Abstract
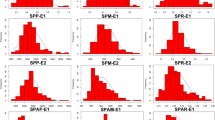
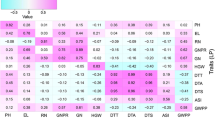
Phosphorus (P) deficiency is one of the major limitations for soybean production. Moreover, it has been well reported P and other mineral elements function interdependently or antagonistically to control nutrients homeostasis in plants. Thus, it is urgently needed to understand the genetic mechanism of the accumulation of mineral elements in response to low-P stress. In this study, to identify single nucleotide polymorphisms (SNPs) and candidate genes controlling the accumulation of mineral elements suffering low-P stress in seedling stage of soybean plants, we measured concentrations of mineral elements, including P, Zn, Fe, Mn, Mg and Ca, in shoots of 211 soybean accessions under normal phosphorus (+P) and low phosphorus (−P) conditions in two hydroponic experiments. And genome-wide association study (GWAS) using high density NJAU 355K SoySNP array and concentrations of five of these mineral elements except P was performed. A total of 36 SNPs distributed on 13 chromosomes were identified to be significantly associated with low-P tolerance, and nine SNPs on chromosome 10 formed a SNP cluster. Meanwhile, the candidate gene GmFeB1 was found to serve as a negative regulator element involved in soybean P metabolism and the haplotype1 (Hap1) of GmFeB1 showed significantly higher shoot Fe concentration under -P condition than that of Hap2. In summary, we uncover 36 SNPs significantly associated with shoot mineral elements concentrations under different P conditions and a soybean low-P related gene GmFeB1, which will provide additional genetic information for soybean low-P tolerance and new gene resources for P-efficient soybean varieties breeding.





Similar content being viewed by others

Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Billard V, Maillard A, Garnica M, Cruz F, Garcia-Mina JM, Yvin JC, Ourry A, Etienne P (2015) Zn deficiency in Brassica napus induces Mo and Mn accumulation associated with chloroplast proteins variation without Zn remobilization. Plant Physiol Biochem 86:66–71. https://doi.org/10.1016/j.plaphy.2014.11.005
Bournier M, Tissot N, Mari S, Boucherez J, Lacombe E, Briat JF, Gaymardet F (2013) Arabidopsis ferritin 1 (AtFer1) gene regulation by the phosphate starvation response 1 (AtPHR1) transcription factor reveals a direct molecular link between iron and phosphate homeostasis. J Biol Chem 288:22670–22680. https://doi.org/10.1074/jbc.M113.482281
Briat JF, Rouached H, Tissot N, Gaymard F, Dubos C (2015) Integration of P, S, Fe, and Zn nutrition signals in Arabidopsis thaliana: potential involvement of PHOSPHATE STARVATION RESPONSE 1 (PHR1). Front Plant Sci 6:290. https://doi.org/10.3389/fpls.2015.00290
Brumbarova T, Ivanov R (2019) The nutrient response transcriptional regulome of Arabidopsis. iScience 19:358–368. https://doi.org/10.1016/j.isci.2019.07.045
Chen H, Seguin P, Archambault A, Constan L, Jabaji S (2009) Gene expression and isoflavone concentrations in soybean sprouts treated with chitosan. Crop Sci 49:224–236. https://doi.org/10.2135/cropsci2007.09.0536
Chen L, Qin L, Zhou L, Li X, Chen Z, Sun L, Wang W, Lin Z, Zhao J, Yamaji N, Ma JF, Gu M, Xu G, Liao H (2019) A nodule-localized phosphate transporter GmPT7 plays an important role in enhancing symbiotic N2 fixation and yield in soybean. New Phytol 221:2013–2025. https://doi.org/10.1111/nph.15541
Cheng Q, Dong L, Gao T, Liu T, Li N, Wang L, Chang X, Wu J, Xu P, Zhang S (2018) The bHLH transcription factor GmPIB1 facilitates resistance to Phytophthora sojae in Glycine max. J Exp Bot 69:2527–2541. https://doi.org/10.1093/jxb/ery103
Cheng L, Min W, Li M, Zhou L, Hsu CC, Yang X, Jiang X, Ruan Z, Zhong Y, Wang ZY, Wang W (2021) Quantitative proteomics reveals that GmENO2 proteins are involved in response to phosphate starvation in the leaves of Glycine max L. Int J Mol Sci 22:920. https://doi.org/10.3390/ijms22020920
Chiou TJ, Lin SI (2011) Signaling network in sensing phosphate availability in plants. Annu Rev Plant Biol 62:185–206. https://doi.org/10.1146/annurev-arplant-042110-103849
Chu S, Wang J, Zhu Y, Liu S, Zhou X, Zhang H, Wang C, Yang W, Tian Z, Cheng H, Yu D (2017) An R2R3-type MYB transcription factor, GmMYB29, regulates isoflavone biosynthesis in soybean. PLoS Genet 13:e1006770. https://doi.org/10.1371/journal.pgen.1006770
Costello R, Wallace TC, Rosanoff A (2016) Magnesium. Adv Nutr 7:199–201. https://doi.org/10.3945/an.115.008524
Cruz TM, Carvalho RF, Richardson DN, Duque P (2014) Abscisic acid (ABA) regulation of Arabidopsis SR protein gene expression. Int J Mol Sci 15:17541–17564. https://doi.org/10.3390/ijms151017541
Danisman S, van Dijk ADJ, Bimbo A, van der Wal F, Hennig L, de Folter S, Angenent GC, Immink RGH (2013) Analysis of functional redundancies within the Arabidopsis TCP transcription factor family. J Exp Bot 64:5673–5685. https://doi.org/10.1093/jxb/ert337
Devaiah BN, Nagarajan VK, Raghothama KG (2007) Phosphate homeostasis and root development in Arabidopsis are synchronized by the zinc finger transcription factor ZAT6. Plant Physiol 145:147–159. https://doi.org/10.1104/pp.107.101691
Du W, Ning L, Liu Y, Zhang S, Yang Y, Wang Q, Chao S, Yang H, Huang F, Cheng H, Yu D (2020) Identification of loci and candidate gene GmSPX-RING1 responsible for phosphorus efficiency in soybean via genome-wide association analysis. BMC Genomics 21:725. https://doi.org/10.1186/s12864-020-07143-3
Gao H, Wang C, Li L, Fu D, Wang C, Zhang Y, Yang P, Zhang T (2020) A novel role of the calcium sensor CBL1 in response to phosphate deficiency in Arabidopsis thaliana. J Plant Physiol 253:153266. https://doi.org/10.1016/j.jplph.2020.153266
Guo M, Ruan W, Zhang Y, Zhang Y, Wang X, Guo Z, Wang L, Zhou T, Paz-Ares J, Yi K (2022) A reciprocal inhibitory module for Pi and iron signaling. Mol Plant 15:138–150. https://doi.org/10.1016/j.molp.2021.09.011
Hammond JP, White PJ (2011) Sugar signaling in root responses to low phosphorus availability. Plant Physiol 156:1033–1040. https://doi.org/10.1104/pp.111.175380
Hansch R, Mendel RR (2009) Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr Opin Plant Biol 12:259–266. https://doi.org/10.1016/j.pbi.2009.05.006
He Y, Zhang X, Li L, Sun Z, Li J, Chen X, Hong G (2021) SPX4 interacts with both PHR1 and PAP1 to regulate critical steps in phosphorus-status-dependent anthocyanin biosynthesis. New Phytol 230:205–217. https://doi.org/10.1111/nph.17139
Hepler PK (2005) Calcium: a central regulator of plant growth and development. Plant Cell 17:2142–2155. https://doi.org/10.1105/tpc.105.032508
Hermans C, Verbruggen N (2005) Physiological characterization of Mg deficiency in Arabidopsis thaliana. J Exp Bot 56:2153–2161. https://doi.org/10.1093/jxb/eri215
Hernandez G, Valdes-Lopez O, Ramirez M, Goffard N, Weiller G, Aparicio-Fabre R, Fuentes SI, Erban A, Kopka J, Udvardi MK, Vance CP (2009) Global changes in the transcript and metabolic profiles during symbiotic nitrogen fixation in phosphorus-stressed common bean plants. Plant Physiol 151:1221–1238. https://doi.org/10.1104/pp.109.143842
Hu D, Kan G, Hu W, Li Y, Hao D, Li X, Yang H, Yang Z, He X, Huang F, Yu D (2019) Identification of loci and candidate genes responsible for pod dehiscence in soybean via genome-wide association analysis across multiple environments. Front Plant Sci 10:811. https://doi.org/10.3389/fpls.2019.00811
Huang C, Barker SJ, Langridge P, Smith FW, Graham RD (2000) Zinc deficiency up-regulates expression of high-affinity phosphate transporter genes in both phosphate-sufficient and -deficient barley roots. Plant Physiol 124:415–422. https://doi.org/10.1104/pp.124.1.415
Jin T, Sun Y, Shan Z, He J, Wang N, Gai J, Li Y (2021) Natural variation in the promoter of GsERD15B affects salt tolerance in soybean. Plant Biotechnol J 19:1155–1169. https://doi.org/10.1111/pbi.13536
Karthikeyan AS, Varadarajan DK, Jain A, Held MA, Carpita NC, Raghothama KG (2007) Phosphate starvation responses are mediated by sugar signaling in Arabidopsis. Planta 225:907–918. https://doi.org/10.1007/s00425-006-0408-8
Kim SA, LaCroix IS, Gerber SA, Guerinot ML (2019) The iron deficiency response in Arabidopsis thaliana requires the phosphorylated transcription factor URI. Proc Natl Acad Sci USA 116:24933–24942. https://doi.org/10.1073/pnas.1916892116
Kumar S, Kumar S, Mohapatra T (2021) Interaction between macro- and micro-nutrients in plants. Front Plant Sci 12:665583. https://doi.org/10.3389/fpls.2021.665583
Leskova A, Giehl RFH, Hartmann A, Fargasova A, von Wiren N (2017) Heavy metals induce iron deficiency responses at different hierarchic and regulatory levels. Plant Physiol 174:1648–1668. https://doi.org/10.1104/pp.16.01916
Li W, Lan P (2015) Genome-wide analysis of overlapping genes regulated by iron deficiency and phosphate starvation reveals new interactions in Arabidopsis roots. BMC Res Notes 8:555. https://doi.org/10.1186/s13104-015-1524-y
Li H, Yang Y, Zhang H, Chu S, Zhang X, Yin D, Yu D, Zhang D (2016) A genetic relationship between phosphorus efficiency and photosynthetic traits in soybean as revealed by QTL analysis using a high-density genetic map. Front Plant Sci 7:924. https://doi.org/10.3389/fpls.2016.00924
Li L, Gao W, Peng Q, Zhou B, Kong Q, Ying Y, Shou H (2018) Two soybean bHLH factors regulate response to iron deficiency. J Integr Plant Biol 60:608–622. https://doi.org/10.1111/jipb.12651
Li T, Zhang W, Yang H, Dong Q, Ren J, Fan H, Zhang X, Zhou Y (2019) Comparative transcriptome analysis reveals differentially expressed genes related to the tissue-specific accumulation of anthocyanins in pericarp and aleurone layer for maize. Sci Rep 9:2485. https://doi.org/10.1038/s41598-018-37697-y
Li Y, Yuan W, Li L, Miao R, Dai H, Zhang J, Xu W (2020) Light-dark modulates root hydrotropism associated with gravitropism by involving amyloplast response in Arabidopsis. Cell Rep 32:108198. https://doi.org/10.1016/j.celrep.2020.108198
Li X, Zhong M, Qu L, Yang J, Liu X, Zhao Q, Liu X, Zhao X (2021) AtMYB32 regulates the ABA response by targeting ABI3, ABI4 and ABI5 and the drought response by targeting CBF4 in Arabidopsis. Plant Sci 310:110983. https://doi.org/10.1016/j.plantsci.2021.110983
Liu J, Yang J, Li R, Shi L, Zhang C, Long Y, Xu F, Meng J (2009) Analysis of genetic factors that control shoot mineral concentrations in rapeseed (Brassica napus) in different boron environments. Plant Soil 320:255–266. https://doi.org/10.1007/s11104-009-9891-6
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆CT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lopez-Arredondo DL, Leyva-Gonzalez MA, Gonzalez-Morales SI, Lopez-Bucio J, Herrera-Estrella L (2014) Phosphate nutrition: improving low-phosphate tolerance in crops. Annu Rev Plant Biol 65:95–123. https://doi.org/10.1146/annurev-arplant-050213-035949
Lu M, Cheng Z, Zhang XM, Huang P, Fan C, Yu G, Chen F, Xu K, Chen Q, Miao Y, Han Y, Feng X, Liu L, Fu YF (2020a) Spatial divergence of PHR-PHT1 modules maintains phosphorus homeostasis in soybean nodules. Plant Physiol 184:236–250. https://doi.org/10.1104/pp.19.01209
Lu S, Dong L, Fang C, Liu S, Kong L, Cheng Q, Chen L, Su T, Nan H, Zhang D, Zhang L, Wang Z, Yang Y, Yu D, Liu X, Yang Q, Lin X, Tang Y, Zhao X, Yang X, Tian C, Xie Q, Li X, Yuan X, Tian Z, Liu B, Weller JL, Kong F (2020b) Stepwise selection on homeologous PRR genes controlling flowering and maturity during soybean domestication. Nat Genet 52:428–436. https://doi.org/10.1038/s41588-020-0604-7
Mattiello EM, Ruiz HA, Neves JC, Ventrella MC, Araujo WL (2015) Zinc deficiency affects physiological and anatomical characteristics in maize leaves. J Plant Physiol 183:138–143. https://doi.org/10.1016/j.jplph.2015.05.014
Ning L, Sun P, Wang Q, Ma D, Hu Z, Zhang D, Zhang G, Cheng H, Yu D (2015) Genetic architecture of biofortification traits in soybean (Glycine max L. merr.) revealed through association analysis and linkage mapping. Euphytica 204:353–369. https://doi.org/10.1007/s10681-014-1340-9
Ogura T, Kobayashi NI, Hermans C, Ichihashi Y, Shibata A, Shirasu K, Aoki N, Sugita R, Ogawa T, Suzuki H, Iwata R, Nakanishi TM, Tanoi K (2020) Short-term magnesium deficiency triggers nutrient retranslocation in Arabidopsis thaliana. Front Plant Sci 11:563. https://doi.org/10.3389/fpls.2020.00563
O’Rourke JA, Yang SS, Miller SS, Bucciarelli B, Liu J, Rydeen A, Bozsoki Z, Uhde-Stone C, Tu ZJ, Allan D, Gronwald JW, Vance CP (2013) An RNA-Seq transcriptome analysis of orthophosphate-deficient white lupin reveals novel insights into phosphorus acclimation in plants. Plant Physiol 161:705–724. https://doi.org/10.1104/pp.112.209254
Perea-Garcia A, Garcia-Molina A, Andres-Colas N, Vera-Sirera F, Perez-Amador MA, Puig S, Penarrubia L (2013) Arabidopsis copper transport protein COPT2 participates in the cross talk between iron deficiency responses and low-phosphate signaling. Plant Physiol 162:180–194. https://doi.org/10.1104/pp.112.212407
Pilon M, Cohu CM, Ravet K, Abdel-Ghany SE, Gaymard F (2009) Essential transition metal homeostasis in plants. Curr Opin Plant Biol 12:347–357. https://doi.org/10.1016/j.pbi.2009.04.011
Ried MK, Wild R, Zhu J, Pipercevic J, Sturm K, Broger L, Harmel RK, Abriata LA, Hothorn LA, Fiedler D, Hiller S, Hothorn M (2021) Inositol pyrophosphates promote the interaction of SPX domains with the coiled-coil motif of PHR transcription factors to regulate plant phosphate homeostasis. Nat Commun 12:384. https://doi.org/10.1038/s41467-020-20681-4
Tokunaga N, Kaneta T, Sato S, Sato Y (2009) Analysis of expression profiles of three peroxidase genes associated with lignification in Arabidopsis thaliana. Physiol Plant 136:237–249. https://doi.org/10.1111/j.1399-3054.2009.01233.x
Tu B, Liu C, Tian B, Zhang Q, Liu X, Herbert SJ (2017) Reduced abscisic acid content is responsible for enhanced sucrose accumulation by potassium nutrition in vegetable soybean seeds. J Plant Res 130:551–558. https://doi.org/10.1007/s10265-017-0912-x
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157:423–447. https://doi.org/10.1046/j.1469-8137.2003.00695.x
Wang J, Chu S, Zhang H, Zhu Y, Cheng H, Yu D (2016) Development and application of a novel genome-wide SNP array reveals domestication history in soybean. Sci Rep 6:20728. https://doi.org/10.1038/srep20728
Wu J, Yuan Y, Zhang X, Zhao J, Song X, Li Y, Li X, Sun R, Koornneef M, Aarts MGM, Wang XW (2008) Mapping QTLs for mineral accumulation and shoot dry biomass under different Zn nutritional conditions in Chinese cabbage (Brassica rapa L. ssp. pekinensis). Plant Soil 310:25–40. https://doi.org/10.1007/s11104-008-9625-1
Wu J, Xu P, Liu L, Zhang S, Wang J, Lin W, Zhong P, Wei L, Dong D (2012) Mapping QTLs for phosphorus-deficiency tolerance in soybean at seedling stage. Int Conf Biomed Eng Biotechnol. https://doi.org/10.1109/iCBEB.2012.269
Wu D, Li C, Jing Y, Wang J, Zhao X, Han Y (2020) Identification of quantitative trait loci underlying soybean (Glycine max) 100-seed weight under different levels of phosphorus fertilizer application. Plant Breed 139:959–968. https://doi.org/10.1111/pbr.12850
Yang Z, Gao Z, Zhou H, He Y, Liu Y, Lai Y, Zheng J, Li X, Liao H (2021) GmPTF1 modifies root architecture responses to phosphate starvation primarily through regulating GmEXPB2 expression in soybean. Plant J 107:525–543. https://doi.org/10.1111/tpj.15307
Yu FW, Zhu XF, Li GJ, Kronzucker HJ, Shi WM (2016) The chloroplast protease AMOS1/EGY1 affects phosphate homeostasis under phosphate stress. Plant Physiol 172:1200–1208. https://doi.org/10.1104/pp.16.00786
Zakikhani H, Khanif YM, Anuar AR, Radziah O, Soltangheisi A (2014) Effects of different levels of molybdenum on uptake of nutrients in rice cultivars. Asian J Crop Sci 6:236–244. https://doi.org/10.3923/ajcs.2014.236.244
Zhang Z, Ersoz E, Lai CQ, Todhunter RJ, Tiwari HK, Gore MA, Bradbury PJ, Yu J, Arnett DK, Ordovas JM, Buckler ES (2010) Mixed linear model approach adapted for genome-wide association studies. Nat Genet 42:355–360. https://doi.org/10.1038/ng.546
Zhang D, Song H, Cheng H, Hao D, Wang H, Kan G, Jin H, Yu D (2014) The acid phosphatase-encoding gene GmACP1 contributes to soybean tolerance to low-phosphorus stress. PLoS Genet 10:e1004061. https://doi.org/10.1371/journal.pgen.1004061
Zhang H, Yang Y, Sun C, Liu X, Lv L, Hu Z, Yu D, Zhang D (2020) Up-regulating GmETO1 improves phosphorus uptake and use efficiency by promoting root growth in soybean. Plant Cell Environ 43:2080–2094. https://doi.org/10.1111/pce.13816
Zhang S, Du H, Ma Y, Li H, Kan G, Yu D (2021) Linkage and association study discovered loci and candidate genes for glycinin and β-conglycinin in soybean (Glycine max L. Merr.). Theor Appl Genet 134:1201–1215. https://doi.org/10.1007/s00122-021-03766-6
Zheng L, Huang F, Narsai R, Wu J, Giraud E, He F, Cheng L, Wang F, Wu P, Whelan J, Shou H (2009) Physiological and transcriptome analysis of iron and phosphorus interaction in rice seedlings. Plant Physiol 151:262–274. https://doi.org/10.1104/pp.109.141051
Zhu XF, Zhao XS, Wu Q, Shen RF (2018) Abscisic acid is involved in root cell wall phosphorus remobilization independent of nitric oxide and ethylene in rice (Oryza sativa). Ann Bot 121:1361–1368. https://doi.org/10.1093/aob/mcy034
Acknowledgements
This work was supported in part by National Natural Science Foundation of China (32090065, 31871649), Ministry of Science and Technology (2017YFE0111000), EUCLEG Horizon 2020 of European Union (727312), and the Bioinformatics Center of Nanjing Agricultural University.
Author information
Authors and Affiliations
Contributions
HC and DY designed the experiment. QW, WD and WY carried out the experiment and analyzed the data. WZ assisted in performing the experiment. QW and WD wrote the manuscript. FH, HC and DY edited the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Additional information
Communicated by Bing Yang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Du, W., Yu, W. et al. Genome-wide association analysis discovered new loci and candidate genes associated with low-phosphorus tolerance based on shoot mineral elements concentrations in soybean. Mol Genet Genomics 297, 843–858 (2022). https://doi.org/10.1007/s00438-022-01895-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-022-01895-1