Abstract
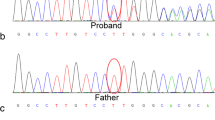
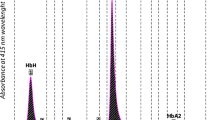
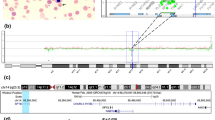
Diamond–Blackfan anemia (DBA) is an inherited red blood cell aplasia that usually presents during the first year of life. The main features of the disease are normochromic and macrocytic anemia, reticulocytopenia, and nearly absent erythroid progenitors in the bone marrow. The patients also present with growth retardation and craniofacial, upper limb, heart and urinary system congenital malformations in ~30–50 % of cases. The disease has been associated with point mutations and large deletions in ten ribosomal protein (RP) genes RPS19, RPS24, RPS17, RPL35A, RPL5, RPL11, RPS7, RPS10, RPS26, and RPL26 and GATA1 in about 60–65 % of patients. Here, we report a novel large deletion in RPL15, a gene not previously implicated to be causative in DBA. Like RPL26, RPL15 presents the distinctive feature of being required both for 60S subunit formation and for efficient cleavage of the internal transcribed spacer 1. In addition, we detected five deletions in RP genes in which mutations have been previously shown to cause DBA: one each in RPS19, RPS24, and RPS26, and two in RPS17. Pre-ribosomal RNA processing was affected in cells established from the patients bearing these deletions, suggesting a possible molecular basis for their pathological effect. These data identify RPL15 as a new gene involved in DBA and further support the presence of large deletions in RP genes in DBA patients.



Similar content being viewed by others
References
Ball SE, McGuckin CP, Jenkins G, Gordon-Smith EC (1996) Diamond–Blackfan anaemia in the UK: analysis of 80 cases from a 20-year birth cohort. Br J Haematol 94:645–653
Beggs AH, Koenig M, Boyce FM, Kunkel LM (1990) Detection of 98% of DMD/BMD gene deletions by polymerase chain reaction. Hum Genet 86:45–48
Campagnoli MF, Garelli E, Quarello P, Carando A, Sv SV, Nobili B, Dl DL, Pecile V, Zecca M, Dufour C, Ramenghi U, Dianzan I (2004) Molecular basis of Diamond–Blackfan anemia: new findings from the Italian registry and a review of the literature. Haematologica 89:480–489
Ceulemans S, van der Ven K, Del-Favero J (2012) Targeted screening and validation of copy number variations. Methods Mol Biol 838:311–328
Choesmel V, Bacqueville D, Rouquette J, Noaillac-Depeyre J, Fribourg S, Cretien A, Leblanc T, Tchernia G, Da Costa L, Gleizes PE (2007) Impaired ribosome biogenesis in Diamond–Blackfan anemia. Blood 109:1275–1283
Choesmel V, Fribourg S, Aguissa-Toure AH, Pinaud N, Legrand P, Gazda HT, Gleizes PE (2008) Mutation of ribosomal protein RPS24 in Diamond–Blackfan anemia results in a ribosome biogenesis disorder. Hum Mol Genet 17:1253–1263
Cmejla R, Cmejlova J, Handrkova H, Petrak J, Pospisilova D (2007) Ribosomal protein S17 gene (RPS17) is mutated in Diamond–Blackfan anemia. Hum Mutat 28:1178–1182
Doherty L, Sheen MR, Vlachos A, Choesmel V, O’Donohue MF, Clinton C, Schneider HE, Sieff CA, Newburger PE, Ball SE, Niewiadomska E, Matysiak M, Glader B, Arceci RJ, Farrar JE, Atsidaftos E, Lipton JM, Gleizes PE, Gazda HT (2010) Ribosomal protein genes RPS10 and RPS26 are commonly mutated in Diamond–Blackfan anemia. Am J Hum Genet 86:222–228
Draptchinskaia N, Gustavsson P, Andersson B, Pettersson M, Willig TN, Dianzani I, Ball S, Tchernia G, Klar J, Matsson H, Tentler D, Mohandas N, Carlsson B, Dahl N (1999) The gene encoding ribosomal protein S19 is mutated in Diamond–Blackfan anaemia. Nat Genet 21:169–175
Farrar JE, Nater M, Caywood E, McDevitt MA, Kowalski J, Takemoto CM, Talbot CC Jr, Meltzer P, Esposito D, Beggs AH, Schneider HE, Grabowska A, Ball SE, Niewiadomska E, Sieff CA, Vlachos A, Atsidaftos E, Ellis SR, Lipton JM, Gazda HT, Arceci RJ (2008) Abnormalities of the large ribosomal subunit protein, Rpl35a, in Diamond–Blackfan anemia. Blood 112:1582–1592
Farrar JE, Vlachos A, Atsidaftos E, Carlson-Donohoe H, Markello TC, Arceci RJ, Ellis SR, Lipton JM, Bodine DM (2011) Ribosomal protein gene deletions in Diamond–Blackfan anemia. Blood 118:6943–6951
Flygare J, Aspesi A, Bailey JC, Miyake K, Caffrey JM, Karlsson S, Ellis SR (2007) Human RPS19, the gene mutated in Diamond–Blackfan anemia, encodes a ribosomal protein required for the maturation of 40S ribosomal subunits. Blood 109:980–986
Gazda HT, Grabowska A, Merida-Long LB, Latawiec E, Schneider HE, Lipton JM, Vlachos A, Atsidaftos E, Ball SE, Orfali KA, Niewiadomska E, Da Costa L, Tchernia G, Niemeyer C, Meerpohl JJ, Stahl J, Schratt G, Glader B, Backer K, Wong C, Nathan DG, Beggs AH, Sieff CA (2006) Ribosomal protein S24 gene is mutated in Diamond–Blackfan anemia. Am J Hum Genet 79:1110–1118
Gazda HT, Sheen MR, Vlachos A, Choesmel V, O’Donohue MF, Schneider H, Darras N, Hasman C, Sieff CA, Newburger PE, Ball SE, Niewiadomska E, Matysiak M, Zaucha JM, Glader B, Niemeyer C, Meerpohl JJ, Atsidaftos E, Lipton JM, Gleizes PE, Beggs AH (2008) Ribosomal protein L5 and L11 mutations are associated with cleft palate and abnormal thumbs in Diamond–Blackfan anemia patients. Am J Hum Genet 83:769–780
Gazda HT, Preti M, Sheen MR, O’Donohue MF, Vlachos A, Davies SM, Kattamis A, Doherty L, Landowski M, Buros C, Ghazvinian R, Sieff CA, Newburger PE, Niewiadomska E, Matysiak M, Glader B, Atsidaftos E, Lipton JM, Gleizes PE, Beggs AH (2012) Frameshift mutation in p53 regulator RPL26 is associated with multiple physical abnormalities and a specific pre-ribosomal RNA processing defect in Diamond–Blackfan anemia. Hum Mutat 33:1037–1044
Glader BE, Backer K (1988) Elevated red cell adenosine deaminase activity: a marker of disordered erythropoiesis in Diamond–Blackfan anaemia and other haematologic diseases. Br J Haematol 68:165–168
Gregory LA, Aguissa-Toure AH, Pinaud N, Legrand P, Gleizes PE, Fribourg S (2007) Molecular basis of Diamond–Blackfan anemia: structure and function analysis of RPS19. Nucleic Acids Res 35:5913–5921
Idol RA, Robledo S, Du HY, Crimmins DL, Wilson DB, Ladenson JH, Bessler M, Mason PJ (2007) Cells depleted for RPS19, a protein associated with Diamond Blackfan anemia, show defects in 18S ribosomal RNA synthesis and small ribosomal subunit production. Blood Cells Mol Dis 39:35–43
Kuramitsu M, Sato-Otsubo A, Morio T, Takagi M, Toki T, Terui K, Wang R, Kanno H, Ohga S, Ohara A, Kojima S, Kitoh T, Goi K, Kudo K, Matsubayashi T, Mizue N, Ozeki M, Masumi A, Momose H, Takizawa K, Mizukami T, Yamaguchi K, Ogawa S, Ito E, Hamaguchi I (2012) Extensive gene deletions in Japanese patients with Diamond–Blackfan anemia. Blood 119:2376–2384
Lipton JM, Atsidaftos E, Zyskind I, Vlachos A (2006) Improving clinical care and elucidating the pathophysiology of Diamond Blackfan anemia: an update from the Diamond Blackfan anemia registry. Pediatr Blood Cancer 46:558–564
O’Donohue MF, Choesmel V, Faubladier M, Fichant G, Gleizes PE (2010) Functional dichotomy of ribosomal proteins during the synthesis of mammalian 40S ribosomal subunits. J Cell Biol 190:853–866
Orfali KA, Ohene-Abuakwa Y, Ball SE (2004) Diamond Blackfan anaemia in the UK: clinical and genetic heterogeneity. Br J Haematol 125:243–252
Preti M, O’Donohue MF, Montel-Lehry N, Bortolin-Cavaille ML, Choesmel V, Gleizes PE (2013) Gradual processing of the ITS1 from the nucleolus to the cytoplasm during synthesis of the human 18S rRNA. Nucleic Acids Res 41:4709–4723
Quarello P, Garelli E, Brusco A, Carando A, Mancini C, Pappi P, Vinti L, Svahn J, Dianzani I, Ramenghi U (2012) High frequency of ribosomal protein gene deletions in Italian Diamond Blackfan anemia patients detected by multiplex ligation-dependent probe amplification (MLPA) assay. Haematologica 97:1813–1817
Sankaran VG, Ghazvinian R, Do R, Thiru P, Vergilio JA, Beggs AH, Sieff CA, Orkin SH, Nathan DG, Lander ES, Gazda HT (2012) Exome sequencing identifies GATA1 mutations resulting in Diamond–Blackfan anemia. J Clin Invest 122:2439–2443
Song MJ, Yoo EH, Lee KO, Kim GN, Kim HJ, Kim SY, Kim SH (2010) A novel initiation codon mutation in the ribosomal protein S17 gene (RPS17) in a patient with Diamond–Blackfan anemia. Pediatr Blood Cancer 54:629–631
Vlachos A, Ball S, Dahl N, Alter BP, Sheth S, Ramenghi U, Meerpohl J, Karlsson S, Liu JM, Leblanc T, Paley C, Kang EM, Leder EJ, Atsidaftos E, Shimamura A, Bessler M, Glader B, Lipton JM (2008) Diagnosing and treating Diamond Blackfan anaemia: results of an international clinical consensus conference. Br J Haematol 142:859–876
Willig TN, Niemeyer CM, Leblanc T, Tiemann C, Robert A, Budde J, Lambiliotte A, Kohne E, Souillet G, Eber S, Stephan JL, Girot R, Bordigoni P, Cornu G, Blanche S, Guillard JM, Mohandas N, Tchernia G (1999) Identification of new prognosis factors from the clinical and epidemiologic analysis of a registry of 229 Diamond–Blackfan anemia patients. DBA group of Societe d’Hematologie et d’Immunologie Pediatrique (SHIP), Gesellshaft fur Padiatrische Onkologie und Hamatologie (GPOH), and the European Society for Pediatric Hematology and Immunology (ESPHI). Pediatr Res 46:553–561
Acknowledgments
We wish to thank the patients and their families for participating in the study. This work was supported by the following Grants: NIH Grants R01 HL107558 and K02 HL111156, a Grant from the DBA Foundation (to H.T.G.), and a Grant from the Agence Nationale de la Recherche (RIBOCRASH) (to P.E.G.).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Landowski, M., O’Donohue, MF., Buros, C. et al. Novel deletion of RPL15 identified by array-comparative genomic hybridization in Diamond–Blackfan anemia. Hum Genet 132, 1265–1274 (2013). https://doi.org/10.1007/s00439-013-1326-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-013-1326-z