Abstract
Hyperthermophilic microorganisms play a key role in the hyper-thermophilic composting (HTC) technique. However, little information is available about the hyperthermophilic microorganisms prevalent in HTC systems, except for the Calditerricola satsumensis, Calditerricola yamamurae, and Thermaerobacter. To obtain effective hyper-thermophilic microorganisms, a continuous thermo-acclimation of the suitable thermophilic microorganisms was demonstrated in this study. Bacillus thermoamylovorans with high-temperature endurance (70 °C) were newly isolated from sludge composting, and an adequate slow heating rate (2 °C per cycle) was applied to further improve its thermostability. Finally, a strain with a maximum growth temperature of 80 °C was obtained. Moreover, structural and hydrophobic changes in cell proteins, the special amino acid content ratio, and the membrane permeability of the thermophilic bacterium after thermo-acclimation were evaluated for improved thermostability. In addition, the acclimated hyperthermophilic bacterium was further inoculated into the HTC system, and an excellent performance with a maximum operating temperature of 82 °C was observed.
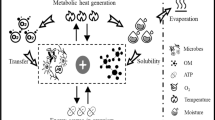
Graphic abstract










Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Zhou W, Liu M, Chai Y, Chen Y, Chen M, Wei Y, Min Y (2018) Discussion on establishment of technical standard system of domestic wastewater in villages and towns of China. Water Wastewater Eng 44(9):2–14. https://doi.org/10.13789/j.cnki.wwe1964.2018.0029
Zhou G, Gu Y, Yuan H, Gong Y, Wu Y (2020) Selecting sustainable technologies for disposal of municipal sewage sludge using a multi-criterion decision-making method: a case study from China. Resour Conserv Recy 161:104881. https://doi.org/10.1016/j.resconrec.2020.104881
Alves D, Villar I, Mato S (2019) Thermophilic composting of hydrocarbon residue with sewage sludge and fish sludge as cosubstrates: microbial changes and TPH reduction. J Environ Manag 239:30–37. https://doi.org/10.1016/j.jenvman.2019.03.028
Song U, Lee EJ (2010) Environmental and economical assessment of sewage sludge compost application on soil and plants in a landfill. Resour Conserv Recy 54(12):1109–1116. https://doi.org/10.1016/j.resconrec.2010.03.005
Tan XB, Yang LB, Zhang WW, Zhao XC (2020) Lipids production and nutrients recycling by microalgae mixotrophic culture in anaerobic digestate of sludge using wasted organics as carbon source. Bioresour Technol 297:122379. https://doi.org/10.1016/j.biortech.2019.122379
Raheem A, Sikarwar VS, He J, Dastyar W, Dionysiou DD, Wang W, Zhao M (2018) Opportunities and challenges in sustainable treatment and resource reuse of sewage sludge: a review. Chem Eng J 337:616–641. https://doi.org/10.1016/j.cej.2017.12.149
Wu LJ, Higashimori A, Qin Y, Hojo T, Kubota K, Li YY (2016) Comparison of hyper-thermophilic–mesophilic two-stage with single-stage mesophilic anaerobic digestion of waste activated sludge: process performance and microbial community analysis. Chem Eng J 290:290–301. https://doi.org/10.1016/j.cej.2016.01.067
Shao L, Wang T, Li T, Lü F, He P (2013) Comparison of sludge digestion under aerobic and anaerobic conditions with a focus on the degradation of proteins at mesophilic temperature. Bioresour Technol 140:131–137. https://doi.org/10.1016/j.biortech.2013.04.081
Zhang J, Chen G, Sun H, Zhou S, Zou G (2016) Straw biochar hastens organic matter degradation and produces nutrient-rich compost. Bioresour Technol 200:876–883. https://doi.org/10.1016/j.biortech.2015.11.016
Wei Y, Li J, Shi D, Liu G, Zhao Y, Shimaoka T (2017) Environmental challenges impeding the composting of biodegradable municipal solid waste: a critical review. Resour Conserv Recy 122:51–65. https://doi.org/10.1016/j.resconrec.2017.01.024
Ge J, Huang G, Huang J, Zeng J, Han L (2015) Modeling of oxygen uptake rate evolution in pig manure-wheat straw aerobic composting process. Chem Eng J 276:29–36. https://doi.org/10.1016/j.cej.2015.04.067
Onwosi CO, Igbokwe VC, Odimba JN, Eke IE, Nwankwoala MO, Iroh IN, Ezeogu LI (2017) Composting technology in waste stabilization: on the methods, challenges and future prospects. J Environ Manag 190:140–157. https://doi.org/10.1016/j.jenvman.2016.12.051
Bialobrzewski I, Miks-Krajnik M, Dach J, Markowski M, Czekala W, Gluchowska K (2015) Model of the sewage sludge-straw composting process integrating different heat generation capacities of mesophilic and thermophilic microorganisms. Waste Manag 43(5):72–83. https://doi.org/10.1016/j.wasman.2015.05.036
Deive FJ, Domínguez A, Barrio T, Moscoso F, Morán P, Longo MA, Sanromán MA (2010) Decolorization of dye reactive black 5 by newly isolated thermophilic microorganisms from geothermal sites in Galicia (Spain). J Hazard Mater 182(1–3):735–742. https://doi.org/10.1016/j.jhazmat.2010.06.096
Sunil K (2011) Composting of municipal solid waste. Crit Rev Biotechnol 31(2):112–136. https://doi.org/10.3109/07388551.2010.492207
Muibat OF, Ngole-Jeme VM, Babalola OO (2015) Diversity of acidophilic bacteria and archaea and their roles in bioremediation of acid mine drainage. Br Microbiol Res J 8(3):443–456. https://doi.org/10.9734/bmrj/2015/14365
Bhattacharya A, Pletschke BI (2014) Thermophilic bacilli and their enzymes in composting. In: Dinesh KM (ed) Composting for sustainable agriculture, 6th edn. Springer, New York, pp 103–124
Yu Z, Tang J, Liao H, Liu X, Zhou P, Zhi C, Christopher R, Zhou S (2018) The distinctive microbial community improves composting efficiency in a full-scale hyperthermophilic composting plant. Bioresour Technol 265:146–154. https://doi.org/10.1016/j.biortech.2018.06.011
Liang J, Tang S, Gong J, Zeng G, Luo Y (2019) Responses of enzymatic activity and microbial communities to biochar/compost amendment in sulfamethoxazole polluted wetland soil. J Hazard Mater 385:121533. https://doi.org/10.1016/j.jhazmat.2019.121533
Bari Q, Koenig A, Guihe T (2010) Kinetic analysis of forced aeration composting- I. Reaction rates and temperature. Waste Manag Res 18(4):303–312. https://doi.org/10.1034/j.1399-3070.2000.00114.x
Liu X, Hou Y, Li Z, Yu Z, Zhou S (2020) Hyperthermophilic composting of sewage sludge accelerates humic acid formation: elemental and spectroscopic evidence. Waste Manag 103:342–351. https://doi.org/10.1016/j.wasman.2019.12.053
Cui P, Liao H, Bai Y, Li X, Zhou S (2019) Hyperthermophilic composting reduces nitrogen loss via inhibiting ammonifiers and enhancing nitrogenous humic substance formation. Sci Total Environ 692:98–106. https://doi.org/10.1016/j.scitotenv.2019.07.239
Liao H, Lu X, Rensing C, Friman VP, Geisen S, Chen Z, Yu Z, Wei Z, Zhou S, Zhu Y (2017) Hyperthermophilic composting accelerates the removal of antibiotic resistance genes and mobile genetic elements in sewage sludge. Environ Sci Technol 52(1):266–276. https://doi.org/10.1021/acs.est.7b04483
Oshima T, Moriya TA (2008) Preliminary analysis of microbial and biochemical properties of high-temperature compost. Ann N Y Acad Sci 1125(1):338–344. https://doi.org/10.1196/annals.1419.012
Cheng Y, Inamori R, Ruike K, Inamori Y, Zhang Z (2018) Optimum dosage of hyper-thermophilic aerobic compost (HTAC) produced from sewage sludge for rice yield. Int J Bio 10(3):27. https://doi.org/10.5539/ijbv10n3p27
Moriya T, Hikota T, Yumoto I, Ito T, Terui Y, Yamagishi A, Oshima T (2011) Calditerricola satsumensis gen. nov., sp. Nov. and Calditerricola yamamurae sp. Nov., extreme thermophiles isolated from a high-temperature compost. Int J Syst Evol Micr 61(3):631–636. https://doi.org/10.1099/ijs.0.018416-0
Wang Z, Wu D, Lin Y, Wang X (2021) Role of temperature in sludge composting and hyperthermophilic systems: a Review. Bioenerg Res. https://doi.org/10.1007/s12155-021-10281-5
Shiotsuka K, Tanaka A, Sakai K (2010) Occurrence of extreme thermophiles, thermaerobacter spp., in sewage sludge. J Jpn Soc Extrem 9(2):67–71. https://doi.org/10.3118/jjse.9.67
Stetter KO (2013) A brief history of the discovery of hyperthermophilic life. Biochem Soc Trans 41(1):416–420. https://doi.org/10.1042/BST20120284
Steven DH, Bo-Barker J, Jay M, Anja B, Ruth B, Barry AC, Heribert C, Gerald RD, Timothy F, Kai-Uwe H, Nils GH, Richard M, Arthur S, Guizhi W, Barbara B, Bert E, Kathryn F, Glen G, Scott DR, Henrik SC, Gregory S, Ivano WA, Gilles G, Christopher HH, Fumio I, Patrick M, Thomas N, Sachiko NR, John DCS, Andreas T, Juergen W, Christian NP, Acosta JLS (2004) Distributions of microbial activities in deep subseafloor sediments. Science 306:2216–2221. https://doi.org/10.1126/science.1101155
Atomi H, Sato T, Kanai T (2011) Application of hyperthermophiles and their enzymes. Curr Opin Biotechnol 22(5):618–626. https://doi.org/10.1016/j.copbio.2011.06.010
Vieille C, Zeikus GJ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65(1):1–43. https://doi.org/10.1128/MMBR.65.1.1-43.2001
Sakuraba H, Goda S, Ohshima T (2010) Unique sugar metabolism and novel enzymes of hyperthermophilic archaea. Chem Rec 3(5):281–287. https://doi.org/10.1002/tcr.10066
Pittera J, Jouhet J, Breton S, Garczarek L, Partensky F, Maréchal É, Nguyen NA, Doré H, Ratin M, Pitt FD (2017) Thermoacclimation and genome adaptation of the membrane lipidome in marine Synechococcus. Environ Microbiol 20(2):612–631. https://doi.org/10.1111/1462-2920.13985
Guyot S, Pottier L, Hartmann A, Ragon M, Hauck TJ, Molin P, Ferret E, Gervais P (2014) Extremely rapid acclimation of Escherichia coli to high temperature over a few generations of a fed-batch culture during slow warming. Microbiol Open 3(1):52–63. https://doi.org/10.1002/mbo3.146
Rudolph B, Gebendorfer KM, Buchner J, Winter J (2010) Evolution of Escherichia coli for growth at high temperatures. J Biol Chem 285(25):19029–19034. https://doi.org/10.1074/jbc.M110.103374
Khan S, Farooq U, Kurnikova M (2016) Exploring protein stability by comparative molecular dynamics simulation of homologous hyperthermophilic, mesophilic and psychrophilic proteins. J Chem Inf Model 56:2129–2139. https://doi.org/10.1021/acs.jcim.6b00305
Sterpone F, Melchionna S (2012) Thermophilic proteins: insight and perspective from in silico experiments. Chem Soc Rev 41(5):1665–1676. https://doi.org/10.1039/c1cs15199a
Beadle BM, Baase WA, Wilson DB, Gilkes NR, Shoichet BK (1999) Comparing the thermodynamic stabilities of a related thermophilic and mesophilic enzyme. Biochemistry 38(8):2570–2576. https://doi.org/10.1021/bi9824902
Grimalt-Alemany A, Łężyk M, Kennes-Veiga D, Gavala SI, H, (2020) Enrichment of mesophilic and thermophilic mixed microbial consortia for syngas biomethanation: the role of kinetic and thermodynamic competition. Waste Biomass Valori 2:465–481. https://doi.org/10.1007/s12649-019-00595-z
Gao X, Ding Y (2017) Using the residue interaction network improve the classification of thermophilic and mesophilic proteins. Curr Bioinform 12(3):249–257. https://doi.org/10.2174/1574893611666160502122132
Brinda KV (2005) A network representation of protein structures: implications for protein stability. Biophys J 6(89):4159–4170. https://doi.org/10.1529/biophysj.105.064485
Khadem H, Tirtouil A, Drabo S, Boubakeur B (2020) Ultrasound conditioning of Streptococcus thermophilus CNRZ 447: growth, biofilm formation, exopolysaccharide production, and cell membrane permeability. Biotechnologia 101:159–165. https://doi.org/10.5114/bta.2020.94774
Qiu S, Ma F, Wo Y, Xu S (2010) Study on the biological effect of tourmaline on the cell membrane of E. coli. Surf Interface Anal 43:1069–1073. https://doi.org/10.1002/sia.3694
Funnekotter B, Kaczmarczyk A, Turner SR, Bunn E, Zhou W, Smith S, Flematti G, Mancera RL (2013) Acclimation-induced changes in cell membrane composition and influence on cryotolerance of in vitro shoots of native plant species. Plant Cell Tiss Org 114(1):83–96. https://doi.org/10.1007/s11240-013-0308-0
Xu L, Tian C, Lu X, Ling L, Lv J, Wu M, Zhu G (2015) Photoreactivation of Escherichia coli is impaired at high growth temperatures. J Photoch Photobio B 285(25):19029. https://doi.org/10.1016/j.jphotobiol.2015.03.012
Ali ABA, Danial K, Asma A, Hoda B, Rosa F (2018) Identification of Thiobacillus bacteria in agricultural soil in Iran using the 16S rRNA gene. Mol Biol Rep 45(6):1723–1731. https://doi.org/10.1007/s11033-018-4316-3
Geng WW, Yang G, Xie XY, Yang B (2009) Determination of protein hydrophobicity by sodium dodecyl sulfate binding method. Food Sci 30(24):416–418. https://doi.org/10.1007/978-3-540-85168-4_52
Haralambiev L, Nitsch A, Jacoby M, Strakeljahn S, Stope B (2020) Cold atmospheric plasma treatment of chondrosarcoma cells affects proliferation and cell membrane permeability. Int J Mol Sci 21(7):2291. https://doi.org/10.3390/ijms21072291
Xiao Y, Zeng GM, Yang ZH, Shi WJ, Huang C, Fan CZ, Xu ZY (2009) Continuous thermophilic composting (CTC) for rapid biodegradation and maturation of organic municipal solid waste. Bioresour Technol 100(20):4807–4813. https://doi.org/10.1016/j.biortech.2009.05.013
Kulikowska D, Klimiuk E (2011) Organic matter transformations and kinetics during sewage sludge composting in a two-stage system. Bioresour Technol 102(23):10951–10958. https://doi.org/10.1016/j.biortech.2011.09.009
Gromiha MM, Pathak MC, Saraboji K, Ortlund EA, Gaucher EA (2013) Hydrophobic environment is a key factor for the stability of thermophilic proteins. Proteins 81(4):715–721. https://doi.org/10.1002/prot.24232
Pica A, Graziano G (2016) Shedding light on the extra thermal stability of thermophilic proteins. Biopolymers 105(12):856–863. https://doi.org/10.1002/bip.22923
Yuan H, Xu J, Van-Dam EP, Giubertoni G, Rezus YLA, Hammink R, Bakker HJ, Zhan Y, Rowan AE, Xing C (2017) Strategies to increase the thermal stability of truly biomimetic hydrogels: combining hydrophobicity and directed hydrogen bonding. Macromolecules 50(22):9058–9065. https://doi.org/10.1021/acs.macromol.7b01832
Hazel JR, Schuster VL (2010) The effects of temperature and thermal acclimation upon the osmotic properties and nonelectrolyte permeability of liver and gill mitochondria from rainbow trout (salmo gairdneri). J Exp Zool 195(3):425–438. https://doi.org/10.1002/jez.1401950309
Blicher A, Wodzinska K, Fidorra M, Winterhalter M, Heimburg T (2008) The temperature dependence of lipid membrane permeability, its quantized nature, and the influence of anesthetics. Biophys J 96(11):4581–4591. https://doi.org/10.1016/j.bpj.2009.01.062
Acknowledgements
The authors acknowledge that this work was supported by the National Natural Science Foundation of China (22078194) and National Key Research and Development Program (2017YFE0127100).
Funding
Funding provided by the National Natural Science Foundation of China (22078194) and National Key Research and Development Program (2017YFE0127100).
Author information
Authors and Affiliations
Contributions
ZW: conceptualization, investigation, methodology, experiments and writing (original draft preparation). SW: article revision and supervision. CF: methodology and article revision. XZ: data collection and writing (review and editing). DW: methodology and investigation. XW: formal analysis and investigation. HK: methodology, writing (review and editing), supervision and funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Z., Wu, S., Fan, C. et al. Isolation and thermo-acclimation of thermophilic bacteria in hyperthermophilic fermentation system. Bioprocess Biosyst Eng 45, 75–85 (2022). https://doi.org/10.1007/s00449-021-02640-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-021-02640-5