Abstract
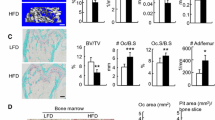
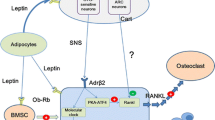
The accumulation of fat cells (adipocytes) in bone marrow is now thought to be a factor contributing to age-related bone loss. Women with osteoporosis have higher numbers of marrow adipocytes than women with healthy bone, and bone formation rate is inversely correlated with adipocyte number in bone tissue biopsies from both men and women. Adipogenic differentiation of bone marrow stromal cells increases with age, but the factors regulating populations of mature adipocytes are not well understood. Leptin is thought to regulate adipose tissue mass via its receptors in the ventromedial hypothalamus (VMH). We have therefore tested the hypothesis that stimulation of leptin receptors in the VMH regulates adipocyte number in bone marrow. Results indicate that unilateral twice-daily injections of leptin into the rat VMH for only 4 or 5 days cause a significant reduction in the number of adipocytes in peripheral fat pads and bone marrow and indeed eliminate adipocytes almost entirely from bone marrow of the proximal tibia. Osteoblast surface is not affected with leptin treatment. Apoptosis assays performed on bone marrow samples from control and treated rats have revealed a significant increase in protein concentration of the apoptosis marker caspase-3 with leptin treatment. We conclude that stimulation of leptin receptors in the VMH significantly decreases the adipocyte population in bone marrow, primarily through apoptosis of marrow adipocytes. Elimination of marrow adipocytes via this central pathway may represent a useful strategy for the treatment and prevention of osteoporosis.








Similar content being viewed by others
References
Abbott C, Kennedy A, Wren A, Rossi M, Murphy K, Seal L, Todd J, Ghatei M, Small C, Bloom S (2003) Identification of hypothalamic nuclei involved in the orexigenic effect of melanin-concentrating hormone. Endocrinology 144:3943–3949
Ahdjoudj S, Lasmoles F, Holy X, Zerath E, Marie P (2002) Transforming growth factor beta-2 inhibits adipocyte differentiation induced by skeletal unloading in rat bone marrow stroma. J Bone Miner Res 17:668–677
Bathija A, Davis S, Trubowitz S (1979) Bone marrow adipose tissue: response to acute starvation. Am J Hematol 6:191–198
Ceddia RB, William WN Jr, Lima FB, Carpinelli AR, Curi R (1998) Pivotal role of leptin in insulin effects. Braz J Med Biol Res 31:715–722
Cousin B, Agou K, Leturque A, Ferre P, Girard J, Penicaud L (1992) Molecular and metabolic changes in white adipose tissue of the rat during development of ventromedial hypothalamic obesity. Eur J Biochem 207:377–382
Custer R, Ahlfeldt F (1932) Studies on the structure and function of bone marrow. II. Variations in the cellularity in various bones with advancing years of life and their relative response to stimuli. J Lab Clin Med 17:960–962
Della-Fera MA, Qian H, Baile CA (2001) Adipocyte apoptosis in the regulation of body fat mass by leptin. Diabetes Obes Metab 3:299–310
Ducy P, Amling M, Takeda S, Premel M, Schilling A, Beil F, Shen J, Vinson C, Rueger J, Karsenty G (2000) Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell 100:197–207
Fried S, Bunkin D, Greenberg A (1998) Omental and subcutaneous adipose tissues of obese subjects release interleukin-6: depot difference and regulation by glucocorticoid. J Clin Endocrinol Metab 83:847–850
Guidobono F, Pagani F, Sibilia V, Netti C, Lattuada N, Rapetti D, Mrak E, Villa I, Cavani F, Bertoni L, Palumbo C, Ferretti M, Marotti G, Rubinacci G (2006) Different skeletal regional response to continuous brain infusion of leptin in the rat. Peptides 27:1426–1433
Gullicksen P, Hausman D, Dean R, Hartzell D, Baile C (2003) Adipose tissue cellularity and apoptosis after intracerebroventricular injections of leptin and 21 days recovery in rats. Int J Obesity Relat Metab Disord 27:302–312
Gullicksen P, Dean R, Baile C (2004) Detection of DNA fragmentation and apoptotic proteins and quantification of uncoupling protein expression by real-time RT-PCR in adipose tissue. J Biochem Biophys Methods 58:1–13
Hamrick MW, Pennington C, Newton D, Xie D, Isales C (2004) Leptin deficiency produces contrasting phenotypes in bones of the limb and spine. Bone 34:376–383
Hamrick M, Della-Fera MA, Choi Y-H, Pennington C, Hartzell D, Baile CA (2005) Leptin treatment induces loss of bone marrow adipocytes & increases bone formation in leptin-deficient ob/ob mice. J Bone Mineral Res 20:994–1001
Jacob RJ, Dziura J, Medwick MB et al (1997) The effect of leptin is enhanced by microinjection into the ventromedial hypothalamus. Diabetes 46:150–152
Jilka R (2002) Osteoblast progenitor cell fate and age-related bone loss. J Musculoskel Neuron Interact 2:581–583
Justesen J, Stenderup K, Ebbesen E, Mosekilde L, Steiniche T, Kassem M (2001) Adipocyte tissue volume in bone marrow is increased with aging and in patients with osteoporosis. Biogerontology 2:165–171
Kajkenova O, Lecka-Czernik F, Gubrij I, Hauser S, Takahashi K, Parfitt A, Jilka R, Manolagas S, Lipschitz D (1997) Increased adipogenesis and myelopoiesis in the bone marrow of SAMP6, a murine model of defective osteoblastogenesis and low turnover osteopenia. J Bone Miner Res 12:1772–1779
Kalra PS, Dube MG, Xu B, Farmerie WG, Kalra SP (1998) Neuropeptide Y (NPY) Y1 receptor mRNA is upregulated in association with transient hyperphagia and body weight gain: evidence for a hypothalamic site for concurrent development of leptin resistance. J Neuroendocrinol 10:43–49
Kurabayashi T, Tomita M, Matsushita H, Honda A, Takakuwa K, Tanaka K (2001) Effects of a β3 adrenergic receptor agonist on bone and bone marrow adipocytes in the tibia and lumbar spine of the ovariectomized rat. Calcif Tissue Int 68:248–254
Laroche M (2002) Intraosseous circulation from physiology to disease. Joint Bone Spine 69:262–269
Mach D, Rogers S, Sabino M, Luger N, Schwei M, Pomonis M, Keyser C, Clohisy D, Adams D, O’Leary P, Mantyh P (2002) Origins of skeletal pain: sensory and sympathetic innervation of the mouse femur. Neuroscience 113:155–166
Maurin A, Chavassieux P, Frappart L, Delmas P, Serre C, Meunier P (2000) Influence of mature adipocytes on osteoblast proliferation in human primary cocultures. Bone 26:485–489
Meunier P, Aaron J, Edouard C, Vignon G (1971) Osteoporosis and the replacement of cell populations of the marrow by adipose tissue. Clin Orthop Rel Res 80:147–154
Minaire P, Meunier P, Edouard C, Berbard J, Courpron J, Bourret J (1974) Quantitative histological data on disuse osteoporosis. Calcif Tissue Res 13:371–382
Minokoshi Y, Haque MS, Shimazu T (1999) Microinjection of leptin into the ventromedial hypothalamus increases glucose uptake in peripheral tissues in rats. Diabetes 48:287–291
Moerman E, Teng K, Lipschitz D, Lecka-Czernik B (2004) Aging activates adipogenic and suppresses osteogenic programs in mesenchymal marrow stromal/stem cells: the role of PPAR-gamma2 transcription factor and TGF-beta/BMP signaling pathways. Aging Cell 3:379–389
Nuttall M, Gimble J (2000) Is there a therapeutic opportunity to either prevent or treat osteopenic disorders by inhibiting marrow adipogenesis? Bone 27:177–184
Orban Z, Remaley AT, Sampson M, Trajanoski Z, Chrousos GP (1999) The differential effect of food intake and beta-adrenergic stimulation on adipose-derived hormones and cytokines in man. J Clin Endocrinol Metab 84:2126–2133
Page K, Hartzell D, Li C, Westby A, Della-Fera MA, Azain M, Pringle T, Baile CA (2004) Beta-adrenergic receptor agonists increase apoptosis of adipose tissue in mice. Dome Animal Endocrinol 26:23–31
Parfitt AM, Drezner M, Glorieux F, Kanis J, Malluche H, Meunier P, Ott S, Recker R (1987) Bone histomorphometry: standardization of nomenclature, symbols, units. J Bone Miner Res 2:595–610
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Elsevier, New York
Pierroz D, Muzzin P, Glatt V, Bouxsein M, Rizzoli R, Ferrari S (2004) β1β2-Adrenergic receptor KO mice have decreased total body and cortical bone mass despite increased trabecular bone number. J Bone Miner Res 19 (Suppl 1):S1121
Pierroz D, Bouxsein M, Muzzin P, Rizzoli R, Ferrari S (2005) Bone loss following ovariectomy is maintained in absence of adrenergic receptor β1 and β2 signaling. J Bone Miner Res 20 (Suppl 1):S277
Qian H, Azain M, Compton M, Hartzell D, Hausman G, Baile C (1998) Brain administration of leptin causes deletion of adipocytes by apoptosis. Endocrinology 139:791–794
Satoh N, Ogawa Y, Katsuura G et al (1997) Pathophysiological significance of the obese gene product, leptin, in ventromedial hypothalamus (VMH)-lesioned rats: evidence for loss of its satiety effect in VMH-lesioned rats. Endocrinology 138:947–954
Takeda S, Eleftriou F, Levasseur R, Liu X, Zhao L, Parker K, Armstrong D, Ducy P, Karsenty G (2002) Leptin regulates bone formation via the sympathetic nervous system. Cell 111:305–317
Verma S, Rajaratnman J, Dneton J, Hoyland J, Byers R (2002) Adipocytic proportion of bone marrow is inversely related to bone formation in osteoporosis. J Clin Pathol 55:693–698
Wronski T, Morey-Holton E, Jee W (1981) Skeletal alterations in rats during spaceflight. Adv Space Res 1:135–140
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamrick, M.W., Della Fera, M.A., Choi, YH. et al. Injections of leptin into rat ventromedial hypothalamus increase adipocyte apoptosis in peripheral fat and in bone marrow. Cell Tissue Res 327, 133–141 (2007). https://doi.org/10.1007/s00441-006-0312-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-006-0312-3