Abstract
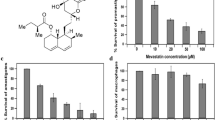
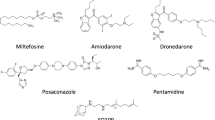
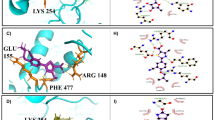
Visceral leishmaniasis (VL, also known as kala-azar) is a vector borne disease caused by obligate intracellular protozoan parasite Leishmania donovani. To overcome the limitations of currently available drugs for VL, molecular target-based study is a promising tool to develop new drugs to treat this neglected tropical disease. One such target we recently identified from L. donovani (Ld) genome (WGS, clinical Indian isolate; BHU 1220, AVPQ01000001) is a small GTP-binding protein, Rab6 protein. We now report a specific inhibitor of the GTPase activity of Rab6 protein of L. donovani (LdRab6) without restricting host enzyme activity. First, to understand the nature of LdRab6 protein, we generated recombinant LdRab6 mutant proteins (rLdRab6) by systematically introducing deletion (two cysteine residues at C-terminal) and mutations [single amino acid substitutions in the conserved region of GTP (Q84L)/GDP(T38N) coding sequence]. The GTPase activity of rLdRab6:GTP and rLdRab6:GDP locked mutant proteins showed ~ 8-fold and ~ 1.5-fold decreases in enzyme activity, respectively, compared to the wild type enzyme activity. The mutant protein rLdRab6:ΔC inhibited the GTPase activity. Sequence alignment analysis of Rab6 protein of L. donovani with Homo sapiens showed identical amino acids in the G conserved region (GTP/GDP-binding sites) but it differed in the C-terminal region. We then evaluated the inhibitory activity of trans-dibenzalacetone (DBA, a synthetic analog of curcumin with strong antileishmanial activity reported earlier by us) in the GTPase activity of LdRab6 protein. Comparative molecular docking analysis of DBA and specific inhibitors of Rab proteins (Lovastatin, BFA, Zoledronate, and NE10790) indicated that DBA had optimum binding affinity with LdRab6 protein. This was further confirmed by the GTPase activity of DBA-treated LdRab6 which showed a basal GTP level significantly lower than that of the wild-type rLdRab6. The results confirm that DBA inhibits the GTPase activity of LdRab6 protein from L. donovani (LdRab6), a potential target for its antileishmanial effect.








Similar content being viewed by others
References
Bhuin T, Roy JK (2014) Rab proteins: the key regulators of intracellular vesicle transport. Exp Cell Res 328:1–19
Chauhan IS, Kaur J, Krishna S, Ghosh A, Singh P, Siddiqi MI, Singh N (2015) Evolutionary comparison of prenylation pathway in kinetoplastid Leishmania and its sister Leptomonas. BMC Evol Biol 15:261
Chauhan IS, Shukla R, Krishna S, Sekhri S, Kaushik U, Baby S, Pal C, Siddiqi MI, Sundar S, Singh N (2016) Recombinant Leishmania Rab6 (rLdRab6) is recognized by sera from visceral leishmaniasis patients. Exp Parasitol 170:135–147
Chauhan IS, Rao GS, Shankar J, Chauhan LKS, Kapadia GJ, Singh N (2018) Chemoprevention of Leishmaniasis: in-vitro antiparasitic activity of dibenzalacetone, a synthetic curcumin analog leads to apoptotic cell death in Leishmania donovani. Parasitol Int 67:627–636
Chauhan IS, Rao GS, Singh N (2019) Enhancing the copy number of Ldrab6 gene in Leishmania donovani parasites mediates drug resistance through drug-thiol conjugate dependent multidrug resistance protein A (MRPA). Acta Trop 199(10518):1–5
Cuif MH, Possmayer F, Zander H, Bordes N, Jollivet F, Couedel-Courteille A, Janoueix-Lerosey I, Langsley G, Bornens M, Goud B (1999) Characterization of GAPCenA, a GTPase activating protein for Rab6, part of which associates with the centrosome. EMBO J 18:1772–1782
Echard A, Jollivet F, Martinez O, Lacapère J-J, Rousselet A, Janoueix-Lerosey I, Goud B (1998) Interaction of a Golgi-associated kinesin-like protein with Rab6. Science 279:580–585
Frech M, Darden T, Pedersen L, Foley C, Charifson P, Anderson M, Wittinghofer A (1994) Role of glutamine-61 in the hydrolysis of GTP by p21H-ras: an experimental and theoretical study. Biochemistry 33:3237–3244
Gabe Lee MT, Mishra A, Lambright DG (2009) Structural mechanisms for regulation of membrane traffic by rab GTPases. Traffic 10:1377–1389
Ghorbani M, Farhoudi R (2018) Leishmaniasis in humans: drug or vaccine therapy? Drug Des, Devel Ther 12:25
Hennies HC, Kornak U, Zhang H, Egerer J, Zhang X, Seifert W, Kühnisch J, Budde B, Nätebus M, Brancati F (2008) Gerodermia osteodysplastica is caused by mutations in SCYL1BP1, a Rab-6 interacting golgin. Nat Genet 40:1410–1412
Indran SV, Britt WJ (2011) A role for the small GTPase Rab6 in assembly of human cytomegalovirus. J Virol 85:5213–5219
Itzen A, Goody RS (2011) GTPases involved in vesicular trafficking: structures and mechanisms. Semin Cell Dev Biol 22:48–56
Jurnak F, Heffron S, Bergmann E (1990) Conformational changes involved in the activation of ras p21: implications for related proteins. Cell 60:525–528
Kapadia GJ, Soares IAO, Rao GS, Badoco FR, Furtado RA, Correa MB, Tavares DC, Cunha WR, Magalhaes LG (2017) Antiparasitic activity of menadione (Vitamin K3) against Schistosoma mansoni in BABL/c mice. Acta Trop 147:163–173
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291
Li G (2011) Rab GTPases, membrane trafficking and diseases. Curr Drug Targets 12:1188–1193
Li B, Warner JR (1996) Mutation of the Rab6 homologue of Saccharomyces cerevisiae, YPT6, inhibits both early Golgi function and ribosome biosynthesis. J Biol Chem 271:16813–16819
Magalhaes LG, Rao GS, Soares IAO, Badocco FR, Cunha WR, Rodrigues V, Kapadia GJ (2014) Chemoprevention of Schistosomiasis: in vitro antiparasitic activity of nineteen plant-derived and synthetic simple naphthoquinones and naphthols against Schistosoma mansoni adult worms. J Trop Dis 2:146–153
Magalhaes, L.G., Rao, G.S. et al., 2020. Phytotherapy of tropical parasitic diseases: in vitro antiparasitic activity of synthetic curcumin monoketone analogs of dibenzalacetone and its methoxy derivatives against Trypanosoma cruzi and Leishmania donovani. (Manuscript in preparation)
Martinez O, Schmidt A, Salaméro J, Hoflack B, Roa M, Goud B (1994) The small GTP-binding protein rab6 functions in intra-Golgi transport. J Cell Biol 127:1575–1588
Miserey-Lenkei S, Waharte F, Boulet A, Cuif MH, Tenza D, El Marjou A, Raposo G, Salamero J, Héliot L, Goud B (2007) Rab6-interacting protein 1 links Rab6 and Rab11 function. Traffic 8:1385–1403
Nuoffer C, Davidson HW, Matteson J, Meinkoth J, Balch WE (1994) A GDP-bound of rab1 inhibits protein export from the endoplasmic reticulum and transport between Golgi compartments. J Cell Biol 125:225–237
Okwor I, Uzonna J (2016) Social and economic burden of human leishmaniasis. The Am J Trop Med Hyg 94:489–493
Park H (2013) Structural basis of membrane trafficking by Rab family small g protein. Int J Mol Sci 14:8912–8923
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF chimera a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612
Pylypenko O, Hammich H, Yu I-M, Houdusse A (2018) Rab GTPases and their interacting protein partners: structural insights into Rab functional diversity. Small GTPases 9:22–48
Rao GS, Tokuda H, Ichiishi E, Takasaki M, Iida A, Suzuki S, Konoshima T, Kapadia GJ (2013) Oral chemoprevention of skin cancer in mice by benzophenone sunscreens dioxybenzone and octabenzone in drinking water. Anticancer Res 33:2535–2540
Riederer MA, Soldati T, Shapiro AD, Lin J, Pfeffer SR (1994) Lysosome biogenesis requires Rab9 function and receptor recycling from endosomes to the trans482 Golgi network. J Cell Biol 125:573–582
Šali A, Blundell TL (1993) Comparative protein modelling by satisfaction of spatial restraints. J Mol Biol 234:779–815
Seabra MC, Mules EH, Hume AN (2002) Rab GTPases, intracellular traffic and disease. Trends Mol Med 8:23–30
Shetty KM, Kurada P, O’Tousa JE (1998) Rab6 regulation of rhodopsin transport in Drosophila. J Biol Chem 273:20425–20430
Short B, Preisinger C, Schaletzky J, Kopajtich R, Barr FA (2002) The Rab6 GTPase regulates recruitment of the dynactin complex to Golgi membranes. Curr Biol 12:1792–1795
Singh N, Chauhan IS (2018) MicroRNA expression profiling of dibenzalacetone (DBA)-treated amastigotes of Leishmania donovani. Exp Parasitol 193:5–19
Stenmark H (2009) Rab GTPases as coordinators of vesicle traffic. Nat Rev Mol Cell Biol 10:513–525
Sundar S, Rai M (2002) Laboratory diagnosis of visceral leishmaniasis. Clin Diagn Lab Immunol 9:951–958
Thaman LA, Deckner GE, Sottery JP (1996) Photoprotection compositions having improved efficiency. US Patent 5:516,508
Tisdale EJ, Bourne JR, Khosravi-Far R, Der CJ, Balch W (1992) GTP binding mutants of rab1 and rab2 are potent inhibitors of vesicular transport from the endoplasmic reticulum to the Golgi complex. J Cell Biol 119:749–761
White J, Johannes L, Mallard F, Girod A, Grill S, Reinsch S, Keller P, Tzschaschel B, Echard A, Goud B (1999) Rab6 coordinates a novel Golgi to ER retrograde transport pathway in live cells. J Cell Biol 147:743–760
Young J, Stauber T, del Nery E, Vernos I, Pepperkok R, Nilsson T (2005) Regulation of microtubule-dependent recycling at the trans-Golgi network by Rab6A and Rab6A. Mol Biol Cell 16:162–177
Zerial M, McBride H (2001) Rab proteins as membrane organizers. Nat Rev Mol Cell Biol 2:107–117
Zhao Y, Gui W, Niu F, Chong S (2019) The MAPK signaling pathways as a novel way in regulation and treatment of parasitic diseases. Diseases 7(1):E9
Funding
This work was supported by the Council of Scientific & Industrial Research (CSIR), India (grant number BSC-0114). Indira Singh Chauhan was supported by fellowships from Indian Council of Medical Research (ICMR). This is CDRI manuscript number 51/2019/NS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Sarah Hendrickx
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chauhan, I.S., Marwa, S., Rao, G.S. et al. Antiparasitic dibenzalacetone inhibits the GTPase activity of Rab6 protein of Leishmania donovani (LdRab6), a potential target for its antileishmanial effect. Parasitol Res 119, 2991–3003 (2020). https://doi.org/10.1007/s00436-020-06810-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06810-4