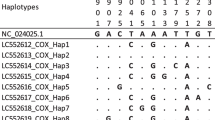
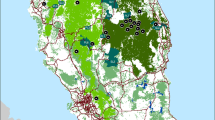
Abstract
Ticks are hematophagous ectoparasites associated with a wide range of vertebrate hosts. Within this group, the Ixodidae family stands out, in which the Ixodes genus contains at least 245 species worldwide, from which 55 species are present in the Neotropical region. Ixodes affinis, a tick described in 1899, has a wide distribution from the Southern Cone of America to the United States. However, since its description, morphological variability has been reported among its populations. Furthermore, attempts have been made to clarify its status as a species complex using mitochondrial markers, but mainly in restricted populations of South and Central America. Thus, information related to populations of the transition region between the Neotropical and Nearctic zones is lacking. For these reasons, the objectives of the study were to evaluate the genetic diversity and structure of I. affinis across the Americas and to compile all the published records of I. affinis in America, to elucidate the host-parasite relationships and to identify their geographical distribution. For this, a phylogeny, and AMOVA analyses were performed to assess the genetic structure of samples obtained by field work in South Carolina, USA and Yucatán, Mexico. A total of 86 sequences were retrieved from a fragment of the 16S region. Phylogeny and genetic structure analysis showed four groups that were geographically and genetically related with high branch support and Fst values, all of them statistically significant. The results obtained support the hypothesis that I. affinis it corresponds to a complex of four species, which must be validated through future morphological comparisons.




Similar content being viewed by others
Data availability
The data that support the results of this study are available in GenBank, under the following accessions numbers I. affinis 16 s-rDNA (OR357724-OR357735 and OR751593-OR751608).
References
Arellano E, González-Cozátl FX, Rogers DS (2005) Molecular systematics of Middle American harvest mice Reithrodontomys (Muridae), estimated from mitochondrial cytochrome b gene sequences. Mol Phylogenet Evol 37(2):529–540. https://doi.org/10.1016/j.ympev.2005.07.021
Almendra AL, Rogers DS (2012) Biogeography of Central American Mammals. Patterns and Processes. In: Patterson BD, Costa LB (eds) Bones, Clones, and Biomes: The History and Geography of Recent Neotropical Mammals. University of Chicago Press, 351–378 pp
Beati L, Nava S, Burkman EJ, Barros-Battesti DM, Labruna MB, Guglielmone AA, Cáceres AG, Guzmán-Cornejo CM, León R, Durden LA, Faccini JLH (2013) Amblyomma cajennense (Fabricius, 1787) (Acari: Ixodidae), the Cayenne tick: phylogeography and evidence for allopatric speciation. BMC Evol Biol 13:267. https://doi.org/10.1186/1471-2148-13-267
Bermúdez SE, Esser HJ, Miranda -CR, Moreno RS (2015) Wild carnivores (Mammalia) as hosts for ticks (Ixodida) in Panama. Syst Appl Acarol 20(1):13–19. https://doi.org/10.11158/saa.20.1.2
Bermúdez SE, Castro AM, Trejos D, García GG, Gabster A, Miranda RJ, Zaldívar Y, Paternina LE (2016) Distribution of Spotted Fever group Rickettsiae in hard ticks (Ixodida: Ixodidae) from Panamanian urban and rural environments (2007–2013). EcoHealth 13(2):274–284. https://doi.org/10.1007/s10393-016-1118-8
Bermúdez S, Martínez-Mandiche J, Domínguez L, Gonzalez C, Chavarria O, Moreno A, Góndola J, Correa N, Rodríguez I, Castillo B, Smith D, Martínez AA (2021) Diversity of Rickettsia in ticks collected from wild animals in Panama. Ticks Tick Borne Dis 12(4): 101723. https://doi.org/10.1016/j.ttbdis.2021.101723
Bitencourth K, Amorim M, De Oliveira SV, Caetano RL, Voloch CM, Gazêta GS (2017) Amblyomma sculptum: genetic diversity and rickettsias in the Brazilian Cerrado biome. Med Vet Entomol 31(4):427–437. https://doi.org/10.1111/mve.12249
Carreno RA, Durden LA, Brooks DR, Abrams A, Hoberg EP (2001) Parelaphostrongylus tenuis (Nematoda: Protostrongylidae) and others parasites of white-tailed deer (Odocoileus virginianus) in Costa Rica. Comp Parasitol 68:177–184
Ceballos G (2014) Mammals of Mexico. Johns Hopkins University Press, Baltimore
Clark KL, Oliver JH Jr, Grego JM, James AM, Durden LA, Banks CW (2001) Host associations of ticks parasitizing rodents at Borrelia burgdorferi enzootic sites in South Carolina. J Parasitol 87(6):1379–1386. https://doi.org/10.1645/0022-3395(2001)087[1379:HAOTPR]2.0.CO;2
Clark KL, Oliver JH Jr, James AM, Durden LA, Banks CW (2002) Prevalence of Borrelia burgdorferi sensu lato infection among rodents and host-seeking ticks in South Carolina. J Med Entomol 39(1):198–206. https://doi.org/10.1603/0022-2585-39.1.198
Clark K (2004) Borrelia species in host-seeking ticks and small mammals in northern Florida. J Clin Microbiol 42(11):5076–5086. https://doi.org/10.1128/JCM.42.11.5076-5086.2004
Colunga-Salas P, Hernández-Canchola G (2021) Bats and humans during the SARS-CoV-2 outbreak: The case of bat-coronaviruses from Mexico. Transbound Emerg Dis 68:987–992. https://doi.org/10.1111/tbed.13751
Cooley RA, Kohls GM (1945) The genus ixodes in North America. NIH Bulletin 184:1–246
Cumbie AN, Heller EL, Bement ZJ, Phan A, Walters EL, Hynes WL, Gaff HD (2021) Passerine birds as hosts for Ixodes ticks infected with Borrelia burgdorferi sensu stricto in southeastern Virginia. Ticks Tick-Borne Dis 12(3):101650. https://doi.org/10.1016/j.ttbdis.2021.101650
Domínguez L, Miranda RJ, Torres S, Moreno R, Ortega J, Bermúdez SE (2019) Hard tick (Acari: Ixodidae) survey of Oleoducto trail, Soberania National Park, Panama. Ticks Tick-Borne Dis 10(4):830–837. https://doi.org/10.1016/j.ttbdis.2019.04.001
Dumaidi K, Qaraqe H, Al-Jawabreh A, Abu-Helu R, Samarah F, Al-Jawabreh H (2020) Genetic diversity, haplotype analysis, and risk factor assessment of Hepatitis A virus isolates from the West Bank, Palestine during the period between 2014 and 2016. PLoS One 15(12):e0240339. https://doi.org/10.1371/journal.pone.0240339
Durden LA, Oliver JH Jr, Kinsey AA (2001) Ticks (Acari: Ixodidae) and spirochetes (Spirochaetaceae: Spirochaetales) recovered from birds on a Georgia Barrier Island. J Med Entomol 38(2):231–236. https://doi.org/10.1603/0022-2585-38.2.231
Dzul-Rosado KR, Arroyo-Solís KA, Torres-Monroy AJ, Arias-León JJ, Peniche-Lara GF, Puerto-Manzano FI, Landa-Flores MG, Del Mazo-López JC, Salceda-Sánchez B (2023) Tick-associated diseases identified from hunting dogs during the COVID-19 pandemic in a Mayan community in Yucatan. Mexico. Open Vet J 13(6):794–800. https://doi.org/10.5455/OVJ.2023.v13.i6.14
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797. https://doi.org/10.1093/nar/gkh340
Esser HJ, Foley JE, Bongers F, Herre EA, Miller MJ, Prins HH, Jansen PA (2016a) Host body size and the diversity of tick assemblages on Neotropical vertebrates. Int J Parasitol: Parasites Wildlife 5(3):295–304. https://doi.org/10.1016/j.ijppaw.2016.10.001
Esser HJ, Herre EA, Blüthgen N, Loaiza JR, Bermúdez SE, Jansen PA (2016b) Host specificity in a diverse Neotropical tick community: an assessment using quantitative network analysis and host phylogeny. Parasit Vectors 9(1):372. https://doi.org/10.1186/s13071-016-1655-6
Estrada-Peña A, Guglielmone AA, Nava S (2023) Worldwide host associations of the tick genus Ixodes suggest relationships based on environmental sharing rather than on co-phylogenetic events. Parasit Vectors 16(1):75. https://doi.org/10.1186/s13071-022-05641-9
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10(3):564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Fairchild GB (1943) An annotated list of the bloodsucking insects, ticks and mites known from Panama. Am J Trop Med Hyg 23:569–591. https://doi.org/10.4269/ajtmh.1943.s1-23.569
Fairchild GB, Kohls GM, Tipton VJ (1966) The ticks of Panama (Acarina: Ixodoidea). In: Wenzel WR, Tipton VJ (eds) Ectoparasites of Panama. Field Museum of Natural History, Chicago, pp 167–219
Flores FS, Saracho-Bottero MN, Sebastian PS, Venzal JM, Mangold AJ, Nava S (2020) Borrelia genospecies in Ixodes sp. cf. Ixodes affinis (Acari: Ixodidae) from Argentina. Ticks Tick-Borne Dis 11(6):101546. https://doi.org/10.1016/j.ttbdis.2020.101546
Fonseca F (1960) Notes d’Acarologie XLV. Enquête acarologique au Pérou. Acarologia 2:1–34
Forrester DJ, Conti JA, Belden RC (1985) Parasites of the Florida panther (Felis concolor coryi). Proc Helminthol Soc Wash 52(1):95–97
Forrester DJ, McLaughlin GS, Telford SR Jr, Foster GW, McCown JW (1996) Ectoparasites (Acari, Mallophaga, Anoplura, Diptera) of white-tailed deer, Odocoileus virginianus, from southern Florida. J Med Entomol 33(1):96–101. https://doi.org/10.1093/jmedent/33.1.96
Fu YX, Li WH (1993) Statistical tests of neutrality of mutations. Genetics 133(3):693–709. https://doi.org/10.1093/genetics/133.3.693
Fu YX (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147(2):915–925. https://doi.org/10.1093/genetics/147.2.915
Fumagalli M, Vieira FG, Korneliussen TS, Linderoth T, Huerta-Sánchez E, Albrechtsen A, Nielsen R (2013) Quantifying population genetic differentiation from next-generation sequencing data. Genetics 195(3):979–992. https://doi.org/10.1534/genetics.113.154740
Gerrish RR, Ossorio JM (1965) New record of the tick Ixodes affinis in the United States. J Econ Entomol 58(2):369. https://doi.org/10.1093/jee/58.2.369
Ghosh P, Saleh MN, Sundstrom KD, Ientile M, Little SE (2021) Ixodes spp. from dogs and cats in the United States: Diversity, seasonality, and prevalence of Borrelia burgdorferi and Anaplasma phagocytophilum. Vector Borne Zoonotic Dis 21(1):11–19. https://doi.org/10.1089/vbz.2020.2637
Guglielmone AA, Estrada-Peña A, Keirans JE, Robbins RG (2003) Ticks (Acari: Ixodida) of the neotropical zoogeographic region. International consortium on ticks and tick-borne diseases. Atalanta, Houten, The Netherlands, 173 pp
Guglielmone AA, Robbins RG, Apanaskevich DA, Petney TN, Estrada-Peña A, Horak IG (2014) The hard ticks of the world. Springer, Dordrecht, pp 978–994
Guglielmone AA, Robbins RG (2018) Hard ticks (Acari: Ixodida: Ixodidae) parasitizing humans. Cham: Springer, US, 210 pp. https://doi.org/10.1007/978-3-319-95552-0
Guglielmone AA, Petney TN, Robbins RG (2020) Ixodidae (Acari: Ixodoidea): descriptions and redescriptions of all known species from 1758 to December 31, 2019. Zootaxa 4871(1):1–322. https://doi.org/10.11646/ZOOTAXA.4871.1.1
Guglielmone A, Nava S, Robbins R (2021) Neotropical hard ticks (Acari: Ixodida: Ixodidae). A critical analysis of their taxonomy, distribution, and host relationships Springer Nature Switzerland, Cham 486 p
Guzmán-Cornejo C, Robbins RG, Pérez TM (2007) The Ixodes (Acari: Ixodidae) of Mexico: parasite-host and host-parasite checklists. Zootaxa 1553(1):47–58. https://doi.org/10.11646/zootaxa.1553.1.2
Guzmán-Cornejo C, Robbins RG (2010) The genus Ixodes (Acari: Ixodidae) in Mexico: adult identification keys, diagnoses, hosts and distribution. Rev Mex Biodiversidad 81(2):289–298. https://doi.org/10.22201/ib.20078706e.2010.002.254
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hasegawa M, Kishino H, Yano T (1985) Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J Mol Evol 22:160–174. https://doi.org/10.1007/BF02101694
Harrison BA, Rayburn WH Jr, Toliver M, Powell EE, Engber BR, Durden LA, Robbins RG, Prendergast BF, Whitt PB (2010) Recent discovery of widespread Ixodes affinis (Acari: Ixodidae) distribution in North Carolina with implications for Lyme disease studies. J Vector Ecol 35(1):174–179. https://doi.org/10.1111/j.1948-7134.2010.00044.x
Heller EL, Wright CL, Nadolny RM, Hynes WL, Gaff HD, Walters EL (2016) New records of Ixodes affinis (Acari: Ixodidae) parasitizing avian hosts in Southeastern Virginia. J Med Entomol 53(2):441–445. https://doi.org/10.1093/jme/tjv175
Hernández-Canchola G, León-Paniagua L (2017) Genetic and ecological processes promoting early diversification in the lowland Mesoamerican bat Sturnira parvidens (Chiroptera: Phyllostomidae). Mol Phylogenet Evol 114:334–345. https://doi.org/10.1016/j.ympev.2017.06.015
Hertz JC, Ferree Clemons BC, Lord CC, Allan SA, Kaufman PE (2017) Distribution and host associations of ixodid ticks collected from wildlife in Florida, USA. Exp Appl Acarol 73(2):223–236. https://doi.org/10.1007/s10493-017-0183-1
Hoffmann A (1962) Monografía de los Ixodidae de México. Parte I Rev Soc Mex Hist Nat 23:191–307
Hornok S, Kontschán J, Estrada-Peña A, de Mera IG, Tomanović S, de la Fuente J (2015) Contributions to the morphology and phylogeny of the newly discovered bat tick species, Ixodes ariadnae in comparison with I. vespertilionis and I. simplex. Parasit Vectors 8(1):1–7. https://doi.org/10.1186/s13071-015-0665-0
Kalyaanamoorthy S, Minh BQ, Wong TKF, Von Haeseler A, Jermiin LS (2017) ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat Methods 14(6):587–589. https://doi.org/10.1038/nmeth.4285
Keirans JE, Clifford CM (1978) The genus Ixodes in the United States: a scanning electron microscope study and key to the adults. J Med Entomol Suppl 2:1–149. https://doi.org/10.1093/jmedent/15.suppl2.1
Kellogg FE, Kistner TP, Strickland RK, Gerrish RR (1971) Arthropod parasites collected from white-tailed deer. J Med Entomol 8(5):495–498. https://doi.org/10.1093/jmedent/8.5.495
Klompen JSH, Black WC IV, Keirans JE, Norris DE (2000) Systematics and biogeography of hard ticks, a total evidence approach. Cladistics 16(1):79–102. https://doi.org/10.1006/clad.1999.0126
Kohls GM, Rogers AJ (1953) Note on the occurrence of the tick Ixodes affinis Neumann in the United States. J Parasitol 39(6):669
Lado P, Nava S, Labruna MB, Szabo MPJ, Durden LA, Bermudez SE, Montagna M, Sánchez Quirós AC, Beati L (2016) Amblyomma parvum Aragão, 1908 (Acari: Ixodidae): Phylogeography and systematic considerations. Ticks Tick-Borne Dis 7(5):817–827. https://doi.org/10.1016/j.ttbdis.2016.03.017
Larsson A (2014) AliView: a fast and lightweight alignment viewer and editor for large datasets. Bioinformatics 30(22):3276–3278. https://doi.org/10.1093/bioinformatics/btu531
Lavender DR, Oliver JH Jr (1996) Ticks (Acari: Ixodidae) in Bulloch County, Georgia. J Med Entomol 33(2):224–231. https://doi.org/10.1093/jmedent/33.2.224
Leigh JW, Bryant D (2015) POPART: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116. https://doi.org/10.1111/2041-210X.12410
Lepage D (2016) Avibase. http://avibase.bsc-eoc.org/avibase.jsp?lang=EN (accessed 14 October 2016)
Lepage D, Vaidya G, Guralnick RP (2014) Avibase–a database system for managing and organizing taxonomic concepts. ZooKeys 420:117–135. https://doi.org/10.3897/zookeys.420.7089
Lin T, Oliver JH Jr, Gao L, Kollars TM Jr, Clark KL (2001) Genetic heterogeneity of Borrelia burgdorferi sensu lato in the southern United States based on restriction fragment length polymorphism and sequence analysis. J Clin Microbiol 39(7):2500–2507. https://doi.org/10.1128/JCM.39.7.2500-2507.2001
Lopes MG, Junior JM, Foster RJ, Harmsen BJ, Sanchez E, Martins TF, Quigley H, Marcili A, Labruna MB (2016) Ticks and rickettsiae from wildlife in Belize, Central America. Parasit Vectors 9:62. https://doi.org/10.1186/s13071-016-1348-1
Lozano-Sardaneta YN, Viveros-Santos V, Colunga-Salas P, Rodríguez-Rojas JJ, Becker I (2023) Is Psathyromyia shannoni (Diptera: Psychodidae: Phlebotominae) a species complex? Retrospective study of genetic diversity of COI gene, pathogens and geographic distribution. Acta Trop 238:106807. https://doi.org/10.1016/j.actatropica.2022.106807
Lund HO, Marshall CM, Hayes FA (1962) The Occurrence of Ixodes affinis Neumann on Blackbeard Island, Georgia. Proc Entomol Soc Wash 64:105–106
Maggi RG, Reichelt S, Toliver M, Engber B (2010) Borrelia species in Ixodes affinis and Ixodes scapularis ticks collected from the coastal plain of North Carolina. Ticks Tick-Borne Dis 1(4):168–171. https://doi.org/10.1016/j.ttbdis.2010.08.003
Maggi RG, Toliver M, Richardson T, Mather T, Breitschwerdt EB (2019) Regional prevalences of Borrelia burgdorferi, Borrelia bissettiae, and Bartonella henselae in Ixodes affinis, Ixodes pacificus and Ixodes scapularis in the USA. Ticks Tick-Borne Dis 10(2):360–364. https://doi.org/10.1016/j.ttbdis.2018.11.015
Martínez-Ortiz D, Torres-Castro M, López-Avila K, Koyoc-Cardeña E, Manrique-Saide P (2019) Rickettsia sp. en garrapatas (Acari: Ixodidae) que infestan perros de una comunidad rural con antecedentes de rickettsiosis, Yucatán, Mexico. Biomédicac 30:43–50. https://doi.org/10.32776/revbiomed.v30i2.650
Mattar S, Lopez-Valencia G (1998) Searching for Lyme disease in Colombia: a preliminary study on the vector. J Med Entomol 35(3):324–326. https://doi.org/10.1093/jmedent/35.3.324
Montenegro VM, Delgado M, Miranda RJ, Domínguez L, Vargas-Muñoz M, Bermúdez SE (2021) Free-living hard ticks (Ixodida: Ixodidae) from three different natural environments of Costa Rica. Ticks Tick-Borne Dis 12(6):101811. https://doi.org/10.1016/j.ttbdis.2021.101811
Mysterud A, Hügli C, Viljugrein H (2021) Tick infestation on medium-large-sized mammalian hosts: are all equally suitable to Ixodes ricinus adults? Parasit Vectors 14(1):254. https://doi.org/10.1186/s13071-021-04775-6
Nadolny RM, Wright CL, Hynes WL, Sonenshine DE, Gaff HD (2011) Ixodes affinis (Acari: Ixodidae) in southeastern Virginia and implications for the spread of Borrelia burgdorferi, the agent of Lyme disease. J Vector Ecol 36(2):464–467. https://doi.org/10.1111/j.1948-7134.2011.00191.x
Nadolny RM, Wright CL, Sonenshine DE, Hynes WL, Gaff HD (2014) Ticks and spotted fever group rickettsiae of southeastern Virginia. Ticks Tick-Borne Dis 5(1):53–57. https://doi.org/10.1016/j.ttbdis.2013.09.001
Nadolny R, Gaff H, Carlsson J, Gauthier D (2015) Comparative population genetics of two invading ticks: Evidence of the ecological mechanisms underlying tick range expansions. Infect Genet Evol 35:153–162. https://doi.org/10.1016/j.meegid.2015.08.009
Nadolny RN, Gauthier DT, Gaff HD, Bermúdez SE (2016) Preliminary assessment of the population genetics of Ixodes affinis (Ixodida: Ixodidae) in North and Central America. Syst Appl Acarol 21(10):1300–1308. https://doi.org/10.11158/saa.21.10.2
Nadolny RM, Gaff HD (2018) Natural history of Ixodes affinis in Virginia. Ticks Tick-Borne Dis 9(1):109–119. https://doi.org/10.1016/j.ttbdis.2017.09.016
Nadolny RM, Toliver M, Gaff HD, Snodgrass JG, Robbins RG (2021) Focus stacking images of morphological character states for differentiating the adults of Ixodes affinis and Ixodes scapularis (Acari: Ixodidae) in areas of sympatry. J Med Entomol 58(4):1941–1947. https://doi.org/10.1093/jme/tjab058
Nava S, Beati L, Venzal JM, Durden LA, Bermudez SE, Tarragona EL, Mangold AJ, Gleason B, Mastropaolo M, Guglielmone AA (2023) Description of two new species in the Ixodes ricinus complex from the New World (Acari: Ixodidae), and redescription of Ixodes affinis Neumann, 1899. Zootaxa 5361:53–73. https://doi.org/10.11646/ZOOTAXA.5361.1.2
Nelder MP, Reeves WK (2005) Ectoparasites of road-killed vertebrates in Northwestern South Carolina, USA. Vet Parasitol 129(3–4):313–322. https://doi.org/10.1016/j.vetpar.2004.02.029
Neumann LG (1899) Révision de la famille des ixodidés (3e mémoire). Mémoires Soc Zoologique France 12:107–294
Nguyen LT, Schmidt HA, Von Haeseler A, Minh BQ (2015) IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Norris DE, Klompen JS, Keirans JE, Black WC 4th (1996) Population genetics of Ixodes scapularis (Acari: Ixodidae) based on mitochondrial 16S and 12S genes. J Med Entomol 33(1):78–89. https://doi.org/10.1093/jmedent/33.1.78
Norris DE, Johnson BJ, Piesman J, Maupin GO, Clark JL, Black WC 4th (1999) Population genetics and phylogenetic analysis of Colorado Borrelia burgdorferi. Am J Trop Med Hyg 60(4):699–707. https://doi.org/10.4269/ajtmh.1999.60.699
Ojeda-Chi MM, Rodriguez-Vivas RI, Esteve-Gasent MD, Pérez de León A, Modarelli JJ, Villegas-Perez S (2019a) Molecular detection of rickettsial tick-borne agents in white-tailed deer (Odocoileus virginianus yucatanensis), mazama deer (Mazama temama), and the ticks they host in Yucatan, Mexico. Ticks Tick-Borne Dis 10(2):365–370. https://doi.org/10.1016/j.ttbdis.2018.11.018
Ojeda-Chi MM, Rodriguez-Vivas RI, Esteve-Gasent MD, Pérez de León AA, Modarelli JJ, Villegas-Perez SL (2019b) Ticks infesting dogs in rural communities of Yucatan, Mexico and molecular diagnosis of rickettsial infection. Transbound Emerg Dis 66(1):102–110. https://doi.org/10.1111/tbed.12990
Oliver JH Jr, Keirans JE, Lavender DR, Hutcheson HJ (1987) Ixodes affinis Neumann (Acari: Ixodidae): new host and distribution records, description of immatures, seasonal activities in Georgia, and laboratory rearing. J Parasitol 73(3):646–652. https://doi.org/10.2307/3282149
Oliver Jr JH, Lin T, Gao L, Clark KL, Banks CW, Durden LA, James AM, Chandler Jr FW (2003) An enzootic transmission cycle of Lyme borreliosis spirochetes in the Southeastern United States. Proc Natl Acad Sci U S A 100(20):11642–11645.https://doi.org/10.1073/pnas.1434553100
Onofrio VC, Guglielmone AA, Barros-Battesti D, Gianizella SL, Marcili A, Quadros RM, Marques S, Labruna MB (2020) Description of a new species of Ixodes (Acari: Ixodidae) and first report of Ixodes lasallei and Ixodes bocatorensis in Brazil. Ticks Tick-Borne Dis 11(4):101423. https://doi.org/10.1016/j.ttbdis.2020.101423
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35(3):526–528. https://doi.org/10.1093/bioinformatics/bty633
Patterson BD, Solari S, Velazco PM (2012) The role of the Andes in the diversification and biogeography of neotropical mammals. In: Patterson BD, Costa LB (eds) Bones, Clones, and Biomes: The History and Geography of Recent Neotropical Mammals. University of Chicago Press, pp 351–378
Peniche-Lara G, Jimenez-Delgadillo B, Munoz-Zanzi C, Cárdenas-Marrufo M, Pérez-Osorio C, Arias-León J (2018) Presence of Rickettsia species in a marginalized area of Yucatan, Mexico. J Trop Med 2018:7675828. https://doi.org/10.1155/2018/7675828
Polsomboon S, Hoel DF, Murphy JR, Linton YM, Motoki M, Robbins RG, Bautista K, Bricen OI, Achee NL, Grieco JP, Ching WM, Chao CC (2017) Molecular detection and identification of Rickettsia species in ticks (Acari: Ixodidae) collected from Belize, Central America. J Med Entomol 54(6):1718–1726. https://doi.org/10.1093/jme/tjx141
Poucher KL, Hutcheson HJ, Keirans JE, Durden LA, Black WC IV (1999) Molecular genetic key for the identification of 17 Ixodes species of the United States (Acari: Ixodidae): a methods model. J Parasitol 85(4):623–629
QGIS (2016) QGIS Geographic information system 2.18. Open-source geospatial foundation project. Available from: http://www.qgis.org/ (accessed 18 December 2017)
Ramírez-Pulido J, González-Ruiz N, Gardner AL, Arroyo-Cabrales J (2014) List of recent land mammals of Mexico. Special Publ Museum Texas Tech Univ 63:1–69
Rodríguez-Vivas RI, Apanaskevich DA, Ojeda-Chi MM, Trinidad-Martínez I, Reyes-Novelo E, Esteve-Gassent MD, Pérez de León AA (2016) Ticks collected from humans, domestic animals, and wildlife in Yucatán, Mexico. Vet Parasitol 215:106–113. https://doi.org/10.1016/j.vetpar.2015.11.010
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34(12):3299–3302. https://doi.org/10.1093/molbev/msx248
Sánchez-Montes S, Salceda-Sánchez B, Bermúdez SE, Aguilar-Tipacamú G, Ballados-González GG, Huerta H, Aguilar-Domínguez M, Mora JD, Licona-Enríquez JD, Mora DD, López-Pérez AM, Torres-Castro MA, Alcántara-Rodríguez V, Becker I, Colunga-Salas P (2021a) Rhipicephalus sanguineus complex in the Americas: Systematic, genetic diversity, and geographic insights. Pathogens 10(9):1118. https://doi.org/10.3390/pathogens10091118
Sánchez-Montes S, Blum-Domínguez S, Lozano-Sardaneta YN, Zazueta-Islas HM, Solís-Cortés M, Ovando-Márquez O, Colunga-Salas P, Tamay-Segovia P, Becker I, Fernández-Figueroa E, Rangel-Escareño C (2021) Molecular detection of Rickettsia sp. cf. Rickettsia monacensis in Ixodes sp. cf. Ixodes affinis collected from white-tailed deer in Campeche, Mexico. Parasitol Res 120(5):1891–1895. https://doi.org/10.1007/s00436-021-07128-5
Saracho-Bottero MN, Venzal JM, Tarragona EL, Thompson CS, Mangold AJ, Beati L, Guglielmone AA, Nava S (2020) The Ixodes ricinus complex (Acari: Ixodidae) in the Southern Cone of America: Ixodes pararicinus, Ixodes aragaoi, and Ixodes sp. cf. I. affinis. Parasitol Res 119(1):43–54. https://doi.org/10.1007/s00436-019-06470-z
Sayler K, Rowland J, Boyce C, Weeks E (2017) Borrelia burgdorferi DNA absent, multiple Rickettsia spp. DNA present in ticks collected from a teaching forest in North Central Florida. Ticks Tick-borne Dis 8(1):53–59. https://doi.org/10.1016/j.ttbdis.2016.09.016
Scott JD, Anderson JF, Durden LA (2012) Widespread dispersal of Borrelia burgdorferi-infected ticks collected from songbirds across Canada. J Parasitol 98(1):49–59. https://doi.org/10.1645/GE-2874.1
Scott JD, Clark KL, Foley JE, Durden LA, Manord JM, Smith ML (2016) First record of Ixodes affinis tick (Acari: Ixodidae) infected with Borrelia burgdorferi sensu lato collected from a migratory songbird in Canada. J Bacteriol Parasitol 7(3):1000281. https://doi.org/10.4172/2155-9597.1000281
Sebastian PS, Flores FS, Saracho-Bottero MN, Tarragona EL, Venzal JM, Nava S (2020) Molecular detection of rickettsial bacteria in ticks of the genus Ixodes from the Southern Cone of America. Acta Trop 210:105588. https://doi.org/10.1016/j.actatropica.2020.105588
Solís-Hernández A, Rodríguez-Vivas RI, Esteve-Gassent MD, Villegas-Pérez SL (2018) Detection de Borrelia burgdorferi sensu lato in dogs and its ticks in rural communities of Yucatán, Mexico. Rev Biol Trop 66(1):428–437
Solís-Hernández A, Rodríguez-Vivas RI, Pérez-Barrera MA, Esteve-Gassent MD, Apanaskevich DA (2015) Ixodes affinis (Acari: Ixodidae) in dogs from rural localities of Yucatán, Mexico: Prevalence, abundance and associated factors. Vet Méx OA 2(3):1–9. https://doi.org/10.21753/vmoa.2.3.351
Sonenshine DE (1991) Biology of ticks, vol I. Old Dominion University, Norfolk, VA, USA, p 447
Sultana H, Patel U, Toliver M, Maggi RG, Neelakanta G (2016) Molecular identification and bioinformatics analysis of a potential anti-vector vaccine candidate, 15-kDa salivary gland protein (Salp15), from Ixodes affinis ticks. Ticks Tick-Borne Dis 7(1):46–53. https://doi.org/10.1016/j.ttbdis.2015.08.003
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123(3):585–595. https://doi.org/10.1093/genetics/123.3.585
Varma RGM (1973) Ticks (Ixodidae) of British Honduras. Trans R Soc Trop Med Hyg 67:92–102. https://doi.org/10.1016/0035-9203(73)90325-8
Wehinger KA, Roelke ME, Greiner EC (1995) Ixodid ticks from panthers and bobcats in Florida. J Wildl Dis 31(4):480–485. https://doi.org/10.7589/0090-3558-31.4.480
Wright CL, Hynes WL, White BT, Marshall MN, Gaff HD, Gauthier DT (2014) Single-tube real-time PCR assay for differentiation of Ixodes affinis and Ixodes scapularis. Ticks Tick-Borne Dis 5(1):48–52. https://doi.org/10.1016/j.ttbdis.2013.08.003
Wilson DE, Reeder DM (2005) Mammal species of the world: a taxonomic and geographic reference, 3 ed. Johns Hopkins University Press, Baltimore. https://doi.org/10.1644/06-MAMM-R-422.1
Xu G, Fang QQ, Keirans JE, Durden LA (2003) Molecular phylogenetic analyses indicate that the Ixodes ricinus complex is a paraphyletic group. J Parasitol 89(3):452–457. https://doi.org/10.1645/0022-3395(2003)089[0452:MPAITT]2.0.CO;2
Yabsley MJ, Nims TN, Savage MY, Durden LA (2009) Ticks and tick-borne pathogens and putative symbionts of black bears (Ursus americanus floridanus) from Georgia and Florida. J Parasitol 95(5):1125–1128. https://doi.org/10.1645/GE-2111.1
Acknowledgements
To the local hunters and veterinarians who kindly donated the biological material.
Funding
Consejo Nacional de Humanidades, Ciencias y Tecnologías CONAHCYT: CF-2023-G-174.
Author information
Authors and Affiliations
Contributions
Roger I. Rodríguez-Vivas: acquisition of data, conception and design of the study, analysis and interpretation of data, drafting the manuscript and final approval of the version to be submitted. Melina M. Ojeda-Chi: acquisition of data, revising it critically for important intellectual content, final approval of the version to be submitted. Alec T. Thompson: acquisition of data, revising it critically for important intellectual content, final approval of the version to be submitted. Michael J. Yabsley: acquisition of data, revising it critically for important intellectual content, final approval of the version to be submitted. Pablo Colunga-Salas: conception and design of the study, analysis and interpretation of data, drafting the manuscript and final approval of the version to be submitted. Sokani Sanchez-Montes: conception and design of the study, analysis and interpretation of data, drafting the manuscript and final approval of the version to be submitted.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rodríguez-Vivas, R.I., Ojeda-Chi, M.M., Thompson, A.T. et al. Population genetics of the Ixodes affinis (Ixodida: Ixodidae) complex in America: new findings and a host-parasite review. Parasitol Res 123, 78 (2024). https://doi.org/10.1007/s00436-023-08091-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-023-08091-z