Abstract
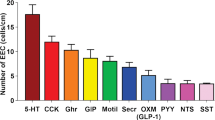
This paper provides quantitative data on the distributions of enteroendocrine cells (EEC), defined by the hormones they contain, patterns of colocalisation between hormones and EEC relations to nerve fibres in the rat gastric mucosa. The rat stomach has three mucosal types: non-glandular stratified squamous epithelium of the fundus and esophageal groove, a region of oxyntic glands in the corpus, and pyloric glands of the antrum and pylorus. Ghrelin and histamine were both contained in closed cells, not contacting the lumen, and were most numerous in the corpus. Gastrin cells were confined to the antrum, and 5-hydroxytryptamine (5-HT) and somatostatin cells were more frequent in the antrum than the corpus. Most somatostatin cells had basal processes that in the antrum commonly contacted gastrin cells. Peptide YY (PYY) cells were rare and mainly in the antrum. The only numerous colocalisations were 5-HT and histamine, PYY and gastrin and gastrin and histamine in the antrum, but each of these populations was small. Peptide-containing nerve fibres were found in the mucosa. One of the most common types was vasoactive intestinal peptide (VIP) fibres. High-resolution analysis showed that ghrelin cells were closely and selectively approached by VIP fibres. In contrast, gastrin cells were not selectively innervated by VIP or CGRP fibres. The study indicates that there are distinct populations of gastric EEC and selective innervation of ghrelin cells. It also shows that, in contrast to EEC of the small intestine, the majority of EEC within the stomach contained only a single hormone.










Similar content being viewed by others
References
Accili EA, Dhatt N, Buchan AMJ (1995) Neural somatostatin, vasoactive intestinal polypeptide and substance P in canine and human jejunum. Neurosci Lett 185:37–40
Andrews PLR, Davis CJ, Bingham S, Davidson HI, Hawthorn J, Maskell L (1990) The abdominal visceral innervation and the emetic reflex: pathways, pharmacology, and plasticity. Can J Physiol Pharmacol 68:325–345
Andrews PLR, Naylor RJ, Joss RA (1998) Neuropharmacology of emesis and its relevance to anti-emetic therapy. Support Care Cancer 6:197–203
Asakawa A, Inui A, Kaga T, Yuzuriha H, Nagata T, Ueno N, Makino S, Fujimiya M, Niijima A, Fujino MA, Kasuga M (2001) Ghrelin is an appetite-stimulatory signal from stomach with structural resemblance to motilin. Gastroenterology 120:337–345
Avau B, Carbone F, Tack J, Depoortere I (2013) Ghrelin signaling in the gut, its physiological properties, and therapeutic potential. Neurogastroenterol Motil 25:720–732
Berthoud H-R (1996) Morphological analysis of vagal input to gastrin releasing peptide and vasoactive intestinal peptide containing neurons in the rat glandular stomach. J Comp Neurol 370:61–70
Berthoud H-R, Powley TL (1992) Vagal afferent innervation of the rat fundic stomach: morphological characterization of the gastric tension receptor. J Comp Neurol 319:261–276
Blundell JE (1986) Serotonin manipulations and the structure of feeding behaviour. Appetite 7:39–56
Buchan AMJ, Sikora LKJ, Levy JG, McIntosh CHS, Dyck I, Brown JC (1985) An immunocytochemical investigation with monoclonal antibodies to somatostatin. Histochemistry 83:175–180
Canfield SP, Spencer JE (1983) The inhibitory effects of 5-hydroxytryptamine on gastric acid secretion by the rat isolated stomach. Br J Pharmacol 78:123–129
Cho H-J, Callaghan B, Bron R, Bravo DM, Furness JB (2014) Identification of enteroendocrine cells that express TRPA1 channels in the mouse intestine. Cell Tissue Res 356:77–82
Cho H-J, Kosari S, Hunne B, Callaghan B, Rivera LR, Bravo DM, Furness JB (2015) Differences in hormone localisation patterns of K and L type enteroendocrine cells in the mouse and pig small intestine and colon. Cell Tissue Res 359:693–698
Choi E, Roland JT, Barlow BJ, O’Neal R, Rich AE, Nam KT, Shi C, Goldenring JR (2014) Cell lineage distribution atlas of the human stomach reveals heterogeneous gland populations in the gastric antrum. Gut 63:1711–1720
Chuang C-N, Tanner M, Lloyd KCK, Wong H, Soll AH (1993) Endogenous somatostatin inhibits histamine release from canine gastric mucosal cells in primary culture. Am J Physiol Gastrointest Liver Physiol 28:G521–G525
Clemmensen C, MT D, Woods SC, Berthoud H-R, Seeley RJ, Tschöp MH (2017) Gut-brain cross-talk in metabolic control. Cell 168:758–774
Cooper SJ, Dourish CT (1990) Multiple cholecystokinin (CCK) receptors and CCK-monoamine interactions are instrumental in the control of feeding. Physiol Behav 48:849–857
Costa M, Furness JB (1984) Somatostatin is present in a subpopulation of noradrenergic nerve fibres supplying the intestine. Neuroscience 13:911–919
Date Y, Kojima M, Hosoda H, Sawaguchi A, Mondal MS, Suganuma T, Matsukura S, Kangawa K, Nakazato M (2000) Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 141:4255–4261
Date Y, Murakami N, Toshinai K, Matsukura S, Niijima A, Matsuo H, Kangawa K, Nakazato M (2002) The role of the gastric afferent vagal nerve in ghrelin-induced feeding and growth hormone secretion in rats. Gastroenterology 123:1120–1128
Dornonville De La Cour C, Björkqvist M, Sandvik AK, Bakke I, Zhao C-M, Chen D, Håkanson R (2001) A-like cells in the rat stomach contain ghrelin and do not operate under gastrin control. Regul Pept 99:141–150
Dornonville de la Cour C, Lindström E, Norlén P, Håkanson R (2004) Ghrelin stimulates gastric emptying but is without effect on acid secretion and gastric endocrine cells. Regul Pept 120:23–32
Egerod KL, Engelstoft MS, Grunddal KV et al (2012) A major lineage of enteroendocrine cells coexpress CCK, secretin, GIP, GLP-1, PYY, and neurotensin but not somatostatin. Endocrinology 153:5782–5795
Ekblad E, Ekelund M, Graffner H, Håkanson R, Sundler F (1985) Peptide-containing nerve fibres in the stomach wall of rat and mouse. Gastroenterology 89:73–85
Ekblad E, M Q, Sundler F (2000) Innervation of the gastric mucosa. Microsc Res Tech 48:241–257
Engelstoft MS, Park W-M, Sakata I et al (2013) Seven transmembrane G protein-coupled receptor repertoire of gastric ghrelin cells. Mol Metab 2:376–392
Fakhry J, Stebbing MJ, Hunne B, Bayguinov Y, Ward SM, Sasse KC, Callaghan B, McQuade RM, Furness JB (2019) Relationships of endocrine cells to each other and to other cell types in the human gastric fundus and corpus. Cell Tissue Res 376:37–49
Fothergill LJ, Callaghan B, Hunne B, Bravo DM, Furness JB (2017) Costorage of enteroendocrine hormones evaluated at the cell and subcellular levels in male mice. Endocrinology 158:2113–2123
Frick C, Rettenberger AT, Lunz ML, Breer H (2016) Complex morphology of gastrin-releasing G-cells in the antral region of the mouse stomach. Cell Tissue Res 366:301–310
Furness JB, Costa M, Walsh JH (1981) Evidence for and significance of the projection of VIP neurons from the myenteric plexus to the taenia coli in the guinea-pig. Gastroenterology 80:1557–1561
Furness JB, Padbury RTA, Baimbridge KG, Skinner JM, Lawson DEM (1989) Calbindin immunoreactivity is a characteristic of enterochromaffin- like cells ECL cells of the human stomach. Histochemistry 92:449–451
Furness JB, Koopmans HS, Robbins HL, Clerc N, Tobin JM, Morris MJ (2001) Effects of vagal and splanchnic section on food intake, weight, serum leptin and hypothalamic neuropeptide Y in rat. Auton Neurosci 92:28–36
Furness JB, Rivera LR, Cho H-J, Bravo DM, Callaghan B (2013) The gut as a sensory organ. Nat Rev Gastroenterol Hepatol 10:729–740
Gong Z, Yoshimura M, Aizawa S, Kurotani R, Zigman JM, Sakai T, Sakata I (2013) G-protein coupled receptor 120 (GPR120) signaling regulates ghrelin secretion in vivo and in vitro. Am J Physiol Endocrinol Metab 306:E28–E35
Gribble FM, Reimann F (2016) Enteroendocrine cells: chemosensors in the intestinal epithelium. Annu Rev Physiol 78:277–299
Habib AM, Richards P, Cairns LS, Rogers GJ, Bannon CAM, Parker HE, Morley TCE, Yeo GSH, Reimann F, Gribble FM (2012) Overlap of endocrine hormone expression in the mouse intestine revealed by transcriptional profiling and flow cytometry. Endocrinology 153:3054–3065
Hauso Ø, Gustafsson BI, Waldum HL (2007) Long slender cytoplasmic extensions: a common feature of neuroendocrine cells? J Neuroendocrinol 19:739–742
Hunyady B, Zólyomi A, Hoffman BJ, Mezey E (1998) Gastrin-producing endocrine cells: a novel source of histamine in the rat stomach. Endocrinology 139:4404–4415
Husted AS, Trauelsen M, Rudenko O, Hjorth SA, Schwartz TW (2017) GPCR-mediated signaling of metabolites. Cell Metab 25:777–796
Ito H, Yokozaki H, Tokumo K, Nakajo S, Tahara E (1986) Serotonin-containing EC cells in normal human gastric mucosa and in gastritis. Virchows Archiv A 409:313–323
Kasacka I, Łebkowski W, Janiuk I, Łapińska J, Lewandowska A (2012) Immunohistochemical identification and localisation of gastrin and somatostatin in endocrine cells of human pyloric gastric mucosa. Folia Morphol (Warsz) 71:39–44
Kestell GR, Anderson RL, Clarke JN, Haberberger RV, Gibbins IL (2015) Primary afferent neurons containing calcitonin gene-related peptide but not substance P in forepaw skin, dorsal root ganglia, and spinal cord of mice. J Comp Neurol 523:2555–2569
Kojima M, Kangawa K (2010) Ghrelin: more than endogenous growth hormone secretagogue. Ann N Y Acad Sci 1200:140–148
Kovacs TO, Lloyd KC, Lawson DC (1997) Inhibition of sham feeding-stimulated acid secretion in dogs by immunoneutralization of gastrin. Am J Physiol Gastrointest Liver Physiol 273:G399–G403
Larsson LI, Goltermann N, De Magistris L, Rehfeld JF, Schwarz TW (1979) Somatostatin cell processess as pathways for paracrine secretion. Science 205:1393–1395
le Roux CW, Neary NM, Halsey TJ, Small CJ, Martinez-Isla AM, Ghatei MA, Theodorou NA, Bloom SR (2005) Ghrelin does not stimulate food intake in patients with surgical procedures involving vagotomy. J Clin Endocrinol Metab 90:4521–4524
LePard KJ, Chi J, Mohammed JR, Gidener S, Stephens RL Jr (1996) Gastric antisecretory effect of serotonin: quantitation of release and site of action. Am J Physiol Endocrinol Metab 271:E669–E677
Levin F, Edholm T, Schmidt PT, Grybäck P, Jacobsson H, Degerblad M, Höybye C, Holst JJ, Rehfeld JF, Hellström PM, Näslund E (2006) Ghrelin stimulates gastric emptying and hunger in normal-weight humans. J Clin Endocrinol Metab 91:3296–3302
Lu K-H, Cao J, Thomas Oleson S, Powley TL, Liu Z (2017) Contrast-enhanced magnetic resonance imaging of gastric emptying and motility in rats. IEEE Trans Biomed Eng 64:2546–2554
Miller AS, Furness JB, Costa M (1989) The relationship between gastrin cells and bombesin-like immunoreactive nerve fibres in the gastric antral mucosa of guinea-pig, rat, dog and man. Cell Tissue Res 257:171–178
Mizutani M, Atsuchi K, Asakawa A, Matsuda N, Fujimura M, Inui A, Kato I, Fujimiya M (2009) Localization of acyl ghrelin- and des-acyl ghrelin-immunoreactive cells in the rat stomach and their responses to intragastric pH. Am J Physiol Gastrointest Liver Physiol 297:G974–G980
Moody TW, Pert CB (1979) Bombesin-like peptides in rat brains: quantitation and biochemical characterization. Biochem Biophys Res Commun 90:7–14
Mumphrey MB, Patterson LM, Zheng H, Berthoud H-R (2013) Roux-en-Y gastric bypass surgery increases number but not density of CCK-, GLP-1-, 5-HT-, and neurotensin-expressing enteroendocrine cells in rats. Neurogastroenterol Motil 25:e70–e79
Phillips RJ, Powley TL (1998) Gastric volume detection after selective vagotomies in rats. Am J Physiol Regul Integr Comp Physiol 271:R766–R769
Powley TL, Spaulding RA, Haglof SA (2011) Vagal afferent innervation of the proximal gastrointestinal tract mucosa: chemoreceptor and mechanoreceptor architecture. J Comp Neurol 519:644–660
Powley TL, Hudson CN, McAdams JL, Baronowsky EA, Martin FN, Mason JK, Phillips RJ (2014) Organization of vagal afferents in pylorus: mechanoreceptors arrayed for high sensitivity and fine spatial resolution? Auton Neurosci 183:36–48
Pustovit RV, Callaghan B, Ringuet MT, Kerr NF, Hunne B, Smyth IM, Pietra C, Furness JB (2017) Evidence that central pathways that mediate defecation utilize ghrelin receptors but do not require endogenous ghrelin. Physiol Rep 5:e13385
Reynaud Y, Fakhry J, Fothergill L, Callaghan B, Ringuet MT, Hunne B, Bravo DM, Furness JB (2016) The chemical coding of 5-hydroxytryptamine containing enteroendocrine cells in the mouse gastrointestinal tract. Cell Tissue Res 364:489–497
Rindi G, Necchi V, Savio A, Torsello A, Zoli M, Locatelli V, Raimondo F, Cocchi D, Solcia E (2002) Characterisation of gastric ghrelin cells in man and other mammals: studies in adult and fetal tissues. Histochem Cell Biol 117:511–519
Sanger GJ, Furness JB (2016) Ghrelin and motilin receptors as drug targets for gastrointestinal disorders. Nat Rev Gastroenterol Hepatol 19:38–48
Schubert ML, Peura DA (2008) Control of gastric acid secretion in health and disease. Gastroenterology 134:1842–1860
Schubert ML, Bitar KN, Makhlouf GM (1982) Regulation of gastrin and somatostatin secretion by cholinergic and noncholinergic intramural neurons. Am J Physiol Gastrointest Liver Physiol 243:G442–G447
Schubert ML, Edwards NF, Makhlouf GM (1988) Regulation of gastric somatostatin secretion in the mouse by luminal acidity: a local feedback mechanism. Gastroenterology 94:317–322
Smolka AJ, Larsen KA, Hammond CE (2000) Location of a cytoplasmic epitope for monoclonal antibody HK 12.18 on H,K-ATPase α subunit. Biochem Biophys Res Commun 273:942–947
Solcia E, Rindi G, Buffa R, Fiocca R, Capella C (2000) Gastric endocrine cells: types, function and growth. Regul Pept 93:31–35
Steensels S, Vancleef L, Depoortere I (2016) The sweetener-sensing mechanisms of the ghrelin cell. Nutrients 8:795
Stengel A, Hofmann T, Goebel-Stengel M, Lembke V, Ahnis A, Elbelt U, Lambrecht NWG, Ordemann J, Klapp BF, Kobelt P (2013) Ghrelin and NUCB2/nesfatin-1 are expressed in the same gastric cell and differentially correlated with body mass index in obese subjects. Histochem Cell Biol 139:909–918
Sykaras AG, Demenis C, Cheng L, Pisitkun T, Mclaughlin JT, Fenton RA, Smith CP (2014) Duodenal CCK cells from male mice express multiple hormones including ghrelin. Endocrinology 155:3339–3351
Tanaka-Shintani M, Watanabe M (2005) Distribution of ghrelin-immunoreactive cells in human gastric mucosa: comparison with that of parietal cells. J Gastroenterol 40:345–349
Veedfald S, Plamboeck A, Hartmann B, Vilsbøll T, Knop FK, Deacon CF, Svendsen LB, Holst JJ (2018) Ghrelin secretion in humans - a role for the vagus nerve? Neurogastroenterol Motil 30:e13295
Vuyyuru L, Harrington L, Arimura A, Schubert ML (1997) Reciprocal inhibitory paracrine pathways link histamine and somatostatin secretion in the fundus of the stomach. Am J Physiol Gastrointest Liver Physiol 273:G106–G111
Walker AK, Park W-M, Chuang J-C, Perello M, Sakata I, Osborne-Lawrence S, Zigman JM (2013) Characterization of gastric and neuronal histaminergic populations using a transgenic mouse model. PLoS One 8:e60276
Weihe E, Schütz B, Hartschuh W, Anlauf M, Schäfer MK, Eiden LE (2005) Coexpression of cholinergic and noradrenergic phenotypes in human and nonhuman autonomic nervous system. J Comp Neurol 492:370–379
Williams DL, Cummings DE, Grill HJ, Kaplan JM (2003) Meal-related ghrelin suppression requires postgastric feedback. Endocrinology 144:2765–2767
Wo JM, Nowak TV, Waseem S, Ward MP (2016) Gastric electrical stimulation for gastroparesis and chronic unexplained nausea and vomiting. Curr Treat Options Gastroenterol 14:386–400
Worthington JJ, Reimann F, Gribble FM (2018) Enteroendocrine cells-sensory sentinels of the intestinal environment and orchestrators of mucosal immunity. Mucosal Immunol 11:3–20
Yakabi K, Ro S, Onouhi T, Tanaka T, Ohno S, Miura S, Johno Y, Takayama K (2006) Histamine mediates the stimulatory action of ghrelin on acid secretion in rat stomach. Dig Dis Sci 51:1313–1321
Yu P-L, Fujimura M, Hayashi N, Nakamura T, Fujimiya M (2001) Mechanisms in regulating the release of serotonin from the perfused rat stomach. Am J Physiol Gastrointest Liver Physiol 280:G1099–G1105
Zheng H, Berthoud H-R (2000) Functional vagal input to gastric myenteric plexus as assessed by vagal stimulation-induced Fos expression. Am J Physiol Gastrointest Liver Physiol 279:G73–G81
Acknowledgements
We thank Josiane Fakhry for helpful comments on the manuscript.
Funding
This work was supported by NIH (SPARC) grant ID # OT2OD023847 (PI Terry Powley) to JBF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Informed consent
None required.
Ethical approval
Procedures were approved by the University of Melbourne Animal Ethics Committee (ethics approval number 1614002). All applicable National and Institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hunne, B., Stebbing, M.J., McQuade, R.M. et al. Distributions and relationships of chemically defined enteroendocrine cells in the rat gastric mucosa. Cell Tissue Res 378, 33–48 (2019). https://doi.org/10.1007/s00441-019-03029-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-019-03029-3