Abstract
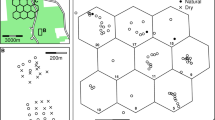
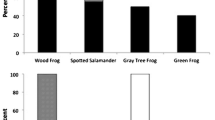
Epidemics in wildlife populations often display a striking seasonality. Ranaviruses can cause rapid, synchronous mass mortality events in populations of wood frog (Rana sylvatica) larvae in the summer. While there are several possible explanations for this pattern—from seasonal introductions of the virus to environmental stressors to windows of susceptibility to mortality from infection during development—most studies have focused on single factors in laboratory settings. We characterized the time course of ranavirus epidemics in eight ephemeral ponds in Connecticut, USA, measuring the prevalence and intensity of infections in wood frog larvae and Ranavirus DNA in water samples using environmental DNA methods. We found little evidence that the timing of pathogen introduction affected the timing of epidemics (rising prevalence) or the resulting die-offs. Instead, we observed a pulse in transmission asynchronous with die-offs; prevalence reached high levels (≥ 50%) up to 6 weeks before mortality was observed, suggesting that die-offs may be uncoupled from this pulse in transmission. Rather, mortality occurred when larvae reached later stages of development (hind limb formation) and coinciding water temperatures rose (≥ 15 °C), both of which independently increase pathogenicity (i.e., probability of host mortality) of infections in laboratory experiments. In summary, the strong seasonality of die-offs appears to be driven by development- and/or temperature-dependent changes in pathogenicity rather than occurring chronologically with pathogen introduction, after a pulse in transmission, or when susceptible host densities are greatest. Furthermore, our study illustrates the potential for eDNA methods to provide valuable insight in aquatic host–pathogen systems.



Similar content being viewed by others
References
Altizer S, Dobson A, Hosseini P, Hudson P, Pascual M, Rohani P (2006) Seasonality and the dynamics of infectious diseases. Ecol Lett 9:467–484. https://doi.org/10.1111/j.1461-0248.2005.00879.x
Altizer S, Ostfeld RS, Johnson PT, Kutz S, Harvell CD (2013) Climate change and infectious diseases: from evidence to a predictive framework. Science 341:514–519
APHA (1998) Standard methods for the examination of water and wastewaters. American Public Health Association, Washington, DC
Araujo A, Kirschman L, Warne RW (2016) Behavioural phenotypes predict disease susceptibility and infectiousness. Biol Lett. https://doi.org/10.1098/rsbl.2016.0480
Ariel E, Nicolajsen N, Christophersen M-B, Holopainen R, Tapiovaara H, Jensen BB (2009) Propagation and isolation of ranaviruses in cell culture. Aquaculture 294:159–164
Bass D, Stentiford GD, Littlewood D, Hartikainen H (2015) Diverse applications of environmental DNA methods in parasitology. Trends Parasitol 31:499–513
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Brand MD et al (2016) Water temperature affects susceptibility to Ranavirus. EcoHealth 13(2):1–10
Brunner JL, Schock DM, Collins JP (2007) Transmission dynamics of the amphibian ranavirus Ambystoma tigrinum virus. Dis Aquat Org 77:87–95. https://doi.org/10.3354/dao01845
Brunner JL, Storfer A, Gray MJ, Hoverman JT (2015) Ranavirus ecology and evolution: from epidemiology to extinction. Ranaviruses. Springer, New York, pp 71–104
Brunner JL, Beaty L, Guitard A, Russell D (2017) Heterogeneities in the infection process drive ranavirus transmission. Ecology 98:576–582. https://doi.org/10.1002/ecy.1644
Chinchar V (2002) Ranaviruses (family Iridoviridae): emerging cold-blooded killers. Adv Virol 147:447–470
Crespi EJ et al (2015) Geophysiology of wood frogs: landscape patterns of prevalence of disease and circulating hormone concentrations across the Eastern Range. Integr Comp Biol 55:602–617. https://doi.org/10.1093/icb/icv096
Denver RJ, Glennemeier KA, Boorse GC (2002) Endocrinology of complex life cycles: amphibians. Horm Brain Behav 2:469–513
Dhabhar FS (2009) Enhancing versus suppressive effects of stress on immune function: implications for immunoprotection and immunopathology. Neuroimmunomodulation 16:300–317
Dowell SF (2001) Seasonal variation in host susceptibility and cycles of certain infectious diseases. Emerg Infect Dis 7:369
Duffus A, Pauli B, Wozney K, Brunetti C, Berrill M (2008) Frog virus 3-like infections in aquatic amphibian communities. J Wildl Dis 44:109–120
Duffus AL et al (2015) Distribution and host range of ranaviruses. Ranaviruses. Springer, New York, pp 9–57
Echaubard P, Leduc J, Pauli B, Chinchar VG, Robert J, Lesbarreres D (2014) Environmental dependency of amphibian–ranavirus genotypic interactions: evolutionary perspectives on infectious diseases. Evol Appl 7:723–733
Gahl MK, Calhoun AJK (2008) Landscape setting and risk of Ranavirus mortality events. Biol Conserv 141:2679–2689. https://doi.org/10.1016/j.biocon.2008.08.003
Goldberg CS, Pilliod DS, Arkle RS, Waits LP (2011) Molecular detection of vertebrates in stream water: a demonstration using Rocky Mountain tailed frogs and Idaho giant salamanders. PLoS One 6:e22746. https://doi.org/10.1371/journal.pone.0022746
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190. https://doi.org/10.2307/3890061
Grassly NC, Fraser C (2006) Seasonal infectious disease epidemiology. Proc R Soc Lond B Biol Sci 273:2541–2550
Gray MJ, Brunner JL, Earl JE, Ariel E (2015) Design and analysis of ranavirus studies: surveillance and assessing risk. Ranaviruses. Springer, New York, pp 209–240
Green DE, Converse KA, Schrader AK (2002) Epizootiology of sixty-four amphibian morbidity and mortality events in the USA, 1996–2001. Ann N Y Acad Sci 969:323–339
Greer AL, Briggs CJ, Collins JP (2008) Testing a key assumption of host-pathogen theory: density and disease transmission. Oikos 117:1667–1673
Haislip NA, Gray MJ, Hoverman JT, Miller DL (2011) Development and disease: how susceptibility to an emerging pathogen changes through anuran development. PLoS One 6:e22307
Hall EM, Crespi EJ, Goldberg CS, Brunner JL (2016) Evaluating environmental DNA-based quantification of ranavirus infection in wood frog populations. Mol Ecol Resour 16:423–433
Hall EM, Brady SP, Mattheus NM, Earley RL, Diamond M, Crespi EJ (2017) Physiological consequences of exposure to salinized roadside ponds on wood frog larvae and adults. Biol Conserv 209:98–106. https://doi.org/10.1016/j.biocon.2017.02.013
Harp EM, Petranka JW (2006) Ranavirus in wood frogs (Rana sylvatica): potential sources of transmission within and between ponds. J Wildl Dis 42:307–318
Hayes T, Chan R, Licht P (1993) Interactions of temperature and steroids on larval growth, development, and metamorphosis in a toad (Bufo boreas). J Exp Zool Part A Ecol Genet Physiol 266:206–215
Hayhoe K et al (2008) Regional climate change projections for the Northeast USA. Mitig Adapt Strateg Glob Change 13:425–436
Hoverman JT, Gray MJ, Haislip NA, Miller DL (2011) Phylogeny, life history, and ecology contribute to differences in amphibian susceptibility to ranaviruses. EcoHealth 8:301–319
Johnson A, Brunner J (2014) Persistence of an amphibian ranavirus in aquatic communities. Dis Aquat Org 111:129–138
Johnson PT, Kellermanns E, Bowerman J (2011) Critical windows of disease risk: amphibian pathology driven by developmental changes in host resistance and tolerance. Funct Ecol 25:726–734
Kelly DW, Thomas H, Thieltges DW, Poulin R, Tompkins DM (2010) Trematode infection causes malformations and population effects in a declining New Zealand fish. J Anim Ecol 79:445–452
Kirschman LJ, Crespi EJ, Warne RW (2018) Critical disease windows shaped by stress exposure alter allocation trade-offs between development and immunity. J Anim Ecol 87:235–246
Kosmidis I (2013) brglm: Bias reduction in binomial-response generalized linear models. http://www.ucl.ac.uk/~ucakiko/software.html. Accessed 27 Aug 2018
Kuznetsova A, Brockhoff PB, Christensen RHB (2016) lmerTest: tests in linear mixed effects models. R package version 2.0–30. https://CRAN.R-project.org/package=lmerTest. Accessed 27 Aug 2018
Lafferty KD, Holt RD (2003) How should environmental stress affect the population dynamics of disease? Ecol Lett 6:654–664
Langwig KE et al (2015) Host and pathogen ecology drive the seasonal dynamics of a fungal disease, white-nose syndrome. Philos Trans R Soc Lond Ser B Biol Sci 282:20142335
Moore MK, Townsend VR Jr (1998) The interaction of temperature, dissolved oxygen and predation pressure in an aquatic predator-prey system. Oikos 81(2):329–336
Munro J, Bayley AE, McPherson NJ, Feist SW (2016) Survival of frog virus 3 in freshwater and sediment from an English lake. J Wildl Dis 52:138–142
Nazir J, Spengler M, Marschang R (2012) Environmental persistence of amphibian and reptilian ranaviruses. Dis Aquat Org 98:177–184
Nelson RJ, Demas GE, Klein SL, Kriegsfeld LJ (2002) Seasonal patterns of stress, immune function, and disease. Cambridge University Press, Cambridge
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org. Accessed 27 Aug 2018
Robert J, George E, Andino FDJ, Chen G (2011) Waterborne infectivity of the Ranavirus frog virus 3 in Xenopus laevis. Virology 417:410–417
Rollins-Smith LA (1998) Metamorphosis and the amphibian immune system. Immunol Rev 166:221–230
Stilwell NK, Whittington RJ, Hick PM, Becker JA, Ariel E, Van Beurden S, Vendramin N, Olesen NJ, Waltzek TB (2018) Partial validation of a TaqMan real-time quantitative PCR for the detection of ranaviruses. Dis Aquat Org 128(2):105–116
Todd-Thompson M (2010) Seasonality, variation in species prevalence, and localized disease for Ranavirus in Cades Cove (Great Smoky Mountains National Park) amphibians. Master's Thesis, University of Tennessee. https://trace.tennessee.edu/utk_gradthes/665
Warne RW, Crespi EJ, Brunner JL (2011) Escape from the pond: stress and developmental responses to ranavirus infection in wood frog tadpoles. Funct Ecol 25:139–146. https://doi.org/10.1111/j.1365-2435.2010.01793.x
Wells K et al (2015) Timing and severity of immunizing diseases in rabbits is controlled by seasonal matching of host and pathogen dynamics. J R Soc Interface 12:20141184
Werner EE, Skelly DK, Relyea RA, Yurewicz KL (2007) Amphibian species richness across environmental gradients. Oikos 116:1697–1712
White K, Grenfell B, Hendry R, Lejeune O, Murray J (1996) Effect of seasonal host reproduction on host-macroparasite dynamics. Math Biosci 137:79–99
Acknowledgements
This publication was developed under STAR Fellowship Assistance Agreement no. 91767901 awarded by the U.S. Environmental Protection Agency (EPA) to EMH. It has not been formally reviewed by EPA. The views expressed in this publication are solely those of E. M. Hall and co-authors, and EPA does not endorse any products or commercial services mentioned in this publication. This work was also funded by the Washington State University Elling Foundation. We would like to graciously thank A. Helton lab from University of Connecticut (Storrs, CT) for processing biogeochemical water samples. We are grateful for constructive feedback from P. Johnson and anonymous reviewers.
Author information
Authors and Affiliations
Contributions
EMH, CSG, JLB, and EJC designed the experiment. EMH performed field work and processed samples. EMH and JLB analyzed data, and everyone contributed to writing the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed. This research was approved by the Animal Care and Use Committee (Protocol #04520-001) of Washington State University. Collections were approved by the Connecticut Department of Energy and Environmental Protection (Scientific collections permit #1115003) and Yale Myers Forest.
Conflict of interest
The authors declare that they have no conflict of interest.
Data accessibility
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Additional information
Communicated by Pieter Johnson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hall, E.M., Goldberg, C.S., Brunner, J.L. et al. Seasonal dynamics and potential drivers of ranavirus epidemics in wood frog populations. Oecologia 188, 1253–1262 (2018). https://doi.org/10.1007/s00442-018-4274-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4274-4