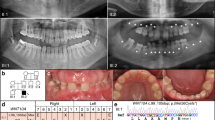
Abstract
Tooth agenesis (TA), the failure of development of one or more permanent teeth, is a common craniofacial abnormality observed in different world populations. The genetic etiology of TA is heterogeneous; more than a dozen genes have been associated with isolated or nonsyndromic TA, and more than 80 genes with syndromic forms. In this study, we applied whole exome sequencing (WES) to identify candidate genes contributing to TA in four Turkish families. Likely pathogenic variants with a low allele frequency in the general population were identified in four disease-associated genes, including two distinct variants in TSPEAR, associated with syndromic and isolated TA in one family each; a variant in LAMB3 associated with syndromic TA in one family; and a variant in BCOR plus a disease-associated WNT10A variant in one family with syndromic TA. With the notable exception of WNT10A (Tooth agenesis, selective, 4, MIM #150400), the genotype-phenotype relationships described in the present cohort represent an expansion of the clinical spectrum associated with these genes: TSPEAR (Deafness, autosomal recessive 98, MIM #614861), LAMB3 (Amelogenesis imperfecta, type IA, MIM #104530; Epidermolysis bullosa, junctional, MIMs #226700 and #226650), and BCOR (Microphthalmia, syndromic 2, MIM #300166). We provide evidence supporting the candidacy of these genes with TA, and propose TSPEAR as a novel nonsyndromic TA gene. Our data also suggest potential multilocus genomic variation, or mutational burden, in a single family, involving the BCOR and WNT10A loci, underscoring the complexity of the genotype–phenotype relationship in the common complex trait of TA.




Similar content being viewed by others
References
1000 Genomes Project Consortium et al (2015) A global reference for human genetic variation. Nature 526:68–74. https://doi.org/10.1038/nature15393
Adaimy L et al (2007) Mutation in WNT10A is associated with an autosomal recessive ectodermal dysplasia: the odonto-onycho-dermal dysplasia. Am J Hum Genet 81:821–828. https://doi.org/10.1086/520064
Arte S, Parmanen S, Pirinen S, Alaluusua S, Nieminen P (2013) Candidate gene analysis of tooth agenesis identifies novel mutations in six genes and suggests significant role for WNT and EDA signaling and allele combinations. PLoS One 8:e73705. https://doi.org/10.1371/journal.pone.0073705
Bainbridge MN et al (2011) Targeted enrichment beyond the consensus coding DNA sequence exome reveals exons with higher variant densities. Genome Biol 12:R68. https://doi.org/10.1186/gb-2011-12-7-r68
Bayram Y et al (2017) REST final-exon-truncating mutations cause hereditary gingival fibromatosis. Am J Hum Genet 101:149–156. https://doi.org/10.1016/j.ajhg.2017.06.006
Ben-Shachar S, Khajavi M, Withers MA, Shaw CA, van Bokhoven H, Brunner HG, Lupski JR (2009) Dominant versus recessive traits conveyed by allelic mutations—to what extent is nonsense-mediated decay involved? Clin Genet 75:394–400. https://doi.org/10.1111/j.1399-0004.2008.01114.x
Bergendal B, Klar J, Stecksen-Blicks C, Norderyd J, Dahl N (2011) Isolated oligodontia associated with mutations in EDARADD. Am J Med Genet A 155A:1616–1622. https://doi.org/10.1002/ajmg.a.34045
Bircher AJ, Lang-Muritano M, Pfaltz M, Bruckner-Tuderman L (1993) Epidermolysis bullosa junctionalis progressiva in three siblings. Br J Dermatol 128:429–435. https://doi.org/10.1111/j.1365-2133.1993.tb00204.x
Bohring A et al (2009) WNT10A mutations are a frequent cause of a broad spectrum of ectodermal dysplasias with sex-biased manifestation pattern in heterozygotes. Am J Hum Genet 85:97–105. https://doi.org/10.1016/j.ajhg.2009.06.001
Burgeson RE et al (1994) A new nomenclature for the laminins. Matrix Biol 14:209–211
Cai J et al (2010) Function analysis of mesenchymal Bcor in tooth development by using RNA interference. Cell Tissue Res 341:251–258. https://doi.org/10.1007/s00441-010-0996-2
Cai X, Gong P, Huang Y, Lin Y (2011) Notch signalling pathway in tooth development and adult dental cells. Cell Prolif 44:495–507. https://doi.org/10.1111/j.1365-2184.2011.00780.x
Celikoglu M, Kazanci F, Miloglu O, Oztek O, Kamak H, Ceylan I (2010) Frequency and characteristics of tooth agenesis among an orthodontic patient population. Med Oral Patol Oral Cir Bucal 15:e797–e801. https://doi.org/10.4317/medoral.15.e797
Chen YT, Liu HC, Han D, Liu Y, Feng HL (2017) Association between EDAR Polymorphisms and Non-Syndromic Tooth Agenesis in the Chinese Han Population. Chin J Dent Res 20:153–159. https://doi.org/10.3290/j.cjdr.a38770
Coban-Akdemir Z et al (2018) Identifying genes whose mutant transcripts cause dominant disease traits by potential gain-of-function alleles. Am J Hum Genet. In press
Creighton TE (1988) Disulphide bonds and protein stability. Bioessays 8:57–63. https://doi.org/10.1002/bies.950080204
Delmaghani S, Aghaie A, Michalski N, Bonnet C, Weil D, Petit C (2012) Defect in the gene encoding the EAR/EPTP domain-containing protein TSPEAR causes DFNB98 profound deafness. Hum Mol Genet 21:3835–3844. https://doi.org/10.1093/hmg/dds212
Dinckan N et al (2018) Whole-exome sequencing identifies novel variants for tooth agenesis. J Dent Res 97:49–59. https://doi.org/10.1177/0022034517724149
Doerks T, Copley RR, Schultz J, Ponting CP, Bork P (2002) Systematic identification of novel protein domain families associated with nuclear functions. Genome Res 12:47–56. https://doi.org/10.1101/gr.203201
Fan Z et al (2009) BCOR regulates mesenchymal stem cell function by epigenetic mechanisms. Nat Cell Biol 11:1002–1009. https://doi.org/10.1038/ncb1913
Felszeghy S, Suomalainen M, Thesleff I (2010) Notch signalling is required for the survival of epithelial stem cells in the continuously growing mouse incisor. Differentiation 80:241–248. https://doi.org/10.1016/j.diff.2010.06.004
Floeth M, Bruckner-Tuderman L (1999) Digenic junctional epidermolysis bullosa: mutations in COL17A1 and LAMB3 genes. Am J Hum Genet 65:1530–1537. https://doi.org/10.1086/302672
Gache Y, Allegra M, Bodemer C, Pisani-Spadafora A, de Prost Y, Ortonne JP, Meneguzzi G (2001) Genetic bases of severe junctional epidermolysis bullosa presenting spontaneous amelioration with aging. Hum Mol Genet 10:2453–2461. https://doi.org/10.1093/hmg/10.21.2453
Gambin T et al (2017) Homozygous and hemizygous CNV detection from exome sequencing data in a Mendelian disease cohort. Nucleic Acids Res 45:1633–1648. https://doi.org/10.1093/nar/gkw1237
Gong M et al (2015) Association of BMP4 polymorphisms with isolated tooth agenesis in a Chinese Han population: a case-control study. Eur Rev Med Pharmacol Sci 19:2188–2194
Gonzaga-Jauregui C et al (2015) Exome sequence analysis suggests that genetic burden contributes to phenotypic variability and complex neuropathy. Cell Rep 12:1169–1183. https://doi.org/10.1016/j.celrep.2015.07.023
Gorlin RJ, Marashi AH, Obwegeser HL (1996) Oculo-facio-cardio-dental (OFCD) syndrome. Am J Med Genet 63:290–292. 10.1002/(SICI)1096-8628(19960503)63:1<290::AID-AJMG47>3.0.CO;2-G
Hilton E et al (2009) BCOR analysis in patients with OFCD and Lenz microphthalmia syndromes, mental retardation with ocular anomalies, and cardiac laterality defects. Eur J Hum Genet 17:1325–1335. https://doi.org/10.1038/ejhg.2009.52
Jumlongras D et al (2001) A nonsense mutation in MSX1 causes witkop syndrome. Am J Hum Genet 69:67–74. https://doi.org/10.1086/321271
Kantaputra P, Sripathomsawat W (2011) WNT10A and isolated hypodontia. Am J Med Genet A 155A:1119–1122. https://doi.org/10.1002/ajmg.a.33840
Kantaputra P, Kaewgahya M, Kantaputra W (2014) WNT10A mutations also associated with agenesis of the maxillary permanent canines, a separate entity. Am J Med Genet A 164A:360–363. https://doi.org/10.1002/ajmg.a.36280
Kantaputra PN, Kaewgahya M, Hatsadaloi A, Vogel P, Kawasaki K, Ohazama A, Ketudat Cairns JR (2015) GREMLIN 2 Mutations and Dental Anomalies. J Dent Res 94:1646–1652. https://doi.org/10.1177/0022034515608168
Karaca E et al (2018) Phenotypic expansion illuminates multilocus pathogenic variation. Genet Med. https://doi.org/10.1038/gim.2018.33
Karadas M, Celikoglu M, Akdag MS (2014) Evaluation of tooth number anomalies in a subpopulation of the North-East of Turkey. Eur J Dent 8:337–341. https://doi.org/10.4103/1305-7456.137641
Kere J et al (1996) X-linked anhidrotic (hypohidrotic) ectodermal dysplasia is caused by mutation in a novel transmembrane protein. Nat Genet 13:409–416. https://doi.org/10.1038/ng0895-409
Khajavi M, Inoue K, Lupski JR (2006) Nonsense-mediated mRNA decay modulates clinical outcome of genetic disease. Eur J Hum Genet 14:1074–1081. https://doi.org/10.1038/sj.ejhg.5201649
Kim JW et al (2013) LAMB3 mutations causing autosomal-dominant amelogenesis imperfecta. J Dent Res 92:899–904. https://doi.org/10.1177/0022034513502054
Kim YJ, Shin TJ, Hyun HK, Lee SH, Lee ZH, Kim JW (2016) A novel de novo mutation in LAMB3 causes localized hypoplastic enamel in the molar region. Eur J Oral Sci 124:403–405. https://doi.org/10.1111/eos.12280
Kircher M, Witten DM, Jain P, O’Roak BJ, Cooper GM, Shendure J (2014) A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 46:310–315. https://doi.org/10.1038/ng.2892
Kiritsi D, Has C, Bruckner-Tuderman L (2013) Laminin 332 in junctional epidermolysis bullosa. Cell Adh Migr 7:135–141. https://doi.org/10.4161/cam.22418
Kivirikko S, McGrath JA, Pulkkinen L, Uitto J, Christiano AM (1996) Mutational hotspots in the LAMB3 gene in the lethal (Herlitz) type of junctional epidermolysis bullosa. Hum Mol Genet 5:231–237. https://doi.org/10.1093/hmg/5.2.231
Lammi L et al (2004) Mutations in AXIN2 cause familial tooth agenesis and predispose to colorectal cancer. Am J Hum Genet 74:1043–1050. https://doi.org/10.1086/386293
Lee KE, Ko J, Le CG, Shin TJ, Hyun HK, Lee SH, Kim JW (2015) Novel LAMB3 mutations cause non-syndromic amelogenesis imperfecta with variable expressivity. Clin Genet 87:90–92. https://doi.org/10.1111/cge.12340
Lek M et al (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536:285–291. https://doi.org/10.1038/nature19057
Liu HC, Zhang J, Wong S, Han D, Zhao HS, Feng HL (2014) Association between rs11001553 of DKK1 and non-syndromic tooth agenesis in the Chinese Han population. Genet Mol Res 13:7133–7139. https://doi.org/10.4238/2014.April.3.4
Lu Y et al (2016) Genetic variants of BMP2 and their association with the risk of non-syndromic tooth agenesis. PLoS One 11:e0158273. https://doi.org/10.1371/journal.pone.0158273
Massink MP et al (2015) Loss-of-function mutations in the WNT co-receptor LRP6 cause autosomal-dominant oligodontia. Am J Hum Genet 97:621–626. https://doi.org/10.1016/j.ajhg.2015.08.014
McGrath JA et al (1995a) Mutations in the 180-kD bullous pemphigoid antigen (BPAG2), a hemidesmosomal transmembrane collagen (COL17A1), in generalized atrophic benign epidermolysis bullosa. Nat Genet 11:83–86. https://doi.org/10.1038/ng0995-83
McGrath JA, Pulkkinen L, Christiano AM, Leigh IM, Eady RAJ, Uitto J (1995b) Altered Laminin 5 Expression Due to Mutations in the Gene Encoding the β3 Chain (LAMB3) in Generalized Atrophic Benign Epidermolysis Bullosa. J Investig Dermatol 104:467–474. https://doi.org/10.1111/1523-1747.ep12605904
Mellerio JE, Eady RA, Atherton DJ, Lake BD, McGrath JA (1998) E210K mutation in the gene encoding the beta3 chain of laminin-5 (LAMB3) is predictive of a phenotype of generalized atrophic benign epidermolysis bullosa. Br J Dermatol 139:325–331. https://doi.org/10.1046/j.1365-2133.1998.02377.x
Nagy E, Maquat LE (1998) A rule for termination-codon position within intron-containing genes: when nonsense affects RNA abundance. Trends Biochem Sci 23:198–199. https://doi.org/10.1016/S0968-0004(98)01208-0
Nakano A, Chao SC, Pulkkinen L, Murrell D, Bruckner-Tuderman L, Pfendner E, Uitto J (2002) Laminin 5 mutations in junctional epidermolysis bullosa: molecular basis of Herlitz vs. non-Herlitz phenotypes. Hum Genet 110:41–51. https://doi.org/10.1007/s00439-001-0630-1
Ng D et al (2004) Oculofaciocardiodental and Lenz microphthalmia syndromes result from distinct classes of mutations in BCOR. Nat Genet 36:411–416. https://doi.org/10.1038/ng1321
Ockeloen CW et al (2016) Novel mutations in LRP6 highlight the role of WNT signaling in tooth agenesis. Genet Med 18:1158–1162. https://doi.org/10.1038/gim.2016.10
Peled A et al (2016) Mutations in TSPEAR, Encoding a Regulator of Notch Signaling, Affect Tooth and Hair Follicle Morphogenesis. PLoS Genet 12:e1006369. https://doi.org/10.1371/journal.pgen.1006369
Posteraro P et al (1998) Compound heterozygosity for an out-of-frame deletion and a splice site mutation in the LAMB3 gene causes nonlethal junctional epidermolysis bullosa. Biochem Biophys Res Commun 243:758–764. https://doi.org/10.1006/bbrc.1998.8180
Poulter JA, El-Sayed W, Shore RC, Kirkham J, Inglehearn CF, Mighell AJ (2014) Whole-exome sequencing, without prior linkage, identifies a mutation in LAMB3 as a cause of dominant hypoplastic amelogenesis imperfecta. Eur J Hum Genet 22:132–135. https://doi.org/10.1038/ejhg.2013.76
Pulkkinen L, Uitto J (1998) Heterozygosity for premature termination codon mutations in LAMB3 in siblings with non-lethal junctional epidermolysis bullosa. J Invest Dermatol 111:1244–1246. https://doi.org/10.1046/j.1523-1747.1998.00399.x
Pulkkinen L, Christiano AM, Gerecke D, Wagman DW, Burgeson RE, Pittelkow MR, Uitto J (1994) A homozygous nonsense mutation in the beta 3 chain gene of laminin 5 (LAMB3) in Herlitz junctional epidermolysis bullosa. Genomics 24:357–360. https://doi.org/10.1006/geno.1994.1627
Pulkkinen L et al (1997) Predominance of the recurrent mutation R635X in the LAMB3 gene in European patients with Herlitz junctional epidermolysis bullosa has implications for mutation detection strategy. J Invest Dermatol 109:232–237. https://doi.org/10.1111/1523-1747.ep12319752
Punta M et al (2012) The Pfam protein families database. Nucleic Acids Res 40:D290–D301. https://doi.org/10.1093/nar/gkr1065
Ranta R, Rintala AE (1983) Correlations between microforms of the Van der Woude syndrome and cleft palate. Cleft Palate J 20:158–162
Reid JG et al (2014) Launching genomics into the cloud: deployment of Mercury, a next generation sequence analysis pipeline. BMC Bioinformatics 15:30. https://doi.org/10.1186/1471-2105-15-30
Song S, Zhao R, He H, Zhang J, Feng H, Lin L (2014) WNT10A variants are associated with non-syndromic tooth agenesis in the general population. Hum Genet 133:117–124. https://doi.org/10.1007/s00439-013-1360-x
Stockton DW, Das P, Goldenberg M, D’Souza RN, Patel PI (2000) Mutation of PAX9 is associated with oligodontia. Nat Genet 24:18–19. https://doi.org/10.1038/71634
Takizawa Y, Pulkkinen L, Shimizu H, Lin L, Hagiwara S, Nishikawa T, Uitto J (1998a) Maternal uniparental meroisodisomy in the LAMB3 region of chromosome 1 results in lethal junctional epidermolysis bullosa. J Invest Dermatol 110:828–831. https://doi.org/10.1046/j.1523-1747.1998.00186.x
Takizawa Y et al (1998b) Novel mutations in the LAMB3 gene shared by two Japanese unrelated families with Herlitz junctional epidermolysis bullosa, and their application for prenatal testing. J Invest Dermatol 110:174–178. https://doi.org/10.1046/j.1523-1747.1998.00105.x
Tao R et al (2006) A novel missense mutation of the EDA gene in a Mongolian family with congenital hypodontia. J Hum Genet 51:498–502. https://doi.org/10.1007/s10038-006-0389-2
The ARIC Investigators (1989) The Atherosclerosis Risk in Communities (ARIC) Study: design and objectives. Am J Epidemiol 129:687–702. https://doi.org/10.1093/oxfordjournals.aje.a115184
Timberlake AT et al (2016) Two locus inheritance of non-syndromic midline craniosynostosis via rare SMAD6 and common BMP2 alleles. Elife 5 https://doi.org/10.7554/eLife.20125
van den Boogaard MJ et al (2012) Mutations in WNT10A are present in more than half of isolated hypodontia cases. J Med Genet 49:327–331. https://doi.org/10.1136/jmedgenet-2012-100750
Vastardis H (2000) The genetics of human tooth agenesis: new discoveries for understanding dental anomalies. Am J Orthod Dentofacial Orthop 117:650–656. https://doi.org/10.1067/mod.2000.103257
Vastardis H, Karimbux N, Guthua SW, Seidman JG, Seidman CE (1996) A human MSX1 homeodomain missense mutation causes selective tooth agenesis. Nat Genet 13:417–421. https://doi.org/10.1038/ng0896-417
Wang X, Zhao Y, Yang Y, Qin M (2015) Novel ENAM and LAMB3 mutations in Chinese families with hypoplastic amelogenesis imperfecta. PLoS One 10:e0116514. https://doi.org/10.1371/journal.pone.0116514
Wu N et al (2015) TBX6 null variants and a common hypomorphic allele in congenital scoliosis. N Engl J Med 372:341–350. https://doi.org/10.1056/NEJMoa1406829
Yin W, Bian Z (2015) The Gene Network Underlying Hypodontia. J Dent Res 94:878–885. https://doi.org/10.1177/0022034515583999
Yu P et al (2016) Mutations in WNT10B Are Identified in Individuals with Oligodontia. Am J Hum Genet 99:195–201. https://doi.org/10.1016/j.ajhg.2016.05.012
Yuan Q et al (2017) Role of WNT10A in failure of tooth development in humans and zebrafish. Mol Genet Genomic Med 5:730–741. https://doi.org/10.1002/mgg3.332
Zhu X, Dai FR, Wang J, Zhang Y, Tan ZP, Zhang Y (2015) Novel BCOR mutation in a boy with Lenz microphthalmia/oculo-facio-cardio-dental (OFCD) syndrome. Gene 571:142–144. https://doi.org/10.1016/j.gene.2015.07.061
Acknowledgements
We appreciate the participation of the patients and their families in this project. We also thank Dr. Ousheng Liu and Dr. Davut Pehlivan for helpful discussions. This work was supported in part by the Scientific and Technological Research Institution of Turkey, TUBITAK-ERA NET (CRANIRARE-2, grant number: SBAG-112S398), Istanbul University Research Fund (Project No: 48398), National Institute of Dental and Craniofacial Research (NIDCR) R03-DE024596 (to AL) and the Baylor-Hopkins Center for Mendelian Genomics, BHCMG (UM1 HG006542, to JRL). The BHCMG is jointly funded by the National Human Genome Research Institute (NHGRI) and National Heart Lung and Blood Institute (NHLBI). The GSP Coordinating Center (U24 HG008956) contributed to cross-program scientific initiatives and provided logistical and general study coordination. JEP was supported by NHGRI K08 HG008986. NW was supported by 2016 Milstein Medical Asian American Partnership Foundation Fellowship Award in Translational Medicine.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
JRL has stock ownership in 23andMe and Lasergen, is a paid consultant for Regeneron, and a co-inventor on multiple United States and European patents related to molecular diagnostics for inherited neuropathies, eye diseases and bacterial genomic fingerprinting. Other authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Du, R., Dinckan, N., Song, X. et al. Identification of likely pathogenic and known variants in TSPEAR, LAMB3, BCOR, and WNT10A in four Turkish families with tooth agenesis. Hum Genet 137, 689–703 (2018). https://doi.org/10.1007/s00439-018-1907-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-018-1907-y