Abstract
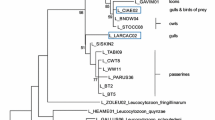
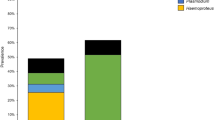
Hemosporidian parasites rarely infect aquatic birds. Few studies have been conducted in South America identifying some lineages of the genera Plasmodium, Leucocytozoon, and Haemoproteus, but none has been done in the Neotropic cormorant (Phalacrocorax brasilianus). This species is widely distributed through the American continent, from Southern USA to Tierra del Fuego, using a wide range of aquatic habitats. We molecularly studied the occurrence and diversity of hemosporidian lineages infecting individuals of Neotropic cormorant across a broad latitudinal gradient in Chile (Arica to Tierra del Fuego). As expected, a very low occurrence of individuals infected by Plasmodium sp. (4/123, 3.3%) and Leucocytozoon sp. (2/123, 1.6%) was detected. We found no evidence of Haemoproteus sp. We identified one lineage of Plasmodium (ZEMAC01) and one new lineage of Leucocytozoon (PHABRA01) infecting cormorants. Individuals infected by Plasmodium sp. were birds from only one site (i.e., Chillán), whereas Leucocytozoon sp. was found in one bird from Valdivia and another one from Tierra del Fuego. Our results expand the known range of hemosporidian parasite lineages in aquatic birds providing an essential baseline data that contribute to a better understanding of the geographic range of hemosporidian parasites infecting Phalacrocoracidae in South America.


Similar content being viewed by others
References
Adlard RA, Peirce MA, Lederer R (2004) Blood parasites of birds from south-east Queensland. EMU 104:191–196
Adler PH, Currie DC, Wood DM (2004) The black flies (Simuliidae) of North America. Cornell University Press, New York
Apanius V (1998) Stress and immune response. In: Møller AP, Milinski M, Slater PJB (eds) Stress and behavior. Academic Press, New York, pp 133–154
Arriero E, Møller AP (2008) Host ecology and life-history traits associated with blood parasite species richness in birds. J Evol Biol 21:1504–1513
Asghar M, Hasselquist D, Hansson B, Zehtindjiev P, Westerdahl H, Bensch S (2015) Hidden costs of infection: chronic malaria accelerates telomere degradation and senescence in wild birds. Science 347:436–438
Atkinson CT (2008) Haemoproteus. In: Atkinson CT, Thomas NJ, Hunter BC (eds) Parasitic diseases of wild birds. Wiley-Blackwell, Ames, pp 13–35
Atkinson CT, Dusek RJ, Woods KL, Iko WM (2000) Pathogenicity of avian malaria in experimentally-infected Hawaii Amakihi. J Wildl Dis 36:197–204
Barquete V, Bugoni L, Vooren CM (2008) Diet of Neotropic cormorant (Phalacrocorax brasilianus) in an estuarine environment. Mar Biol 153:431–443
Bastien M, Jaeger A, Le Corre M, Tortosa P, Lebarbenchon C (2014) Haemoproteus iwa in great frigatebirds (Fregata minor) in the islands of the Western Indian Ocean. PLoS One 9:e97185
Beadell JS, Gering E, Austin J, Dumbacher JP, Peirce MA, Pratt TK, Atkinson CT, Fleischer RC (2004) Prevalence and differential host-specificity of two avian blood parasite genera in the Australo-Papuan region. Mol Ecol 13:3829–3844
Bennett GF, Bishop MA, Peirce MA (1993) Checklist of the avian species of Plasmodium Marchiafava and Celli, 1885 (Apicomplexa) and their distribution by avian family and Wallacean life zones. Syst Parasitol 26:171–179
Bennett GF, Pierce MA, Earlé RA (1994) An annotated checklist of the valid avian species of Haemoproteus, Leucocytozoon (Apicomplexa: Haemosporida) and Hepatozoon (Apicomplexa: Haemogredgarinidae). Syst Parasitol 29:61–73
Bensch S, Stjernman M, Hasselquist D, Ostman O, Hannson B, Westerdahl H, Pinheiro RT (2000) Host specificity in avian blood parasites: a study of Plasmodium and Haemoproteus mitochondrial DNA amplified from birds. Proc Biol Sci 267:1583–1589
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Resour 9:1353–1358
BirdLife International (2016) Phalacrocorax brasilianus. The IUCN Red List of Threatened Species 2016: e.T22696773A93585558
Campioni L, Martínez-de la Puente J, Figuerola J, Granadeiro JP, Silva MC, Catry P (2018) Absence of haemosporidian parasite infections in the long-lived Cory’s shearwater: evidence from molecular analyses and review of the literature. Parasitol Res 117:323–329
Casaux RJ, di Prinzio CY, Bertolin ML, Tartara MA (2009) Diet of the Neotropic cormorant Phalacrocorax olivaceus at West Chubut, Patagonia, Argentina. Waterbirds 32:444–449
Cazorla CG, Spinelli GR (2007) A new species of Patagonian Stilobezzia (Acanthohelea) and a redescription of S. (A.) nigerrima Ingram and Macfie (Diptera: Ceratopogonidae). Trans Am Entomol Soc 133:181–187
Cereceda P, Larrain H, Osses P, Farías M, Egaña I (2008) The climate of the coast and fog zone in the Tarapacá Region, Atacama Desert, Chile. Atmos Res 87:301–311
Chagas CR, Guimarães LO, Monteiro EF et al (2016) Hemosporidian parasites of free-living birds in the São Paulo Zoo, Brazil. Parasitol Res 115:1443–1452
Clark NJ, Clegg SM, Lima MR (2014) A review of global diversity in avian haemosporidians (Plasmodium and Haemoproteus: Haemosporida): new insights from molecular data. Int J Parasitol 44:329–338
Coscarón S, Coscarón-Arias CL (1995) Distribution of Neotropical Simuliidae (Insecta-Diptera) and its areas of endemism. Rev Acad Colomb Cienc 19:717–732
Currie DC, Adler PH (2008) Global diversity of black flies (Diptera: Simuliidae) in freshwater. Hydrobiol 595:469–475
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772–772
Dawson RD, Bortolotti GR (2000) Effects of hematozoan parasites on condition and return rates of American kestrels. Auk 117:373–380
Engström H, Dufva R, Olsson G (2000) Absence of haematozoa and ectoparasites in a highly sexually ornamented species, the crested auklet. Waterbirds 23:486–488
Ferraguti M, Martínez-de la Puente J, Muñoz J, Roiz D, Ruiz S, Soriguer R, Figuerola J (2013) Avian Plasmodium in Culex and Ochlerotatus mosquitoes from southern Spain: effects of season and host-feeding source on parasite dynamics. PLoS One 8:e66238
Figuerola J (1999) Effects of salinity on rates of infestation of waterbirds by haematozoa. Ecography 22:681–685
Forrester DJ, Greiner EC, McFarlane RW (1977) Blood parasites of some columbiform and passeriform birds from Chile. J Wildl Dis 13:94–96
Forrester DJ, Foster GW, Morrison JL (2001) Leucocytozoon toddi and Haemoproteus tinnunculi (Protozoa: Haemosporina) in the Chimango caracara (Milvago chimango) in southern Chile. Mem Inst Osvaldo Cruz 96:1023–1024
Frederiksen M, Lebreton J, Bregnballe T (2001) The interplay between culling and density-dependence in the great cormorant: a modeling approach. J Appl Ecol 38:617–627
Fuentes-Castillo D, Cicchino A, Mironov S, Moreno L, Landaeta-Aqueveque C, Barrientos C, González-Acuña D (2016) Ectoparasites of the black-chinned siskin Spinus barbatus (Passeriformes: Fringillidae) in Chile. Rev Bras Parasitol Vet 25:476–483
Garamszegi LZ (2010) The sensitivity of microscopy and PCR-based detection methods affecting estimates of prevalence of blood parasites in birds. J Parasitol 96:1197–1203
González CR, Jercic MI, Muñoz L (2005) The mosquitoes from Chile (Diptera: Culicidae). Acta Ent Chilena 29:31–35
González-Acuña D, Guglielmone A (2005) Ticks (Acari: Ixodoidea: Argasidae, Ixodidae) of Chile. Exp Appl Acarol 35:147–163
Ham-Dueñas JG, Chapa-Vargas L, Stracey CM, Huber-Sannwald (2017) Haemosporidian prevalence and parasitaemia in the black-throated sparrow (Amphispiza bilineata) in central-Mexican dryland habitats. Parasitol Res 116:2527–2537
Henry A (1993) Simuliidae de Chile: Lista de especies y su distribución geográfica (Díptera: Simuliidae). Acta Ent Chilena 18:89–96
Inumaru M, Murata K, Sato Y (2017) Prevalence of avian haemosporidian among injured wild birds in Tokyo and environs, Japan. Int J Parasitol Parasites Wildl 6:299–309
Ishtiaq F, Gering E, Rappole JH, Rahmani AR, Jhala YV, Dove CJ, Milensky C, Olson SL, Peirce MA, Fleischer RC (2007) Prevalence and diversity of avian hematozoan parasites in Asia: a regional survey. J Wildl Dis 43:382–398
Jiménez E (2001) Biología reproductiva y alimentaria del cormorán yeco (Phalacrocorax brasilianus, Gmelin; 1789) en la colonia del Santuario de la Naturaleza del Ríos Cruces, Valdivia. Dissertation, Universidad Austral de Chile
Jovani R, Tella JL, Forero MG, Bertellotti M, Blanco G, Ceballos O, Donázar JA (2001) Apparent absence of blood parasites in the Patagonian seabird community: is it related to marine environment? Waterbirds 24:430–433
Kalmbach E, Ramsay SC, Wendeln H, Becker PH (2001) A study of Neotropic cormorants in Central Chile: possible effects of El Nino. Waterbirds 24:345–351
Knowles SCL, Wood MJ, Sheldon BC (2010) Context-dependent effects of parental effort on malaria infection in a wild bird population, and their role in reproductive trade-offs. Oecologia 164:87–97
LaPointe DA, Goff ML, Atkinson CT (2005) Comparative susceptibility of introduced forest-dwelling mosquitoes in Hawaii to avian malaria, Plasmodium relictum. J Parasitol 91:843–849
Lee-Cruz L, Cunningham A, Martínez P, Cruz M, Goodman S, Hamer K (2016) Prevalence of Haemoproteus sp. in Galápagos blue-footed boobies: effects on health and reproduction. Parasitol Open 2:1–10. https://doi.org/10.1017/pao.2015.6
Levin II, Parker PG (2013) Comparative host–parasite population genetic structures: obligate fly ectoparasites on Galapagos seabirds. Parasitology 140:1061–1010
Levin II, Valkiunas G, Santiago-Alarcon D, Cruz LL, Iezhova TA et al (2011) Hippoboscid-transmitted Haemoproteus parasites (Haemosporida) infect Galapagos Pelecaniform birds: evidence from molecular and morphological studies, with a description of Haemoproteus iwa. Int J Parasitol 41:1019–1027
Levin II, Adkesson MJ, Evans M, Rettke CK, Parker PG (2014) No evidence for Galapagos Plasmodium lineage arriving via Humboldt Current seabirds. Pac Conserv Biol 20:37–40
Mackerras MJ, Mackerras IM (1960) The haematozoa of Australian birds. Aust J Zool 8:226–260
Manimegalai K, Sukanya S (2014) Biology of the filarial vector, Culex quinquefasciatus (Diptera: Culicidae). Int J Curr Microbiol App Sci 3:718–724
Martínez J, Martínez-de la Puente J, Herrero J, del Cerro S, Lobato E, Rivero-de Aguilar J, Vásquez RA, Merino S (2009) A restriction site to differentiate Plasmodium and Haemoproteus infections in birds: on the inefficiency of general primers for detection of mixed infections. Parasitology 136:713–722
Martínez J, Vásquez RA, Venegas C, Merino S (2015) Molecular characterisation of haemoparasites in forest birds from Robison Crusoe Island: is the austral trush a potential threat to endemic birds? Bird Conserv Int 25:139–152
Martínez-Abrain A, Esparza B, Oro D (2004) Lack of blood parasites in bird species: does absence of blood parasite vectors explain it all? Ardeola 5:225–232
Marzal A (2012) Recent advances in studies on avian malaria parasites. In: Okwa O (ed) Malaria parasites. InTech Open, London, pp 135–158
Mendes L, Piersma T, Lecoq M, Spaans B, Ricklefs RE (2005) Disease-limited distributions? Contrasts in the prevalence of avian malaria in shorebird species using marine and freshwater habitats. Oikos 109:396–404
Merino S, Minguez E (1998) Absence of hematozoa in a breeding colony of the storm petrel Hydrobates pelagicus. IBIS 140:180–181
Merino S, Potti J (1995) High prevalence of hematozoa in nestlings of a passerine species, the pied flycatcher (Ficedula hypoleuca). Auk 112:1041–1043
Merino S, Barbosa A, Moreno J, Potti J (1997) Absence of haematozoa in a wild chinstrap penguin Pygoscelis Antarctica population. Polar Biol 18:227–228
Merino S, Moreno J, Sanz JJ, Arriero E (2000) Are avian blood parasites pathogenic in the wild? A medication experiment in blue tits (Parus caeruleus). Proc Biol Sci 267:2507–2510
Merino S, Moreno J, Vásquez RA, Martínez J, Sánchez-Monsálvez I, Estades CF, Ippi S, Sabat P, Rozzi R, McGehee S (2008) Haematozoa in forest birds from southern Chile: latitudinal gradients in prevalence and parasite lineage richness. Austral Ecol 33:329–340
Merino S, Hennicke J, Martínez J, Ludynia K, Torres R, Work TM, Stroud S, Masello JF, Quillfeldt P (2012) Infection by Haemoproteus parasites in four species of frigatebirds and the description of a new species of Haemoproteus (Haemosporida: Haemoproteidae). J Parasitol 98:388–397
Moens MAJ, Valkiunas G, Paca A, Bonaccorso E, Aguirre N, Pérez-Tris J (2016) Parasite specialization in a unique habitat: hummingbirds as reservoirs of generalist blood parasites of Andean birds. J Anim Ecol 85:1234–1245
Outlaw DC, Ricklefs RE (2011) Rerooting the evolutionary tree of malaria parasites. PNAS 108:13183–13187
Padilla LR, Whiteman NK, Merkel J, Huyvaert KP, Parker PG (2006) Health assessment of seabirds on Isla Genovesa, Galápagos Island. Ornithol Monogr 60:86–97
Parsons NJ, Peirce MA, Strauss V (2010) New species of haematozoa in Phalacrocoracidae and Stercorariidae in South Africa. Ostrich 81:103–108
Parsons NJ, Voogt NM, Schaefer AM, Peirce MA, Vanstreels RET (2017) Occurrence of blood parasites in seabirds for rehabilitation in the Western Cape, South Africa, 2001–2013. Vet Parasitol 233:52–61
Peirce MA (2005) A checklist of the valid avian species of Babesia (Apicomplexa: Piroplasmorida), Haemoproteus, Leucocytozoon (Apicomplexa: Haemosporida), and Hepatozoon (Apicomplexa: Haemogregarinidae). J Nat Hist 39:3621–3632
Peirce MA, Brooke M (1993) Failure to detect blood parasites in seabirds from the Pitcairn Islands. Seabird 15:72–74
Pérez-Rodríguez A, Ramírez Á, Richardson DS, Pérez-Tris J (2013) Evolution of parasite island syndromes without long-term host population isolation: parasite dynamics in Macaronesian blackcaps Sylvia atricapilla. Glob Ecol Biogeogr 22:1272–1281
Perkins SL, Schall J (2002) A molecular phylogeny of malarial parasites recovered from cytochrome b gene sequences. J Parasitol 88:972–978
Piersma T (1997) Do global patterns of habitat use and migration strategies co-evolve with relative investment in immunocompetence due to spatial variation in parasites pressure? Oikos 80:623–631
Quillfeldt P, Martínez J, Hennicke J, Ludynia K, Gladbach A, Masello JF, Riou S, Merino S (2010) Hemosporidian blood parasites in seabirds- a comparative genetic study of species from Antarctic to tropical habitats. Naturwissenschaften 97:809–817
Quillfeldt P, Arriero E, Martínez J, Masello JF, Merino S (2011) Prevalence of blood parasites in seabirds – a review. Front Zool 8:26
Quillfeldt P, Martínez J, Bugoni L, Mancini P, Merino S (2014) Blood parasites in noddies and boobies from Brazilian offshore islands – differences between species and influence of nesting habitat. Parasitology 141:399–410
Quintana F, Yorio P, Borboroglu PG (2002) Aspects of the breeding biology of the Neotropic cormorant Phalacrocorax olivaceus at Golfo San Jorge, Argentina. Mar Ornithol 30:25–29
Ricklefs RE, Fallon SM, Bermingham E (2004) Evolutionary relationships, cospeciation, and host switching in avian malaria parasites. Syst Biol 53:111–119
Rodrigues P, Campos E, Micael J, Verdugo C (in press) Sex determination of Neotropic cormorant (Phalacrocorax brasilianus) by molecular sexing. Avian Biol Res
Ronquist F, Teslenko M, Van Der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, M a S, Huelsenbeck JP (2012) Mrbayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Sallaberry-Pincheira N, Gonzalez-Acuña D, Herrera-Tello Y, Dantas GPM, Luna-Jorquera G, Frere E, Valdés-Velasquez A, Simeone A, Vianna J (2015) Molecular epidemiology of avian malaria in wild breeding colonies of Humboldt and Magellanic penguins in South America. EcoHealth 12:267–277
Seimon TA, Gilbert M, Neabore S, Hollinger C, Tomaszewicz A, Newton A, Chang T, McAloose D (2016) Avian haemosporidian parasite lineages in four species of free-ranging migratory waterbirds from Mongolia, 2008. J Wildl Dis 52:682–687
Sepulveda MS, Palma RL, Ochoa-Acuna H (1997) New records of feather lice from some seabirds in Chile. J Wildl Dis 33:371–372
Soares L, Escudero G, Penha V, Ricklefs RE (2016) Low prevalence of haemosporidian parasites in shorebirds. Ardea 104:129–141
Stotz DF, Fitzpatrick J, Parker T, Moskovits D (1996) Neotropical birds. In: Ecology and conservation. The University of Chicago Press, Chicago
Torres P, Ruiz E, Gesche W, Montefusco A (1991) Gastrointestinal helminths of fish-eating birds from Chiloe Island, Chile. J Wildl Dis 27:178–179
Torres P, Schlatter R, Montefusco A, Gesche W, Ruiz E, Contreras A (1993) Helminth parasites of piscivorous birds from lakes in the south of Chile. Mem Inst Oswaldo Cruz 88:341–343
Torres P, Valdivieso J, Schlatter R, Montefusco A, Revenga J, Marín F, Lamilla J, Ramallo G (2000) Infection by Contracaecum rudolphii (Nematoda: Anisakidae) in the Neotropic cormorant Phalacrocorax brasilianus, and fishes from the estuary of the Valdivia river, Chile. Stud Neotropical Fauna Environ 35:101–108
Torres P, Ortega J, Schlatter R (2005) Nematode parasites of the digestive tract in Neotropic cormorant chicks (Phalacrocorax brasilianus) from the River Cruces Ramsar site in southern Chile. Parasitol Res 97:103–107
Valera F, Carrillo CM, Barbosa A, Moreno E (2003) Low prevalence of haematozoa in trumpeter finches Bucanetes githagineus from South-Eastern Spain: additional support for a restricted distribution of blood parasites in arid lands. J Arid Environ 55:209–213
Valkiunas G (2005) Avian malaria parasites and other haemosporidia. CRC Press, Boca Raton
Verdugo C, Pinto A, Ariyama N, Moroni M, Hernandez C (2019) Molecular identification of avian viruses in Neotropic cormorants (Phalacrocorax brasilianus). J Wildl Dis 51. doi: https://doi.org/10.7589/2017-10-256
Waldenström J, Bensch S, Kiboi S, Hasselquist D, Ottosson U (2002) Cross-species infection of blood parasites between resident and migratory songbirds in Africa. Mol Ecol 11:1545–1554
Waldenström J, Bensch S, Hasselquist D, Ostman O (2004) A new nested polymerase chain reaction method very efficient in detecting Plasmodium and Haemoproteus infections from avian blood. J Parasitol 90:191–194
Whiteman NK, Sánchez P, Merkel J, Klompen H, Parker PG (2006) Cryptic host specificity of an avian skin mite (Epidermoptidae) vectored by louse flies (Hippoboscidae) associated with two endemic Galápagos bird species. J Parasitol 92:1218–1228
Yorio P, Quintana F, Campagna C, Harris G (1994) Diversidad, abundancia y dinámica espacio-temporal de la colonia mixta de aves marinas en Punta León, Patagonia. Ornitol Neotrop 5:69–77
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1024
Acknowledgments
P. R. was supported by Fondo Nacional de Desarrollo Científico y Tecnológico (FONDECYT 3150617) and C. V. was supported by Fondo Nacional de Desarrollo Científico y Tecnológico (FONDECYT 11130305). We also thank the Wildlife Conservation Society, José Luis Bastias, Edgar Carcamo, Omar Jimenez, Nicolás Martin, Joana Micael, Miguel Millon, Rodrigo Munzenmayer, Felipe Pontigo, Roberto Rosas, and Pedro Alvarez for field and laboratory support. We thank two anonymous reviewers whose comments and suggestions helped improve this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The present study was conducted according to the animal welfare guidelines of the Bioethical Committee of the Universidad Austral de Chile and with permission 7597/2016 from the Servicio Agrícola y Ganadero (SAG) and permission 019/2015 from the Corporación Nacional Forestal (CONAF). All experiments comply with the current laws of the Republic of Chile.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Larissa Howe
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Pedro, R., Claudio, N., Elena, C. et al. Low occurrence of hemosporidian parasites in the Neotropic cormorant (Phalacrocorax brasilianus) in Chile. Parasitol Res 118, 325–333 (2019). https://doi.org/10.1007/s00436-018-6146-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-6146-6