Abstract
Objective This study was designed to evaluate the feasibility of AlloMEM™, a novel lyophililzed human peritoneal membrane, at peritoneal reconstitution, and decreasing adhesion formation after temporary loop ileostomy.
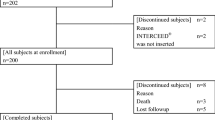
Methods In a pilot study, ten patients had AlloMEM™ used during elective formation of a temporary diverting loop ileostomy for benign or malignant colorectal disease. A blinded investigator and the operating surgeon analyzed the change in adhesion formation and peritoneal remodelling using ileostomy mobilization time and a 5-point adhesion scale grading intra-abdominally and at the subcutaneous and fascial levels.
Results The mean body mass index was 31 [standard deviation (SD) 5.6], and 40 % of patients had previous abdominal surgery. Ileostomies were reversed after a mean 14 weeks (SD 6.0). The mean ileostomy mobilization time was 27.2 min (SD 12.0). From baseline to ileostomy reversal, there were significant increases in adhesions at the subcutaneous (p = 0.0002) and fascial levels (p = 0.0024). The increased subcutaneous adhesions were associated with improved peritoneal remodeling. There was no significant increase in adhesions from baseline to ileostomy reversal at the intra-abdominal points (p = 0.9393) or around the ileostomy site (p = 0.6128). The median hospital length of stay was 2.6 days (range, 2–3). A single adverse event related to product packaging led to redesign of the packaging process.
Conclusions Use of AlloMEM™ in ileostomy closures suggested improvement in adhesions around the fascia and promotion of peritoneal remodeling. AlloMEM™ was safe, feasible, and easy to use in this pilot study. Comparative research is needed to assess the outcomes with this novel product.


Similar content being viewed by others
References
D’Haeninck A, Wolthuis AM, Penninckx F, D’Hondt M, D’Hoore A (2011) Morbidity after closure of a defunctioning loop ileostomy. Acta Chir Belg 111(3):136–141
Ihedioha U, Muhtaseb S, Kalmar K, Donnelly L, Muir V, Macdonald A (2010) Closure of loop ileostomies: is early discharge safe and achievable? Scott Med J 55(1):27–29
Russek K, George JM, Zafar N, Cuevas-Estandia P, Franklin M (2011) Laparoscopic loop ileostomy reversal: reducing morbidity while improving functional outcomes. JSLS 15(4):475–479
Winslet MC, Barsoum G, Pringle W, Fox K, Keighley MR (1991) Loop ileostomy after ileal pouch-anal anastomosis–is it necessary? Dis Colon Rectum 34(3):267–270
Wong KS, Remzi FH, Gorgun E, Arrigain S, Church JM, Preen M, Fazio VW (2005) Loop ileostomy closure after restorative proctocolectomy: outcome in 1,504 patients. Dis Colon Rectum 48(2):243–250
Ray NF, Denton WG, Thamer M, Henderson SC, Perry S (1998) Abdominal adhesiolysis: inpatient care and expenditures in the United States in 1994. J Am Coll Surg 186(1):1–9
Voskerician G, Jin J, Hunter SA, Williams CP, White M, Rosen MJ (2009) Human peritoneal membrane reduces the formation of intra-abdominal adhesions in ventral hernia repair: experimental study in a chronic hernia rat model. J Surg Res 157(1):108–114
Jin J, Voskerician G, Hunter SA, McGee MF, Cavazzola LT, Schomisch S, Harth K, Rosen MJ (2009) Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res 156(2):297–304
Diamond MP (1996) Reduction of adhesions after uterine myomectomy by Seprafilm membrane (HAL-F): a blinded, prospective, randomized, multicenter clinical study. Seprafilm Adhesion Study Group. Fertil Steril 66(6):904–910
Fazio VW, Cohen Z, Fleshman JW, van Goor H, Bauer JJ, Wolff BG, Corman M, Beart RWJ, Wexner SD, Becker JM et al (2006) Reduction in adhesive small-bowel obstruction by Seprafilm adhesion barrier after intestinal resection. Dis Colon Rectum 49(1):1–11
Remzi FH, Oncel M, Church JM, Senagore AJ, Delaney CP, Fazio VW (2003) An unusual complication after hyaluronate-based bioresorbable membrane (Seprafilm) application. Am Surg 69(4):356–357
Acknowledgments
This IRB-approved clinical study (ClinicalTrials.gov Identifier: NCT01113736) was cosponsored by Proxy Biomedical Limited (exclusive distributor of AlloMEM™) and Community Tissue Services (tissue bank processor of AlloMEM™).
Disclosures
The authors have no disclosure.
Author information
Authors and Affiliations
Corresponding author
Additional information
Poster Presentation, SAGES 2013 Annual Conference, Baltimore, MD.
Rights and permissions
About this article
Cite this article
Keller, D.S., Champagne, B.J., Stein, S.L. et al. Pilot study evaluating the efficacy of AlloMEM™ for prevention of intraperitoneal adhesions and peritoneal regeneration after loop ileostomy. Surg Endosc 27, 3891–3896 (2013). https://doi.org/10.1007/s00464-013-3004-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-013-3004-6