Abstract
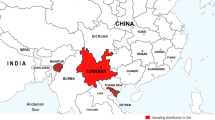
As a result of the experimental infection of rats with metacercariae of Paragonimus heterotremus Chen et Hsia, 1964 from crabs (Potamiscus tannanti) caught in Yen Bai province, Vietnam, it was found that worms migrated into the lungs, to the liver and less frequently to the tissue that lines body cavities of the hosts, where they reached the adult stage, but in the muscles, worms stayed at the larval stage. Studies have shown that for P. heterotremus, rats can simultaneously play the role of the final and paratenic host; herewith, an infection with the trematode of this species can lead to the development of three forms of paragonimiasis: pulmonary, hepatic and muscular. Eggs from the adult worms localised in the liver, unlike eggs from the adult worms localised in the lungs, were not excreted into the external environment, but accumulated inside the organ. Histology and description of changes, which take place on the external surface of organs affected with P. heterotremus, are given in this study. Based on the behavioural characteristics of worms during rat infection and molecular genetic data, we established that worms from Vietnam and India should be assigned to different species of Paragonimus. P. heterotremus distribution is limited to the territory of the Southeast China, Northern Vietnam, Laos and Thailand.





Similar content being viewed by others
References
Adlard RD, Barker SC, Blair D, Cribb TH (1993) Comparison of the second internal transcribed spacer (ribosomal DNA) from populations and species of Fasciolidae (Digenea). Int J Parasitol 23:423–425
Agatsuma T, Arakawa Y, Iwagami M, Honzako Y, Cahyaningsih U, Kang SY, Hong SJ (2000) Molecular evidence of natural hybridization between Fasciola hepatica and F. gigantica. Parasitol Int 49:231–238
Armache J, Jarascha A, Andreas M, Villab E, Beckera T, Bhushana S, Jossinetc F, Habeckd M, Dindara G, Franckenberga S, Marqueza V, Mielkef T, Thommh M, Berninghausena O, Beatrixa B, Södinga J, Westhofc E, Wilsona D, Beckmanna R (2010) Cryo-EM structure and rRNA model of a translating eukaryotic 80S ribosome at 5.5 – a resolution. PNAS 46:748–753
Athokpam VD, Tandon V (2015) A survey of metacercarial infections in commonly edible fish and crab hosts prevailing in Manipur, Northeast India. J Parasit Dis 39:429–440
Bellaousov S, Reuter JS, Seetin MG, Mathews DH (2013) RNAstructure: web servers for RNA secondary structure prediction and analysis. Nucleic Acids Res 41:W471–W474
Besprozvannykh VV (1994) The biology of Paragonimus westermani ichunensis in the Maritime Territory. Experimental research. Med Parazitol 4:28–31 (in Russian)
Besprozvannykh VV (2002) Development of Paragonimus westermani ichunensis in a reservoir host. Parazitologiia 36:427–429 (in Russian)
Besprozvannykh VV (2004) Age-related changes in trematodes Paragonimus westermani ichunensis (Chung, Hsu et Kao, 1978) during development in reservoir and atypical final hosts. In: Materials of the I and II international readings dedicated to the memory and 85th anniversary of the birth of S.S. Shulman. Kaliningrad: KSTU Editore, pp 201-209 (in Russian)
Blair D, Agatsuma T, Watanobe T, Okamoto M, Ito A (1997) Geographical genetic structure within the human lung fluke, Paragonimus westermani, detected from DNA sequences. Parasitology 115:411–417
Blair D, Waikagul J, Honzako Y, Agatsuma T (1998) Phylogenetic relationships among the Thai species of Paragonimus inferred from DNA sequences. In: Tada I, Kojima S, Tsuji M (eds) Proceedings of the 9th International Congress of Parasitology. Bologna: Monduzzi Editore, pp 643–647
Blair D, Xu ZB, Agatsuma T (1999a) Paragonimiasis and the genus Paragonimus. Adv Parasitol 42:113–222
Blair D, Wu B, Chang ZS, Gong X, Agatsuma T, Zhang YN, Chen SH, Lin JX, Chen MG, Waikagul J, Guevara AG, Feng Z, Davis GM (1999b) A molecular perspective on the genera Paragonimus Braun, Euparagonimus Chen and Pagumogonimus Chen. J Helminthol 73:295–299
Blasco-Costa I, Cutmore SC, Miller TL, Nolan MJ (2016) Molecular approaches to trematode systematics: «best practice» and implications for future study. Syst Parasitol 93:295–306
Bowles J, McManus DP (1993) Rapid discrimination of Echinococcus species and strains using a polymerase chain reaction-based RFLP method. Mol Biochem Parasitol 57:231–239
Chang ZS, Wu B, Blair D, Zhang YN, Hu L, Chen SH, Chen MG, Feng Z, Davis GM (2000) Gene sequencing for identification of Paragonimus eggs from a human case. Chin J Parasit Dis 18:213–215
Devi KR, Narain K, Bhattacharya S, Negmu K, Agatsuma T, Blair D, Wickramashinghe S, Mahanta J (2007) Pleuropulmonary paragonimiasis due to Paragonimus heterotremus: molecular diagnosis, prevalence of infection and clinicoradiological features in an endemic area of northeastern India. Trans R Soc Trop Med Hyg 101:786–792
Devi KR, Narain K, Agatsuma T, Blair D, Nagataki M, Wickramasinghe S, Yatawara L, Mahanta J (2010) Morphological and molecular characterization of Paragonimus westermani in northeastern India. Acta Trop 116:31–38
Doanh PN, Dung DT, Thach DTC, Horii Y, Shinohara A, Nawa Y (2011) Human paragonimiasis in Viet Nam: epidemiological survey and identification of the responsible species by DNA sequencing of eggs in patients’ sputum. Parasitol. Int 60:534–537
Doanh PN, Thaenkham U, An PT, Hien HV, Horii Y, Nawa Y (2013) Metacercarial polymorphism and genetic variation of Paragonimus heterotremus (Digenea: Paragonimidae), and a re-appraisal of the taxonomic status of Paragonimus pseudoheterotremus. J Helminthol 89:182–188
Doanh PN, Hien HV, An PT, Tu LA (2015a) Development of lung fluke, Paragonimus heterotremus, in rat and mice, and the role of paratenic host in its life cycle. Tap Chi Sinh Hoc 37:265–271
Doanh PN, Thaenkham U, An PT, Hien HV, Horii Y, Nawa Y (2015b) Metacercarial polymorphism and genetic variation of Paragonimus heterotremus (Digenea: Paragonimidae), and a re-appraisal of the taxonomic status of Paragonimus pseudoheterotremus. J Helminthol 89:182–188
Ghatani S, Shylla JA, Tandon V, Chatterjee A, Roy B (2012) Molecular characterization of pouched amphistome parasites (Trematoda: Gastrothylacidae) using ribosomal ITS2 sequence and secondary structures. J Helminthol 86:117–124
Herrmann KK, Poulin R, Keeney DB, Blasco-Costa I (2014) Genetic structure in a progenetic trematode: signs of cryptic species with contrasting reproductive strategies. Int J Parasitol 44:811–818
Hu W (1998) Studies on the life cycle of Paragonimus heterotremus. Chin J Parasitol Parasit Dis 16:347–352
Huelsenbeck JP, Ronquist F (2001) MrBayes: Bayesian inference of phylogeny. Biometrics 17:754–755
Kasl EL, Fayton TJ, Font WF, Criscione CD (2014) Alloglossidium floridense n. sp. (Digenea: Macroderoididae) from a spring run in North Central Florida. IJP 100:121–126
Kong Y, Doanh P, Nawa Y (2015) Paragonimiasis. In: Xiao L, Ryan U, Feng YY (eds) Biology of foodborne parasites. CRC Press, London, pp 445–462
Kurochkin YV (1987) Trematodes of the fauna of the USSR, Paragonimida. Science, Moscow (In Russian)
Le TH, Van De N, Blair D, McManus DP, Kino H, Agatsuma T (2006) Paragonimus heterotremus Chen and Hsia (1964), in Vietnam: a molecular identification and relationships of isolates from different hosts and geographical origins. Acta Trop 98:25–33
Lockyer AE, Olson PD, Littlewood DTJ (2003) Utility of complete large and small subunit rRNA genes in resolving the phylogeny of the Neodermata (Platyhelminthes): implications and a review of the cercomer theory. Biol J Linn Soc 78:155–171
Mai JC, Coleman AW (1997) The internal transcribed spacer 2 exhibits a common secondary structure in green algae and flowering plants. J Mol Evol 44:258–274
Michot B, Despres L, Bonhomme F, Bachellerie JP (1993) Conserved secondary structures in the ITS2 of trematode prerRNA. FEBS Lett 316:247–252
Miller T, Cribb T (2007) Two new cryptogonimid genera (Digenea, Cryptogonimidae) from Lutjanus bohar (Perciformes, Lutjanidae): analyses of ribosomal DNA reveals wide geographic distribution and presence of cryptic species. Acta Parasitol 52:104–113
Miyazaki I, Habe S (1976) A newly recognized mode of transmission of human infection with lung fluke, Paragonimus westermani (Kerbert, 1878). J Parasitol 62:646–648
Miyazaki I, Terasaki K, Iwata K (1978) Natural infection of muscle of wild boars in Japan by immature Paragonimus westermani (Kerbert, 1878). J Parasitol 64:559–560
Morgan JA, Blair D (1998) Trematode and monogenean rRNA ITS2 secondary structures support a four-domain model. J Mol Evol 47:406–419
Musters W, Boon K, Van der Sande CA, Van Heerikhuizen H, Planta RJ (1990) Functional analysis of transcribed spacers of yeast ribosomal DNA. EMBO J 9:3989–3996
Narain K, Devi KR, Mahanta J (2003) Paragonimus and paragonimiasis - a new focus in Arunachal Pradesh, India. Curr Sci 84:985–987
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, USA
Olson PD, Cribb TH, Tkach VV, Bray RA, Littlewood DTJ (2003) Phylogeny and classification of the Digenea (Platyhelminthes: Trematoda). Int J Parasitol 33:733–755
Park GM, Im K, Yong TS (2003) Phylogenetic relationship of ribosomal ITS2 and mitochondrial CO1 among diploid and triploid Paragonimus westermani isolates. Korean J Parasitol 41:47–55
Pérez-Ponce de León G, Hernández-Mena D (2019) Testing the higher-level phylogenetic classification of Digenea (Platyhelminthes, Trematoda) based on nuclear rDNA sequences before entering the age of the ‘next-generation’ Tree of Life. J Helminthol 93:260–276
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818
Schlotterer C, Hauser MT, Von Haeseler A, Tautz D (1994) Comparative evolutionary analysis of rDNA ITS regions in Drosophila. Mol Biol Evol 11:513–522
Shylla JA, Ghatani S, Tandon V (2013) Utility of divergent domains of 28S ribosomal RNA in species discrimination of paramphistomes (Trematoda: Digenea: Paramphistomoidea). Parasitol Res 112:4239–4253
Singh S, Sugiyama H, Devi KR, Singh LD, Binchai S, Rangsiruji A (2011) Experimental infection with Paragonimus heterotremus metacercariae in laboratory animals in Manipur, India. SE Asian J Trop Med 42:34–38
Singh TS, Sugiyama H, Rangsiruji A (2012) Paragonimus & paragonimiasis in India. Indian J Med Res 136:192–204
Sonnenberg R, Nolte AW, Tautz D (2007) An evaluation of LSU rDNA D1-D2 sequences for their use in species identification. Front Zool 4:6
Sykhanova GI, Kaminsky YV (1998) Paragonimiasis: Typical and larval forms, clinical picture, pathological anatomy, diagnosis, treatment. Vladivostok, Russia
Tacker M, Schuster P (1994) Fast folding and comparison of RNA secondary structures. Monatshefte für Chemie 125:167–188
Tajima F (1983) Evolutionary relationships of DNA sequences in finite populations. Genetics 105:437–467
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol Biol Evol 30:2725–2729
Tkach VV, Littlewood DTJ, Olson PD, Kinsella JM, Swiderski Z (2003) Molecular phylogenetic analysis of the Microphalloidea Ward, 1901 (Trematoda: Digenea). Syst Parasitol 56:1–15
Truett GE, Heeger P, Mynatt RL, Truett AA, Walker JA, Warman ML (2000) Preparation of PCR - quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT). Biotechniques 29:52–54
Van der Sande CAFM, Kwa M, Van Nues RW, Van Heerikhuizen H, Raue HA, Planta RJ (1992) Functional analysis of internal transcribed spacer 2 of Saccharomyces cerevisiae ribosomal DNA. J Mol Biol 223:899–910
Van Nues RW, Rientjes JMJ, Morre SA, Mollee E, Planta RJ, Venema J, Raue HA (1995) Evolutionarily conserved structural elements are critical for processing of internal transcribed spacer 2 from Saccharomyces cerevisiae precursor ribosomal RNA. J Mol Biol 250:24–36
Vilas R, Criscione CD, Blouin MS (2005) A comparison between mitochondrial DNA and the ribosomal internal transcribed regions in prospecting for cryptic species of platyhelminth parasites. Parasitology 131:839–846
Voronova AN, Chelomina GN (2018) Genetic diversity and phylogenetic relations of salmon trematode Nanophyetus japonensis. Parasitol Int 67:267–276
Voronova AN, Chelomina GN, Besprozvannykh VV, Tkach VV (2017) Genetic divergence of human pathogens Nanophyetus spp. (Trematoda: Troglotrematidae) on the opposite sides of the Pacific Rim. Parasitology 144:601–612
Woodruff DS, Merenlender AM, Upatham ES, Viyanant V (1987) Genetic variation and differentiation of three Schistosoma species from the Philippines, Laos, and peninsular Malaysia. Am J Trop Med Hyg 36:345–354
Yan T, Li G, Dong C (1998) Experimental studies on development of Paragonimus heterotremus in rats. Chin J Parasitol Parasit Dis 16:126–129
Zikmundova J, Georgieva S, Faltynkova A, Soldanova M, Kostadinova A (2014) Species diversity of Plagiorchis Luhe, 1899 (Digenea: Plagiorchiidae) in lymnaeid snails from freshwater ecosystems in central Europe revealed by molecules and morphology. Syst Parasitol 88:37–54
Funding
This work was partially supported by the FEB RAS (Far Eastern Branch of the Russian Academy of Sciences) project (18–4–039).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Section Editor: David Bruce Conn
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Voronova, A.N., Besprozvannykh, V.V., Ngo, H.D. et al. Paragonimus heterotremus Chen et Hsia, 1964 (Digenea: Paragonimidae): species identification based on the biological and genetic criteria, and pathology of infection. Parasitol Res 119, 4073–4088 (2020). https://doi.org/10.1007/s00436-020-06929-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06929-4