a radiological study of 12 cases and a review of the literature. Abdom Imaging 21(5):456–460. https://doi.org/10.1007/s002619900104
Roberts KM, Parsons MA (1987) Xanthogranulomatous cholecystitis: clinicopathological study of 13 cases. J Clin Pathol 40(4):412–417
Osborn JB, Keeney GL, Jakub JW, Degnim AC, Boughey JC (2011) Cost-effectiveness analysis of routine frozen-section analysis of breast margins compared with reoperation for positive margins. Ann Surg Oncol 18(11):3204–3209. https://doi.org/10.1245/s10434-011-1956-0
Zanocco K, Heller M, Elaraj D, Sturgeon C (2013) Cost effectiveness of intraoperative pathology examination during diagnostic hemithyroidectomy for unilateral follicular thyroid neoplasms. J Am Coll Surg 217(4):702–710. https://doi.org/10.1016/j.jamcollsurg.2013.05.008
Bonaroti JW, Doane S, McCue PA, Winter JM (2016) Intraoperative frozen section analysis of the pancreas: a case report and review of the literature. Case Rep Pancreat Cancer 2:71–74
Ishizawa T, Bandai Y, Ijichi M, Kaneko J, Hasegawa K, Kokudo N (2010) Fluorescent cholangiography illuminating the biliary tree during laparoscopic cholecystectomy. Br J Surg 97(9):1369–1377. https://doi.org/10.1002/bjs.7125
Dip F, LoMenzo E, Sarotto L, Phillips E, Todeschini H, Nahmod M et al (2019) Randomized trial of near-infrared incisionless fluorescent cholangiography. Ann Surg 270(6):992–999. https://doi.org/10.1097/sla.0000000000003178
Broderick RC, Lee AM, Cheverie JN, Zhao B, Blitzer RR, Patel RJ et al (2021) Fluorescent cholangiography significantly improves patient outcomes for laparoscopic cholecystectomy. Surg Endosc 35(10):5729–5739. https://doi.org/10.1007/s00464-020-08045-x
Hiwatashi K, Okumura H, Setoyama T, Ando K, Ogura Y, Aridome K et al (2018) Evaluation of laparoscopic cholecystectomy using indocyanine green cholangiography including cholecystitis: a retrospective study. Medicine (Baltimore) 97(30):e11654. https://doi.org/10.1097/md.0000000000011654
Funding
No funding information to disclose for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Dr. Horgan is a consultant for Stryker Corporation, Intuitive Surgical, Fortimedix Surgical, and Alume Biosciences. Dr. Jacobsen is a consultant for Gore Medical and Viacyte. Dr. Sandler is a consultant for Intuitive Surgical and Boston Scientific. Dr. Fowler receives grant support from Bayer, Pfizer, Median, FNIH, Siemens, and GE, provides institutional consulting for GE and Bayer, was an expert witness for Quantix Bio, and has the following non-financial roles: ACR GI panel chair, Radiology Editorial Board, SAR portfolio director, ACR LI-RADS leadership. Dr. Broderick is a consultant for Stryker Corporation. Drs. Clary, Huang, Reeves, Serra, Goldhaber, An, and Hosseini have no relevant disclosures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, E.Y., Reeves, J.J., Broderick, R.C. et al. Distinguishing characteristics of xanthogranulomatous cholecystitis and gallbladder adenocarcinoma: a persistent diagnostic dilemma. Surg Endosc 38, 348–355 (2024). https://doi.org/10.1007/s00464-023-10461-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-023-10461-8