Abstract
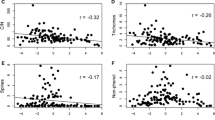
Trade-offs among plant functional traits indicate diversity in plant strategies of growth and survival. The leaf economics spectrum (LES) reflects a trade-off between short-term carbon gain and long-term leaf persistence. A related trade-off, between foliar growth and anti-herbivore defense, occurs among plants growing in contrasting resource regimes, but it is unclear whether this trade-off is maintained within plant communities, where resource gradients are minimized. The LES and the growth-defense trade-off involve related traits, but the extent to which these trade-off dimensions are correlated is poorly understood. We assessed the relationship between leaf economic and anti-herbivore defense traits among sunlit foliage of 345 canopy trees in 83 species on Barro Colorado Island, Panama. We quantified ten traits related to resource allocation and defense, and identified patterns of trait co-variation using multivariate ordination. We tested whether traits and ordination axes were correlated with patterns of phylogenetic relatedness, juvenile demographic trade-offs, or topo-edaphic variation. Two independent axes described ~ 60% of the variation among canopy trees. Axis 1 revealed a trade-off between leaf nutritional and structural investment, consistent with the LES. Physical defense traits were largely oriented along this axis. Axis 2 revealed a trade-off between investments in phenolic defenses versus other foliar defenses, which we term the leaf defense spectrum. Phylogenetic relationships and topo-edaphic variation largely did not explain trait co-variation. Our results suggest that some trade-offs among the growth and defense traits of outer-canopy trees may be captured by the LES, while others may occur along additional resource allocation dimensions.



Similar content being viewed by others
References
Ackerly D (2004) Functional strategies of chaparral shrubs in relation to seasonal water deficit and disturbance. Ecol Monogr 74:25–44. https://doi.org/10.1890/03-4022
Adler PB, Salguero-Gómez R, Compagnoni A et al (2014) Functional traits explain variation in plant life history strategies. Proc Natl Acad Sci USA 111:740–745. https://doi.org/10.1073/pnas.1315179111
Agrawal AA (2007) Macroevolution of plant defense strategies. Trends Ecol Evol 22:103–109. https://doi.org/10.1016/j.tree.2006.10.012
Agrawal AA, Fishbein M (2006) Plant defense syndromes. Ecology 87:S132–S149. https://doi.org/10.1890/0012-9658(2006)87[132:PDS]2.0.CO;2
Agrawal AA, Konno K (2009) Latex: a model for understanding mechanisms, ecology, and evolution of plant defense against herbivory. Annu Rev Ecol Evol Syst 40:311–331. https://doi.org/10.1146/annurev.ecolsys.110308.120307
Ainsworth EA, Gillespie KM (2007) Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin–Ciocalteu reagent. Nat Protoc 2:875–877. https://doi.org/10.1038/nprot.2007.102
Appel HM (1993) Phenolics in ecological interactions: the importance of oxidation. J Chem Ecol 19:1521–1552. https://doi.org/10.1007/BF00984895
Asner GP, Martin RE (2011) Canopy phylogenetic, chemical and spectral assembly in a lowland Amazonian forest. New Phytol 189:999–1012. https://doi.org/10.1111/j.1469-8137.2010.03549.x
Asner GP, Martin RE, Tupayachi R et al (2011) Taxonomy and remote sensing of leaf mass per area (LMA) in humid tropical forests. Ecol Appl 21:85–98. https://doi.org/10.1890/09-1999.1
Asner GP, Martin RE, Tupayachi R et al (2014) Amazonian functional diversity from forest canopy chemical assembly. Proc Natl Acad Sci 111:5604–5609. https://doi.org/10.1073/pnas.1401181111
Ayres MP, Clausen TP, MacLean SF et al (1997) Diversity of structure and antiherbivore activity in condensed tannins. Ecology 78:1696–1712. https://doi.org/10.1890/0012-9658(1997)078[1696:DOSAAA]2.0.CO;2
Baillie I, Elsenbeer H, Barthold F et al (2007) Semi-detailed soil survey of Barro Colorado Island. Smithsonian Tropical Research Institute. Panama City, Panama
Baltzer JL, Thomas SC (2010) A second dimension to the leaf economics spectrum predicts edaphic habitat association in a tropical forest. PLoS One 5:e13163. https://doi.org/10.1371/journal.pone.0013163
Barbehenn RV, Constabel CP (2011) Tannins in plant-herbivore interactions. Phytochemistry 72:1551–1565. https://doi.org/10.1016/j.phytochem.2011.01.040
Basset Y, Aberlenc HP, Delvare G (1992) Abundance and stratification of foliage arthropods in a lowland rain forest of Cameroon. Ecol Entomol 17:310–318. https://doi.org/10.1111/j.1365-2311.1992.tb01063.x/full
Bazzaz FA, Chiariello NR, Coley PD, Pitelka LF (1987) Allocating resources to reproduction and defense. Bioscience 37:58–67
Beven KJ, Kirkby MJ (1979) A physically based, variable contributing area model of basin hydrology. Hydrol Sci Bull 24:43–69. https://doi.org/10.1080/02626667909491834
Bi JL, Felton GW (1995) Foliar oxidative stress and insect herbivory: primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance. J Chem Ecol 21:1511–1530. https://doi.org/10.1007/BF02035149
Blomberg SP, Garland T, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution (NY) 57:717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x
Bloom AJ, Chapin FS, Mooney HA (1985) Resource limitation in plants—an economic analogy. Annu Rev Ecol Syst 16:363–392
Boege K, Marquis RJ (2005) Facing herbivory as you grow up: the ontogeny of resistance in plants. Trends Ecol Evol 20:441–448. https://doi.org/10.1016/j.tree.2005.05.001
Chen Y, Hagerman AE (2005) Reaction pH and protein affect the oxidation products of β-pentagalloyl glucose. Free Radic Res 39:117–124
Clark DA, Clark DB (1992) Life history diversity of canopy and emergent trees in a neotropical rain forest. Ecol Monogr 62:315. https://doi.org/10.2307/2937114
Clark DB, Clark DA (1996) Abundance, growth and mortality of very large trees in neotropical lowland rain forest. For Ecol Manag 80:235–244. https://doi.org/10.1016/0378-1127(95)03607-5
Clark DB, Palmer MW, Clark DA (1999) Edaphic factors and the landscape-scale distribution of tropical rain forest trees. Ecology 80:2662–2675. https://doi.org/10.1890/0012-9658(1999)080[2662:EFATLS]2.0.CO;2
Coley PD (1988) Effects of plant growth rate and leaf lifetime on the amount and type of anti-herbivore defense. Oecologia 74:531–536. https://doi.org/10.1007/BF00380050
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Condit R (2000) Spatial patterns in the distribution of tropical tree species. Science 288:1414–1418. https://doi.org/10.1126/science.288.5470.1414
Condit R, Hubbell SP, Foster RB (1996) Assessing the response of plant functional types to climatic change in tropical forests. J Veg Sci 7:405–416. https://doi.org/10.2307/3236284
Condit R, Pérez R, Daguerre N (2011) Trees of Panama and Costa Rica. Princeton University Press, Princeton
Crisp MD, Cook LG (2012) Phylogenetic niche conservatism: what are the underlying evolutionary and ecological causes? New Phytol 196:681–694. https://doi.org/10.1111/j.1469-8137.2012.04298.x
Croat TB (1978) Flora of Barro Colorado Island. Stanford University Press, Stanford
Dalling JW, Winter K, Nason JD et al (2001) The unusual life history of Alseis blackiana: a shade-persistent pioneer tree? Ecology 82:933–945. https://doi.org/10.1890/0012-9658(2001)082[0933:TULHOA]2.0.CO;2
Diaz-Uriarte R, Garland T (1999) Polytomies and phylogenetically independent contrasts: examination of the bounded degrees of freedom approach. Syst Biol 48:547–558. https://doi.org/10.1080/106351599260139
Dominy NJ, Lucas PW, Wright SJ (2003) Mechanics and chemistry of rain forest leaves: canopy and understorey compared. J Exp Bot 54:2007–2014
Endara M-J, Coley PD (2011) The resource availability hypothesis revisited: a meta-analysis. Funct Ecol 25:389–398. https://doi.org/10.1111/j.1365-2435.2010.01803.x
Everette JD, Bryant QM, Green AM et al (2010) Thorough study of reactivity of various compound classes toward the Folin–Ciocalteu reagent. J Agric Food Chem 58:8139–8144. https://doi.org/10.1021/jf1005935
Feeny P (1976) Plant apparency and chemical defense. Recent Adv Phytochem 10:1–40
Feldman KS, Sahasrabudhe K, Quideau S et al (1999) Prospects and progress in ellagitannin synthesis. In: Gross GG, Hemingway RW, Yoshida T, Branham SJ (eds) Plant polyphenols 2. Springer, New York, pp 101–125
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution (NY) 39:783–791
Fine PVA, Miller ZJ, Mesones I et al (2006) The growth-defense trade-off and habitat specialization by plants in Amazonian forests. Ecology 87:S150–S162. https://doi.org/10.1890/0012-9658(2006)87[150:TGTAHS]2.0.CO;2
Fritz SA, Purvis A (2010) Selectivity in mammalian extinction risk and threat types: a new measure of phylogenetic signal strength in binary traits. Conserv Biol 24:1042–1051. https://doi.org/10.1111/j.1523-1739.2010.01455.x
Garland T, Harvey PH, Ives AR (1992) Procedures for the analysis of comparative data using phylogenetically independent contrasts. Syst Biol 41:18–32. https://doi.org/10.1093/sysbio/41.1.18
Givnish TJ (1988) Adaptation to sun and shade: a whole-plant perspective. Aust J Plant Physiol 15:63. https://doi.org/10.1071/PP9880063
Gleadow R, Bjanrholt N, Jorgensen K et al (2011) Detection, identification and quantitative measurement of cyanogenic glycosides. In: Narwal SS, Szajdak L, Sampietro DA (eds) Soil allelochemicals. Stadium Press, Houston, pp 283–310
Gower JC (1966) A Q-technique for the calculation of canonical variates. Biometrika 53:588–590
Grubb PJ (1977) The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol Rev 52:107–145. https://doi.org/10.1111/j.1469-185X.1977.tb01347.x
Grundhöfer P, Niemetz R, Schilling G, Gross GG (2001) Biosynthesis and subcellular distribution of hydrolyzable tannins. Phytochemistry 57:915–927. https://doi.org/10.1016/S0031-9422(01)00099-1
Hagerman AE (1992) Tannin-protein interactions. In: Ho C, Lee CY, Huang M (eds) Phenolic compounds in food and their effects on health I. American Chemical Society, Washington, DC, pp 236–247
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335
Holdridge LR (1947) Determination of world plant formations from simple climate data. Science 105:367–368
Huang D, Ou B, Prior RL (2005) The chemistry behind antioxidant capacity assays. J Agric Food Chem 53:1841–1856. https://doi.org/10.1021/jf030723c
Iglewicz B, Hoaglin DC (1993) How to detect and handle outliers. ASQC Quality Press, Milwaukee
Kembel SW, Cowan PD, Helmus MR et al (2010) Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464
King G, Zeng L (2001) Logistic regression in rare events data. Polit Anal 9:137–163. https://doi.org/10.1093/oxfordjournals.pan.a004868
Kinner D, Mixon D, Stallard R, Wahl S (2002) BCI GIS version 1.3
Kitajima K, Poorter L (2008) Functional basis for resource niche partitioning by tropical trees. In: Carson WP, Schnitzer SA (eds) Tropical forest community ecology. Blackwell Publishing, Oxford, pp 160–181
Kitajima K, Poorter L (2010) Tissue-level leaf toughness, but not lamina thickness, predicts sapling leaf lifespan and shade tolerance of tropical tree species. New Phytol 186:708–721. https://doi.org/10.1111/j.1469-8137.2010.03212.x
Kitajima K, Mulkey SS, Wright SJ (2005) Variation in crown light utilization characteristics among tropical canopy trees. Ann Bot 95:535–547. https://doi.org/10.1093/aob/mci051
Kitajima K, Llorens A-M, Stefanescu C et al (2012) How cellulose-based leaf toughness and lamina density contribute to long leaf lifespans of shade-tolerant species. New Phytol 195:640–652. https://doi.org/10.1111/j.1469-8137.2012.04203.x
Koenker R (2005) Quantile regression. Cambridge University Press, Cambridge, UK
Konno K (2011) Plant latex and other exudates as plant defense systems: roles of various defense chemicals and proteins contained therein. Phytochemistry 72:1510–1530. https://doi.org/10.1016/j.phytochem.2011.02.016
Koricheva J (2002) Meta-analysis of sources of variation in fitness costs of plant antiherbivore defenses. Ecology 83:176–190. https://doi.org/10.1890/0012-9658(2002)083[0176:MAOSOV]2.0.CO;2
Kress WJ, Erickson DL, Jones FA et al (2009) Plant DNA barcodes and a community phylogeny of a tropical forest dynamics plot in Panama. Proc Natl Acad Sci USA 106:18621–18626. https://doi.org/10.1073/pnas.0909820106
Kursar T, Coley P (2003) Convergence in defense syndromes of young leaves in tropical rainforests. Biochem Syst Ecol 31:929–949. https://doi.org/10.1016/S0305-1978(03)00087-5
Lambert JL, Ramasamy J, Paukstelis JV (1975) Stable reagents for the colorimetric determination of cyanide by modified Koenig reactions. Anal Chem 47:916–918. https://doi.org/10.1021/ac60356a036
Lechtenberg M, Nahrstedt A (1999) Cyanogenic glycosides. In: Ikan R (ed) Naturally occurring glycosides. Wiley, Chichester, pp 147–191
Legendre P, Legendre L (1998) Numerical ecology. Elsevier Science, Amsterdam
Leigh EG (1999) Tropical forest ecology: a view from Barro Colorado Island. Oxford University Press, Oxford
Losos JB (2008) Phylogenetic niche conservatism, phylogenetic signal and the relationship between phylogenetic relatedness and ecological similarity among species. Ecol Lett 11:995–1003. https://doi.org/10.1111/j.1461-0248.2008.01229.x
Lucas P (2000) Mechanical defences to herbivory. Ann Bot 86:913–920. https://doi.org/10.1006/anbo.2000.1261
Makkar HPS, Siddhuraju P, Becker K (2007) Plant secondary metabolites
Mason CM, Donovan LA (2015) Does investment in leaf defenses drive changes in leaf economic strategy? A focus on whole-plant ontogeny. Oecologia 177:1053–1066. https://doi.org/10.1007/s00442-014-3177-2
Mazza CA, Boccalandro HE, Giordano CV et al (2000) Functional significance and induction by solar radiation of ultraviolet-absorbing sunscreens in field-grown soybean crops. Plant Physiol 122:117–126. https://doi.org/10.1104/PP.122.1.117
Messmer T, Elsenbeer H, Wilcke W (2014) High exchangeable calcium concentrations in soils on Barro Colorado Island, Panama. Geoderma 217–218:212–224. https://doi.org/10.1016/j.geoderma.2013.10.021
Mooney HA, Gulmon SL, Johnson ND (1983) Physiological constraints on plant chemical defenses. In: Hedin P (ed) Plant resistance to insects. American Chemical Society, Washington, DC., pp 21–36
Onoda Y, Westoby M, Adler PB et al (2011) Global patterns of leaf mechanical properties. Ecol Lett 14:301–312. https://doi.org/10.1111/j.1461-0248.2010.01582.x
Orme D, Freckleton R, Thomas G et al (2013) Caper: comparative analyses of phylogenetics and evolution in R. R Packag Vers 0.5.2 5:1
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20:289–290
Peeters PJ, Sanson G, Read J (2007) Leaf biomechanical properties and the densities of herbivorous insect guilds. Funct Ecol 21:246–255. https://doi.org/10.1111/j.1365-2435.2006.01223.x
Polly PD, Lawing AM, Fabre A-C, Goswami A (2013) Phylogenetic Principal Components Analysis and Geometric Morphometrics. Hystrix. Ital. J. Mammal. 24:33–41
Poorter L (2007) Are species adapted to their regeneration niche, adult niche, or both? Am Nat 169:433–442. https://doi.org/10.1086/512045
Poorter L, Bongers F (2006) Leaf traits are good predictors of plant performance across 53 rain forest species. Ecology 87:1733–1743. https://doi.org/10.1890/0012-9658(2006)87[1733:LTAGPO]2.0.CO;2
Poorter H, Niinemets Ü, Poorter L et al (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588. https://doi.org/10.1111/j.1469-8137.2009.02830.x
R Core Team (2015) R: A language and environment for statistical computing
Quideau S, Feldman KS, Appel HM (1995) Chemistry of gallotannin-derived o-quinones: reactivity toward nucleophiles. J Org Chem 60:4982–4983. https://doi.org/10.1021/jo00121a012
Read J, Sanson GD, Caldwell E et al (2009) Correlations between leaf toughness and phenolics among species in contrasting environments of Australia and New Caledonia. Ann Bot 103:757–767. https://doi.org/10.1093/aob/mcn246
Reich PB, Wright IJ, Cavender-Bares J et al (2003) The evolution of plant functional variation: traits, spectra, and strategies. Int J Plant Sci 164:S143–S164. https://doi.org/10.1086/374368
Revell LJ (2009) Size-correction and principal components for interspecific comparative studies. Evolution 63:3258–3268. https://doi.org/10.1111/j.1558-5646.2009.00804.x
Revell LJ (2012) Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223
Salminen J-P, Karonen M (2011) Chemical ecology of tannins and other phenolics: we need a change in approach. Funct Ecol 25:325–338. https://doi.org/10.1111/j.1365-2435.2010.01826.x
Schultz JC (1988) Plant responses induced by herbivores. Trends Ecol Evol 3:45–49. https://doi.org/10.1016/0169-5347(88)90047-X
Shipley B, Lechowicz MJ, Wright I, Reich PB (2006) Fundamental trade-offs generating the worldwide leaf economics spectrum. Ecology 87:535–541. https://doi.org/10.1890/05-1051
Silvertown J (2004) Plant coexistence and the niche. Trends Ecol Evol 19:605–611. https://doi.org/10.1016/j.tree.2004.09.003
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Steidinger B (2015) Qualitative differences in tree species distributions along soil chemical gradients give clues to the mechanisms of specialization: why boron may be the most important soil nutrient at Barro Colorado Island. New Phytol. https://doi.org/10.1111/nph.13298
Tilman D (2004) Niche tradeoffs, neutrality, and community structure: a stochastic theory of resource competition, invasion, and community assembly. Proc Natl Acad Sci USA 101:10854–10861
Toth GB, Pavia H (2001) Removal of dissolved brown algal phlorotannins using insoluble polyvinylpolypyrrolidone (PVPP). J Chem Ecol 27:1899–1910. https://doi.org/10.1023/A:1010421128190
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Vitousek PM (1984) Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 65:285–298. https://doi.org/10.2307/1939481
Vitousek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17:137–167. https://doi.org/10.1146/annurev.es.17.110186.001033
Westbrook JW, Kitajima K, Burleigh JG et al (2011) What makes a leaf tough? Patterns of correlated evolution between leaf toughness traits and demographic rates among 197 shade-tolerant woody species in a neotropical forest. Am Nat 177:800–811. https://doi.org/10.1086/659963
Westoby M, Wright IJ (2006) Land-plant ecology on the basis of functional traits. Trends Ecol Evol 21:261–268. https://doi.org/10.1016/j.tree.2006.02.004
Westoby M, Falster DS, Moles AT et al (2002) Plant ecological strategies: some leading dimensions of variation between species. Annu Rev Ecol Syst 33:125–159. https://doi.org/10.1146/annurev.ecolsys.33.010802.150452
Wink M (2003) Evolution of secondary metabolites from an ecological and molecular phylogenetic perspective. Phytochemistry 64:3–19. https://doi.org/10.1016/S0031-9422(03)00300-5
Wink M (2010) Introduction: Biochemistry, physiology, and ecological functions of secondary metabolites. In: Wink M (ed) Annual plant reviews, volume 40: biochemistry of plant secondary metabolism, 2nd edn. Blackwell Publishing, Oxford, pp 1–19
Wright IJ, Reich PB, Westoby M et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Wright SJ, Kitajima K, Kraft NJB et al (2010) Functional traits and the growth–mortality trade-off in tropical trees. Ecology 91:3664–3674. https://doi.org/10.1890/09-2335.1
Yavitt JB (2000) Nutrient dynamics of soil derived from different parent material on Barro Colorado Island, Panama. Biotropica 32:198–207
Yavitt JB, Wieder RK (1988) Nitrogen, phosphorus, and sulfur properties of some forest soils on Barro Colorado Island, Panama. Biotropica 20:2–10
Acknowledgements
We thank K. Kryston, B. Tsang, F. Sinca, R. Tupayachi, N. Jaramillo, J.A. Vega, R. Mena-Werth, and C. Mong for their indispensable help with logistics, field work, and laboratory analyses. The Carnegie Spectranomics Project (http://spectranomics.ciw.edu) is supported by the John D. and Catherine T. MacArthur Foundation.
Author information
Authors and Affiliations
Contributions
KMC, GPA, and CBF conceived and designed this study. GPA, REM, and KMC developed methodology. GPA and SJW collaborated on sampling design. KMC, REM, and GPA conducted fieldwork and chemical analyses. WJK generated the molecular phylogeny. KMC wrote the manuscript; other authors provided editorial advice.
Corresponding author
Ethics declarations
Human/animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Jennifer Thaler.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chauvin, K.M., Asner, G.P., Martin, R.E. et al. Decoupled dimensions of leaf economic and anti-herbivore defense strategies in a tropical canopy tree community. Oecologia 186, 765–782 (2018). https://doi.org/10.1007/s00442-017-4043-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-017-4043-9