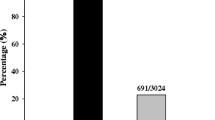
Abstract
Plant major resistance (R) genes are effective in detecting pathogen signal molecules and triggering robust defense responses. Investigating the natural variation in R genes will allow identification of the critical amino acid residues determining recognition specificity in R protein and the discovery of novel R alleles. The rice blast resistance gene Pike, comprising of two adjacent CC-NBS-LRR genes, namely, Pike-1 and Pike-2, confers broad-spectrum resistance to Magnaporthe oryzae. Here, we demonstrated that Pike-1 determined Pike-specific resistance through direct interaction with the pathogen signal molecule AvrPik. Analysis of natural variation in 79 Pike-1 variants in the Asian cultivated rice Oryza sativa and its wild relatives revealed that the CC and NBS regions, particularly the CC region of the Pike-1 protein were the most diversified. We also found that balancing selection had occurred in O. sativa and O. rufipogon to maintain the genetic diversity of the Pike-1 alleles. By analysis of amino acid sequences, we identified 40 Pike-1 variants in these rice germplasms. These variants were divided into three major groups that corresponded to their respective clades. A new Pike allele, designated Pikg, that differed from Pike by a single amino acid substitution (D229E) in the Pike-1 CC region of the Pike protein was identified from wild rice relatives. Pathogen assays of Pikg transgenic plants revealed a unique reaction pattern that was different from that of the previously identified Pike alleles, namely, Pik, Pikh, Pikm, Pikp, Piks and Pi1. These findings suggest that minor amino acid residues in Pike-1/Pikg-1 determine pathogen recognition specificity and plant resistance. As a new blast R gene derived from rice wild relatives, Pikg has potential applications in rice breeding.





Similar content being viewed by others
References
Ashikawa I, Hayashi N, Yamane H, Kanamori H, Wu J, Matsumoto T, Ono K, Yano M (2008) Two adjacent nucleotide-binding site-leucine-rich repeat class genes are required to confer Pikm-specific rice blast resistance. Genetics 180:2267–2276
Bonman JM, De Dios TIV, Khin MM (1986) Physiologic specialization of Pricularia oryzae in the Philippines. Plant Dis 70:767–769
Cesari S, Kanzaki H, Fujiwara T, Bernoux M, Chalvon V, Kawano Y, Shimamoto K, Dodds P, Terauchi R, Kroj T (2014) The NB-LRR proteins RGA4 and RGA5 interact functionally and physically to confer disease resistance. EMBO J 33:1941–1959
Chen X, Shang J, Chen D, Lei C, Zou Y, Zhai W, Liu G, Xu J, Ling Z, Cao G, Ma B, Wang Y, Zhao X, Li S, Zhu L (2006) A B-lectin receptor kinase gene conferring rice blast resistance. Plant J 46:794–804
Chen S, Songkumarn P, Liu J, Wang GL (2009) A versatile zero background T-vector system for gene cloning and functional genomics. Plant Physiol 150:1111–1121
Chen J, Peng P, Tian J, He Y, Zhang L, Liu Z, Yin D, Zhang Z (2015) Pike, a rice blast resistance allele consisting of two adjacent NBS–LRR genes, was identified as a novel allele at the Pik locus. Mol Breeding 35:117
Costanzo S, Jia YL (2010) Sequence variation at the rice blast resistance gene Pi-km locus: Implications for the development of allele specific markers. Plant Sci 178:523–530
Das A, Soubam D, Singh PK, Thakur S, Singh NK, Sharma TR (2012) A novel blast resistance gene, Pi54rh cloned from wild species of rice, Oryza rhizomatis confers broad spectrum resistance to Magnaporthe oryzae. Funct Integr Genomics 12:215–228
De la Concepcion JC, Franceschetti M, Maqbool A, Saitoh H, Terauchi R, Kamoun S, Banfield MJ (2018) Polymorphic residues in rice NLRs expand binding and response to effectors of the blast pathogen. Nat Plants 4:576–585
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1:19–21
Gupta SK, Rai AK, Kanwar SS, Chand D, Singh NK, Sharma TR (2012) The single functional blast resistance gene Pi54 activates a complex defence mechanism in rice. J Exp Bot 63:757–772
He F, Zhang F, Sun W, Ning Y, Wang GL (2018) A versatile vector toolkit for functional analysis of rice genes. Rice 11:27
Hua L, Wu J, Chen C, Wu W, He X, Lin F, Wang L, Ashikawa I, Matsumoto T, Pan Q (2012) The isolation of Pi1, an allele at the Pik locus which confers broad spectrum resistance to rice blast. Theor Appl Genet 125:1047–1055
Huang J, Si W, Deng Q, Li P, Yang S (2014) Rapid evolution of avirulence genes in rice blast fungus Magnaporthe oryzae. BMC Genet 15:45
Imam J, Mandal NP, Variar M, Shukla P (2016) Allele mining and selective patterns of Pi9 gene in a set of rice landraces from India. Front Plant Sci 7:1846
Jones J, Dangl J (2006) The plant immune system. Nature 444:323–329
Kalia S, Rathour R (2019) Current status on mapping of genes for resistance to leaf- and neck-blast disease in rice. 3 Biotech 9:209
Kanzaki H, Yoshida K, Saitoh H, Fujisaki K, Hirabuchi A, Alaux L, Fournier E, Tharreau D, Terauchi R (2012) Arms race co-evolution of Magnaporthe oryzae AVR-Pik and rice Pik genes driven by their physical interactions. Plant J 72:894–907
Kumar GR, Sakthivel K, Sundaram RM, Neeraja CN, Balachandran SM, Rani NS, Viraktamath BC, Madhav MS (2010) Allele mining in crops: prospects and potentials. Biotechnol Adv 28:451–461
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Li J, Wang Q, Li C, Bi Y, Fu X, Wang R (2019) Novel haplotypes and networks of AVR-Pik alleles in Magnaporthe oryzae. BMC Plant Biol 19:204
Longya A, Chaipanya C, Franceschetti M, Maidment JHR, Banfield MJ, Jantasuriyarat C (2019) Gene duplication and mutation in the emergence of a novel aggressive allele of the AVR-Pik effector in the rice blast fungus. Mol Plant-Microbe Interact 32:740–749
Lundberg M, Zhong X, Konrad A, Olsen RA, Råberg L (2020) Balancing selection in pattern recognition receptor signalling pathways is associated with gene function and pleiotropy in a wild rodent. Mol Ecol 29:1990–2003
Maqbool A, Saitoh H, Franceschetti M, Stevenson CEM, Uemura A, Kanzaki H, Kamoun S, Terauchi R, Banfield MJ (2015) Structural basis of pathogen recognition by an integrated HMA domain in a plant NLR immune receptor. Elife 4:e08709
Monteiro F, Nishimura MT (2018) Structural, functional, and genomic diversity of plant NLR proteins: an evolved resource for rational engineering of plant immunity. Annu Rev Phytopathol 56:243–267
Ortiz D, de Guillen K, Cesari S, Chalvon V, Gracy J, Padilla A, Kroj T (2017) Recognition of the Magnaporthe oryzae effector AVR-Pia by the decoy domain of the rice NLR immune receptor RGA5. Plant Cell 29:156–168
Pennisi E (2010) Armed and dangerous. Science 327:804–805
Qu S, Liu G, Zhou B, Bellizzi M, Zeng L, Dai L, Han B, Wang GL (2006) The broad-spectrum blast resistance gene Pi9 encodes a nucleotide-binding site-leucine-rich repeat protein and is a member of a multigene family in rice. Genetics 172:1901–1914
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302
Sanchez PL, Wing RA, Brar DS (2013) The wild relative of rice: genomes and genomics. In: Zhang Q, Wing RA (eds) Plant genetics and genomics: crops and models. Springer, New York, pp 9–25
Shao ZQ, Xue JY, Wu P, Zhang YM, Wu Y, Hang YY, Wang B, Chen JQ (2016) Large-scale analyses of angiosperm nucleotide-binding site-leucine-rich repeat genes reveal three anciently diverged classes with distinct evolutionary patterns. Plant Physiol 170:2095–2109
Shuichi F, Norikuni S, Hironori K, Kazuko O, Takehiko S, Kaworu E, Nagao H, Akira T, Hirohiko H, Kazutoshi O (2009) Loss of function of a proline-containing protein confers durable disease resistance in rice. Science 325:998–1001
Stern A, Doron-Faigenboim A, Erez E, Martz E, Bacharach E, Pupko T (2007) Selecton 2007: advanced models for detecting positive and purifying selection using a Bayesian inference approach. Nucleic Acids Res 35:W506–W511
Swanson WJ, Nielsen R, Yang Q (2003) Pervasive adaptive evolution in mammalian fertilization proteins. Mol Biol Evol 20:18–20
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Tanksley SD, McCouch SR (1997) Seed banks and molecular maps: unlocking genetic potential from the wild. Science 277:1063–1066
Tsunematsu H, Yanoria MJT, Ebron LA, Nagao H, Ikuo A, Hiroshi K, Imbe T, Khush GS (2000) Development of monogenic lines of rice for blast resistance. Breeding Sci 50:229–234
Vo KTX, Lee SK, Halane MK, Song MY, Hoang TV, Kim CY, Park SY, Jeon J, Kim ST, Sohn KH, Jeon JS (2019) Pi5 and Pii paired NLRs are functionally exchangeable and confer similar disease resistance specificity. Mol Cells 42:637–645
Wang ZX, Yano M, Yamanouchi U, Iwamoto M, Monna L, Hayasaka H, Katayose Y, Sasaki T (2010) The Pib gene for rice blast resistance belongs to the nucleotide binding and leucine-rich repeat class of plant disease resistance genes. Plant J 19:55–64
Wang JC, Jia Y, Wen JW, Liu WP, Liu XM, Li L, Jiang ZY, Zhang JH, Guo XL, Ren JP (2013) Identification of rice blast resistance genes using international monogenic differentials. Crop Protect 45:109–116
Xiao G, Yang J, Zhu X, Wu J, Zhou B (2020) Prevalence of ineffective haplotypes at the rice blast resistance (R) gene loci in Chinese elite hybrid rice varieties revealed by sequence-based molecular diagnosis. Rice 13:6
Yang Z, Nielsen R, Goldman N, Pedersen AM (2000) Codon-substitution models for heterogeneous selection pressure at amino acid sites. Genetics 155:431–449
Yang S, Gu T, Pan C, Feng Z, Ding J, Hang Y, Chen JQ, Tian D (2008) Genetic variation of NBS-LRR class resistance genes in rice lines. Theor Appl Genet 116:165–177
Yeatman CW, Kafton D, Wilkes G (1985) Plant genetic resources: a conservation imperative. Westview Press, Boulder
Yoshida K, Saitoh H, Fujisawa S, Kanzaki H, Matsumura H, Tosa Y, Chuma I, Takano Y, Win J, Kamoun S, Terauchi R (2009) Association genetics reveals three novel avirulence genes from the rice blast fungal pathogen Magnaporthe oryzae. Plant Cell 21:1573–1591
Yuan B, Zhai C, Wang W, Zeng X, Xu X, Hu H, Lin F, Wang L, Pan Q (2011) The Pik-p resistance to Magnaporthe oryzae in rice is mediated by a pair of closely linked CC-NBS-LRR genes. Theor Appl Genet 122:1017–1028
Zhai C, Lin F, Dong Z, He X, Yuan B, Zeng X, Wang L, Pan Q (2011) The isolation and characterization of Pik, a rice blast resistance gene which emerged after rice domestication. New Phytol 189:321–334
Zhai C, Zhang Y, Yao N, Lin F, Liu Z, Dong Z, Wang L, Pan Q (2014) Function and interaction of the coupled genes responsible for Pik-h encoded rice blast resistance. PLoS ONE 9:e98067–e98067
Acknowledgements
The authors thank Dr. Zhijian Xu (Guangxi Academy of Agricultural Sciences of China) for providing the wild rice accessions used in the present study. Also thank Dr. Cailin Lei (Chinese Academy of Agricultural Sciences), Prof. Weiren Wu (Fujian Agriculture and Forestry University) for providing rice blast resistance gene monogenic lines, and Dr. Dazhao Yu (Hubei Academy of Agricultural Sciences of China) for providing some of the M. oryzae isolates inoculated in the present study. This work was financed by the Special Transgenic Program of the Ministry of Agriculture in China (No. 2016ZX08001004-002), National Natural Science Foundation of China (No.31901522) and Collaborative Innovation Center of Hubei Province for Hybrid Rice.
Funding
This work was financed by the Special Transgenic Program of the Ministry of Agriculture in China (No. 2016ZX08001004-002), National Natural Science Foundation of China (No.31901522) and Collaborative Innovation Center of Hubei Province for Hybrid Rice.
Author information
Authors and Affiliations
Contributions
FM, YH and ZZ conceived and designed research. FM conducted experiments and analysed data. YH, JC, XL, HW, MZ and SL contributed to the experiment and data analysis. FM, QC and ZZ wrote and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Stefan Hohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Meng, F., He, Y., Chen, J. et al. Analysis of natural variation of the rice blast resistance gene Pike and identification of a novel allele Pikg. Mol Genet Genomics 296, 939–952 (2021). https://doi.org/10.1007/s00438-021-01795-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-021-01795-w