Abstract
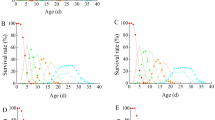
Predators and parasites have been important extrinsic factors influencing the fluctuation of small mammal populations. They can have non-additive effects on a shared group of preys or hosts, which can have important consequences for population dynamics. However, experimental studies incorporating the interactions between predation and parasites are scarce in small mammal populations. Here we systematically examined the synergistic effects of predators and coccidian parasites interaction on overwinter survival and likely mechanisms underlying the synergistic effects in the root vole (Microtus oeconomus). Our aim was to test the general hypothesis that predators and coccidia interact synergistically to decrease overwinter survival of root voles through mediating vole’s physiological traits and body conditions. We carried out a factorial experimental design, by which we manipulated the predator exclusion in combination with the parasitic removal in enclosures, and then measured fecal corticosterone metabolite (FCM) levels, immunocompetence, and body conditions in captured animals via repeated live trapping. We found a strong negative synergistic effect of predators and coccidia on survival. Importantly, we found that predators increased both the prevalence and intensity of coccidian infection in voles through immune suppression induced by predation stress, while increased coccidian infection reduced plasma protein and hematocrit level of voles, which may impair anti-predator ability of voles and lead to an increase in predation. Our finding showed when voles are exposed to both predation risk and infection, their synergistic effects greatly reduce overwinter survival and population density. This may be an important mechanism influencing population dynamics in small mammals.





Similar content being viewed by others
References
Alzaga V, Vicente J, Villanua D, Acevedo P, Casas F, Gortazar C (2008) Body condition and parasite intensity correlates with escape capacity in Iberian hares (Lepus granatensis). Behav Ecol Sociobiol 62:769–775. https://doi.org/10.1007/s00265-007-0502-3
Anderson DR, Burnham KP, White GC (1994) AIC model selection in overdispersed capture—recapture data. Ecology 75:1780–1793. https://doi.org/10.2307/1939637
Bartolomucci A (2007) Social stress, immune functions and disease in rodents. Front Neuroendocrin 28:28–49. https://doi.org/10.1016/j.yfrne.2007.02.001
Behringer DC, Butler MJ (2010) Disease avoidance influences shelter use and predation in Caribbean spiny lobster. Behav Ecol Sociobiol 64:747–755. https://doi.org/10.1007/s00265-009-0892-5
Berdoy M, Webster JP, Macdonald DW (2000) Fatal attraction in rats infected with Toxoplasma gondii. Proc R Soc B Biol Sci 267:1591–1594. https://doi.org/10.1098/rspb.2000.1182
Bian JH, Wu Y, Liu JK (2005) Breeding behavior under temporal risk of predation in male root voles (Microtus oeconomus). J Mammal 86:953–960. https://doi.org/10.1644/1545-1542(2005)86[953:BBUTRO]2.0.CO;2
Bian JH, Wu Y, Getz LL, Cao YF, Chen F, Yang L (2011) Does maternal stress influence winter survival of offspring in root voles Microtus oeconomus? A field experiment. Oikos 120:47–56. https://doi.org/10.1111/j.1600-0706.2010.18165.x
Bian JH, Du SY, Wu Y, Cao YF, Nie XH, He H, You ZB (2015) Maternal effects and population regulation: maternal density-induced reproduction suppression impairs offspring capacity in response to immediate environment in root voles Microtus oeconomus. J Anim Ecol 84:326–336. https://doi.org/10.1111/1365-2656.12307
Boonstra R, Krebs CJ, Beacham TD (1980) Impact of botfly parasitism on Microtus townsendii populations. Can J Zool 58:1683–1692. https://doi.org/10.1139/z80-230
Boonstra R, Hik D, Singleton GR, Tinnikov A (1998) The impact of predator-induced stress on the snowshoe hare cycle. Ecol Monogr 68:371–394. https://doi.org/10.1890/0012-9615(1998)068[0371:TIOPIS]2.0.CO;2
Cáceres CE, Knight CJ, Hall SR (2009) Predator–spreaders: predation can enhance parasite success in a planktonic host-parasite system. Ecology 90:2850–2858. https://doi.org/10.1890/08-2154.1
Caetano JV, Maia MR, Manica LT, Macedo RH (2014) Immune-related effects from predation risk in Neotropical blue-black grassquits (Volatinia jacarina). Behav Process 109:58–63. https://doi.org/10.1016/j.beproc.2014.07.003
Chambers LK, Singleton GR, Hinds LA (1999) Fertility control of wild mouse populations: the effects of hormonal competence and an imposed level of sterility. Wildlife Res 26:579–591. https://doi.org/10.1071/WR98093
Chen F, Du S, Bian J, You ZB, Wu Y (2012) Chronic hypoxia exposure during pregnancy is associated with a decreased active nursing activity in mother and an abnormal birth weight and postnatal growth in offspring of rats. Horm Behav 61:504–511. https://doi.org/10.1016/j.yhbeh.2012.01.009
Crompton DWT, Stephenson LS (1990) Hookworm infection, nutritional status and productivity. In: Schad GA, Warren KS (eds) Hookworm disease: current status and new directions. Taylor & Francis, London, pp 231–264
Daugschies A, Meyer C, Joachim A (1999) Occurrence of Isospora suis in piglet production farms. Prakt Tierarzt 80:530–535
Decaestecker E, De Meester L, Ebert D (2002) In deep trouble: habitat selection constrained by multiple enemies in zooplankton. Proc Natl Acad Sci USA 99:5481–5485. https://doi.org/10.1073/pnas.082543099
Desy EA, Batzli GO (1989) Effects of food availability and predation on prairie vole demography: a field experiment. Ecology 70:411–421. https://doi.org/10.1890/02-0534
Díaz M, Alonso CL (2003) Wood mouse Apodemus sylvaticus winter food supply: density, condition, breeding, and parasites. Ecology 84:2680–2691. https://doi.org/10.1890/02-0534
Du SY, Cao YF, Nie XH, Wu Y, Bian JH (2016) The synergistic effect of density stress during the maternal period and adulthood on immune traits of root vole (Microtus oeconomus) individuals—a field experiment. Oecologia 181:335–346. https://doi.org/10.1007/s00442-015-3445-9
Duffy MA (2009) Staying alive: the post-consumption fate of parasite spores and its implications for disease dynamics. Limnol Oceanogr 54:770–773. https://doi.org/10.4319/lo.2009.54.3.0770
Duffy MA, Housley JM, Penczykowski RM, Caceres CE, Hall SR (2011) Unhealthy herds: indirect effects of predators enhance two drivers of disease spread. Funct Ecol 25:945–953. https://doi.org/10.1111/j.1365-2435.2011.01872.x
Elton C (1942) Voles, mice and lemmings. Problems in population dynamics. Clarendon Press, Oxford
Elton C, Davis DHS, Findlay GM (1935) An epidemic among voles (Microtus agrestis) on the Scottish border in the spring of 1934. J Anim Ecol 4:277–288. https://doi.org/10.2307/1018
Gehman ALM, Byers JE (2017) Non-native parasite enhances susceptibility of host to native predators. Oecologia 183:919–926. https://doi.org/10.1007/s00442-016-3784-1
Goüy de Bellocq J, Krasnov BR, Khokhlova IS, Pinshow B (2006) Temporal dynamics of a T-cell mediated immune response in desert rodents. Comp Biochem Physiol A Mol Integr Physiol 145:554–559. https://doi.org/10.1016/j.cbpa.2006.08.045
Hall SR, Duffy MA, Cáceres CE (2004) Selective predation and productivity jointly drive complex behavior in host-parasite systems. Am Nat 165:70–81. https://doi.org/10.1086/426601
Hatcher MJ, Dick JT, Dunn AM (2006) How parasites affect interactions between competitors and predators. Ecol Lett 9:1253–1271. https://doi.org/10.1111/j.1461-0248.2006.00964.x
Hellgren EC, Rogers LL, Seal US (1993) Serum chemistry and hematology of black bears: physiological indices of habitat quality or seasonal patterns? J Mammal 74:304–315. https://doi.org/10.2307/1382385
Hudson PJ, Dobson AP, Newborn D (1992) Do parasites make prey vulnerable to predation? Red grouse and parasites. J Anim Ecol 61:681–692. https://doi.org/10.2307/5623
Jeanniard du Dot T, Rosen DAS, Trites AW (2009) Energy reallocation during and after periods of nutritional stress in steller sea lions: low-quality diet reduces capacity for physiological adjustments. Physiol Biochem Zool 82:516–530. https://doi.org/10.1086/603637
Jiang Y, Wei S, Wang Z, Zhen Y, Cui R (1991) Productivity investigation of the root vole (Microtus oeconomus) population in the Haibei alpine bushland (Potentilla fruticosa) I. Population dynamics. Acta Theriol Sin 11:270–278 (in Chinese, English abstract)
Jones GW, Parker RJ, Parke CR (1985) Coccidia associated with enteritis in grower pigs. Aust Vet J 62:319. https://doi.org/10.1111/j.1751-0813.1985.tb14917.x
Kamiya T, Poulin R (2012) Parasite-induced behavioural changes to the trade-off between foraging and predator evasion in a marine snail. J Exp Mar Biol Ecol 438:61–67. https://doi.org/10.1016/j.jembe.2012.09.002
Kavaliers M, Colwell DD (1995) Odours of parasitized males induce aversive responses in female mice. Anim Behav 50:1161–1169. https://doi.org/10.1016/0003-3472(95)80032-8
Klein SL (2005) Parasite manipulation of host behaviour: mechanisms, ecology, and future directions. Behav Process 3:219–221. https://doi.org/10.1016/j.beproc.2004.07.009
Krebs CJ, Boutin S, Boonstra R, Sinclair ARE, Smith JNM, Dale MRT, Martin K, Turkington R (1995) Impact of food and predation on the snowshoe hare cycle. Science 269:1112–1115. https://doi.org/10.1126/science.269.5227.1112
Krkošek M, Connors BM, Ford H, Peacock S, Mages P, Ford JS, Morton A, Volpe JP, Hilborn R, Dill LM, Lewis MA (2011) Fish farms, parasites, and predators: implications for salmon population dynamics. Ecol Appl 21:897–914. https://doi.org/10.1890/09-1861.1
Laakkonen J, Oksanen A, Soveri T, Henttonen H (1998) Dynamics of intestinal coccidia in peak density Microtus agrestis, Microtus oeconomus and Clethrionomus glareolus populations in Finland. Ecography 21:135–139. https://doi.org/10.1111/j.1600-0587.1998.tb00667.x
Lebreton JD, Burnham KP, Clobert J, Anderson DR (1992) Modeling survival and testing biological hypotheses using marked animals: a unified approach with case studies. Ecol Monogr 62:67–118. https://doi.org/10.2307/2937171
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Liu JK, Wang X, Liu W (1991) Studies on the nutritional ecology of small herbivorous mammals: patterns of food selection and resource utilization for root voles and Gansu pikas. In: Liu J, Wang Z (eds) Alpine meadow Ecosystem, Fasc.3. Science Press, Beijing, pp 111–124
Marino JA Jr, Werner EE (2013) Synergistic effects of predators and trematode parasites on larval green frog (Rana clamitans) survival. Ecology 94:2697–2708. https://doi.org/10.1890/13-0396.1
Møller AP, Nielsen JT (2007) Malaria and risk of predation: a comparative study of birds. Ecology 88:871–881. https://doi.org/10.1890/06-0747
Murray DL (2002) Differential body condition and vulnerability to predation in snowshoe hares. J Anim Ecol 71:614–625. https://doi.org/10.1046/j.1365-2656.2002.00632.x
Murray DL, Cary JR, Keith LB (1997) Interactive effects of sublethal nematodes and nutritional status on snowshoe hare vulnerability to predation. J Anim Ecol 66:250–264. https://doi.org/10.2307/6026
Navarro C, De Lope F, Marzal A, Møller AP (2004) Predation risk, host immune response, and parasitism. Behav Ecol 15:629–635. https://doi.org/10.1093/beheco/arh054
Newman C, Macdonald DW, Anwar MA (2001) Coccidiosis in the European badger, Meles meles in Wytham Woods: infection and consequences for growth and survival. Parasitology 123:133–142. https://doi.org/10.1017/s0031182001008265
Ondračková M, Valová Z, Kortan J, Vojtek L, Adámek Z (2012) Consequent effects of the great cormorant (Phalacrocorax carbo sinensis) predation on parasite infection and body condition of common carp (Cyprinus carpio). Parasitol Res 110:1487–1493. https://doi.org/10.1007/s00436-011-2652-5
Orlofske SA, Jadin RC, Hoverman JT, Johnson PT (2014) Predation and disease: understanding the effects of predators at several trophic levels on pathogen transmission. Freshwater Biol 59:1064–1075. https://doi.org/10.1111/fwb.12329
Packer C, Holt RD, Hudson PJ, Lafferty KD, Dobson AP (2003) Keeping the herds healthy and alert: implications of predator control for infectious disease. Ecol Lett 6:797–802. https://doi.org/10.1046/j.1461-0248.2003.00500.x
Palacios-Fuentes P, Landaeta MF, Muñoz G, Plaza G, Ojeda FP (2012) The effects of a parasitic copepod on the recent larval growth of a fish inhabiting rocky coasts. Parasitol Res 111:1661–1671. https://doi.org/10.1007/s00436-012-3005-8
Pihl L, Hau J (2003) Fecal corticosterone and immunoglobulin A in young adult rats. Lab Anim 37:166–171. https://doi.org/10.1258/00236770360563822
Ramirez RA, Snyder WE (2009) Scared sick? Predator-pathogen facilitation enhances exploitation of a shared resource. Ecology 90:2832–2839. https://doi.org/10.1890/08-1941.1
Relyea RA (2004) Fine-tuned phenotypes: tadpole plasticity under 16 combinations of predators and competitors. Ecology 85:172–179. https://doi.org/10.1890/03-0169
Rigby MC, Jokela J (2000) Predator avoidance and immune defence: costs and trade-offs in snails. Proc R Soc Lond B Biol 267:171–176. https://doi.org/10.1098/rspb.2000.0983
Sheriff MJ, Krebs CJ, Boonstra R (2009) The sensitive hare: sublethal effects of predator stress on reproduction in snowshoe hares. J Anim Ecol 78:1249–1258. https://doi.org/10.1111/j.1365-2656.2009.01552.x
Sheriff MJ, Krebs CJ, Boonstra R (2011) From process to pattern: how fluctuating predation risk impacts the stress axis of snowshoe hares during the 10-year cycle. Oecologia 166:593–605. https://doi.org/10.1007/s00442-011-1907-2
Solomon NG (1993) Body size and social preferences of male and female prairie voles, Microtus ochrogaster. Anim Behav 45:1031–1033. https://doi.org/10.1006/anbe.1993.1122
Steen H, Taitt M, Krebs CJ (2002) Risk of parasite-induced predation: an experimental field study on Townsend’s voles (Microtus townsendii). Can J Zool 80:1286–1292. https://doi.org/10.1139/z02-115
Stephenson JF, Van Oosterhout C, Mohammed RS, Cable J (2015) Parasites of Trinidadian guppies: evidence for sex-and age-specific trait-mediated indirect effects of predators. Ecology 96:489–498. https://doi.org/10.1890/14-0495.1
Sun RY, Zheng SW, Cui RX (1982) Home range of the root vole microtus oeconomus. Acta Theriol Sin 2:219–231 (in Chinese, English abstract)
Sundell J (2006) Experimental tests of the role of predation in the population dynamics of voles and lemmings. Mammal Rev 36:107–141. https://doi.org/10.1111/j.1365-2907.2006.00083.x
Tompkins DM, Dunn AM, Smith MJ, Telfer S (2011) Wildlife diseases: from individuals to ecosystems. J Anim Ecol 80:19–38. https://doi.org/10.1111/j.1365-2656.2010.01742.x
Werner EE, Peacor SD (2003) A review of trait-mediated indirect interactions in ecological communities. Ecology 84:1083–1100. https://doi.org/10.1890/0012-9658(2003)084[1083:AROTII]2.0.CO;2
Wetzel R (1951) Verbesserte Mcmaster-Kammer zum Auszählen von Wurmeiern. Tierärztl Umsch 6:209–210
White GC, Burnham KP (1999) Program MARK: survival estimation from populations of marked animals. Bird Study 46(Suppl.):S120–S139. https://doi.org/10.1080/00063659909477239
Xu DL, Wang DH (2010) Fasting suppresses T cell-mediated immunity in female Mongolian gerbils (Meriones unguiculatus). Comp Biochem Physiol A: Mol Integr Physiol 155:25–33. https://doi.org/10.1016/j.cbpa.2009.09.003
Xu DL, Liu X, Wang D (2011) Impairment of cellular and humoral immunity in overweight Mongolian gerbils (Meriones unguiculatus). Integr Zool 6:352–365. https://doi.org/10.1111/j.1749-4877.2011.00259.x
Yang YB, Shang GZ, Du SY, Zhang X, Wu Y, Bian JH (2018) Maternal density stress and coccidian parasitism: synergistic effects on overwinter survival in root voles. Funct Ecol 32:2181–2193. https://doi.org/10.1111/1365-2435.13129
Acknowledgements
This work was Funded by the National Key Research and Development Program of China (Grant/Award Number: 2016YFC0501901), Strategic Priority Research Program of Chinese Academy of Sciences (Grant/Award Number: XDA2005010406) and the National Natural Science Foundation of China (Grant No., 31570421). We thank Yan-Bin Yang for his assistance in the field work and biochemical assays.
Author information
Authors and Affiliations
Contributions
JHB and YW conceived and designed the experiments. GZS, YHZ, YFC performed the experiments. JHB and GZS analyzed the data. GZS wrote the manuscript. JHB and YW provided editorial advice.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights statement
The use of animals in this study was in accordance with the guidelines of the regulations of experiments on animals and was approved by the animal Ethics and Welfare committee of the Northwest Institute of Plateau Biology, Chinese Academy of Science.
Additional information
Communicated by Michael Sheriff.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shang, GZ., Zhu, YH., Wu, Y. et al. Synergistic effects of predation and parasites on the overwinter survival of root voles. Oecologia 191, 83–96 (2019). https://doi.org/10.1007/s00442-019-04455-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04455-4