Abstract
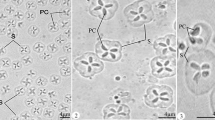
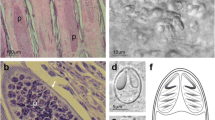
Myxobolus miyairii Kudo, 1919 was first reported from the intestines of the Amur catfish (Silurus asotus) in Japan and then in China and Russia, but with incomplete description. During the investigation of fish myxosporean diversity in Poyang Lake, the biggest lake along the Yangtze River, China, two Amur catfish highly infected with M. miyairii in the intestine wall were sampled. So, the complete description of this species with morphological and molecular data was presented here. A large number of whitish, round or ellipsoidal pseudocysts 0.32–0.78 mm in diameter could be found in the external intestinal wall after dissecting the infected fish. Mature spores of M. miyairii were elongated and ellipsoidal in the frontal view and narrow fusiform in the lateral view, with a slightly pointed anterior end and a bluntly rounded posterior end and measured 13.3 ± 0.49 (12.5–14.7) μm × 6.6 ± 0.27 (6.2–7.4) μm × 5.0 ± 0.26 (4.4–5.7) μm in size. Spore surface was smooth and two spore valves symmetrical, with a thin and straight sutural ridge. Interestingly, two types of caudal appendage (single or bifurcated) were occasionally present on the posterior end of some spores which has not previously been reported. The two equal pyriform polar capsules measured 6.5 ± 0.30 (6.2–7.5) μm long and 1.9 ± 0.14 (1.5–2.3) μm wide and situated at the anterior end of the spore. Polar filaments coiled with eight to nine turns, perpendicularly to the longitudinal axis of the polar capsules. Histopathological analysis showed that the plasmodium developed in the circular muscle layer of intestinal wall of Amur catfish, but no obvious inflammatory responses were observed. Phylogenetic analysis based on the partial 18S small subunit ribosomal DNA sequences indicates that M. miyairii cluster within a clade of Siluriforme-infecting Henneguya species with the support of a high bootstrap value, but also evolutionarily independent from the Henneguya clade infecting the epithelium of fish of the Ictaluridae family. Additionally, Myxobolus species reported with caudal processes dispersed within the Henneguya-Myxobolus clade.








Similar content being viewed by others
References
Atkinson S, Bartošová P, Whipps CM & Bartholomew JL (2015) Approaches for characterizing myxozoan species in myxozoan evolution, ecology and development, edited by Okamura B, Gruhl A, Bartholomew JL. Springer, ISBN 978–3–319–14753–6 (eBook)
Bahri S (2008) Abnormal forms of Myxobolus bizerti and Myxobolus mülleri (Myxosporea: Bivalvulida) spores with caudal appendages. Bull Eur Assoc Fish Pathol 28(6):252–255
Barta JR, Martin DS, Liberator PA, Dashkevica M, Anderson JW, Feighner SD, Elbrecht A, Perkins-Barrow A, Jenkins MC, Danforth HD, Ruff MD, Profous-Juchelka H (1997) Phylogenetic relationships among eight Eimeria species infecting domestic fowl inferred using complete small subunit ribosomal DNA sequences. J Parasitol 83:262–271
Baska F, Voronin VN, Eszterbauer E, Muller L, Marton S, Molnár K (2009) Occurrence of two myxosporean species, Myxobolus hakyi sp. n. and Hoferellus pulvinatus sp. n., in Pangasianodon hypophthalmus fry imported from Thailand to Europe as ornamental fish. Parasitol Res 105(5):1391–1398
Carriero MM, Adriano EA, Silva MRM, Ceccarelli PS, Maia AAM (2013) Molecular phylogeny of the Myxobolus and Henneguya genera with several new south American species. Plos One 8:e73713
Chen QL, Ma CL (1998) Myxozoa: Myxosporea. Science, Beijing ( In Chinese)
Donec ZS, Shulman SS (1984) Parasitic Protozoa. Phylum Cnidosporidia. In: Bauer ON (ed) Key to parasites of freshwater fishes of the USSR, vol 1. Nauka, Leningrad, pp 88–251, In Russian
Eiras JC, Molnár K, Lu YS (2005) Synopsis of the species of Myxobolus Bütschli, 1882 (Myxozoa: Myxosporea: Myxobolidae). Syst Parasitol 61(1):1–46
Eiras JC, Zhang JY, Molnár K (2014) Synopsis of the species of Myxobolus Butschli, 1882 (Myxozoa: Myxosporea, Myxobolidae) described between 2005 and 2013. Syst Parasitol 88(1):11–36
Eszterbauer E (2004) Genetic relationship among gill-infecting Myxobolus species (Myxosporea) of cyprinids: Molecular evidence of importance of tissue-specificity. Dis Aquat Org 58:35–40
Evans NM, Holder MT, Barbeitos MS, Okamura B, Cartwright P (2010) The phylogenetic position of Myxozoa: exploring conflicting signals in phylogenomic and ribosomal data sets. Mol Biol Evol 27:2733–2746
Fiala I (2006) The phylogeny of Myxosporea (Myxozoa) based on small subunit ribosomal RNA gene analysis. Int J Parasitol 36(14):1521–1534
Fiala I, Bartošová P (2010) History of myxozoan character evolution on the basis of rDNA and EF-2 DATA. BMC Evol Biol 10:228
Fiala I, Bartošová P, Whipps CM (2015a) Classification and phylogenetics of myxozoa in Myxozoan Evolution, Ecology and Development, edited by Okamura B, Gruhl A, Bartholomew JL. Springer, ISBN 978–3–319–14753–6 (eBook)
Fiala I, Bartošová P, Okamura B, Hartikainen H (2015b) Adaptive radiation and evolution within the myxozoa in myxozoan evolution, ecology and development, edited by Okamura B, Gruhl A, Bartholomew JL. Springer, ISBN 978–3–319–14753–6 (eBook)
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum–likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59(3):307–321
Hall TA (1999) BioEdit: a user–friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Sym Ser 41:95–98
Kent ML, Andree KB, Bartholomew JL, El-Matbouli M, Desser SS, Devlin RH, Feist SW, Hedrick RP, Hoffmann RW, Khattra J, Hallett SL, Lester RJG, Longshaw M, Palenzeula O, Siddall ME, Xiao C (2001) Recent advances in our knowledge of the Myxozoa. J Eukaryot Microbiol 48:395–413
Kudo R (1919) Illinois biological monographs. 5: p155
Liu Y, Whipps CM, Gu ZM, Zeng LB (2010) Myxobolus turpisrotundus (Myxosporea: Bivalvulida) spores with caudal appendages: investigating the validity of the genus Henneguya with morphological and molecular evidence. Parasitol Res 107:699–706
Liu Y, Whipps CM, Gu ZM, Huang MJ, He C, Yang HL, Molnár K (2013) Myxobolus musseliusae (Myxozoa: Myxobolidae) from the gills of common carp Cyprinus carpio and revision of Myxobolus dispar recorded in China. Parasitol Res 112:289–296
Liu Y, Whipps CM, Nie P, Gu ZM (2014) Myxobolus oralis sp. n. (Myxosporea: Bivalvulida) infecting the palate in the mouth of gibel carp Carassius auratus gibelio (Cypriniformes: Cyprinidae). Folia Parasitol 61:505–511
Lom J, Arthur JR (1989) A guideline for preparation of species descriptions in Myxosporea. J Fish Dis 12:151–156
Lom J, Dyková I (2006) Myxozoan genera: definition and notes on taxonomy, life-cycle terminology and pathogenic species. Folia Parasitol 53:1–36
Molnár K (2002) Site preference of fish myxosporeans in the gill. Dis Aquat Org 48:197–207
Moreira GSA, Adriano EA, Silva MRM, Ceccarelli PS, Maia AAM (2014) The morphological and molecular characterization of Henneguya rotunda n. sp., a parasite of the gill arch and fins of Salminus brasiliensis from the Mogi Guaçu River, Brazil. Parasitol Res 113:1703–1711
Page RD (1996) TreeView: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12(4):357–358
Posada D (2003) Using MODELTEST and PAUP* to select a model of nucleotide substitution. Current Proto Bioinformatics Chapter 6:Unit 6.5
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19(12):1572–1574
Rosser TG, Alberson NR, Baumgartner WA, Mauel MJ, Pote LM, Griffin MJ (2015) Morphological, histological and molecular description of Unicauda fimbrethilae n. sp. (Cnidaria: Myxosporea: Myxobolidae) from the intestinal tract of channel catfish Ictalurus punctatus. J Parasitol (In press)
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X Windows interface: flexible strategies for multiple sequence alignment aided by Quality Analysis Tools. Nucleic Acids Res 25:4876–4883
Whipps CM, Adlard RD, Bryant MS, Lester RJG, Finflay V, Kent ML (2003) First report of three Kudoa species from eastern Australia: Kudoa thyrsites from mahi mahi (Coryphaena hippurus), Kudoa amamiensis and Kudoa minithyrsites n. sp. from sweeper (Pempheris ypsilychnus). J Eukuryot Microbiol 50(3):215–219
Zhang JY, Yokoyama H, Wang JG, Li AH, Gong XN, Ryu–Hasegawa A, Iwashita M, Ogawa K (2010) Utilization oftissue habitats by Myxobolus wulii Landsberg & Lom, 1991 in different carp hosts and disease resistance in allogynogenetic gibel carp: redescription of M. wulii from China and Japan. J Fish Dis 33:57–68
Zhang JY, Al-Quraishy S, Abdel-Baki AAS (2014) The morphological and molecular characterization of Myxobolus khaliji n. sp. (Myxozoa: Myxosporea) from the double bar seabream Acanthopagrus bifasciatus (Forsskål, 1775) in the Arabian Gulf, Saudi Arabia. Parasitol Res 113:2177–2183
Acknowledgments
The study was partially supported by the Chinese Natural Sciences Fund (31101623, 31411130191, 31472296) and Russian Found of Fundamental Investigation (14–04 = 91176).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, X.H., Zhang, J.Y., Batueva, M.D. et al. Supplemental description and molecular characterization of Myxobolus miyarii Kudo, 1919 (Myxosporea: Myxobolidae) infecting intestine of Amur catfish (Silurus asotus). Parasitol Res 115, 1547–1556 (2016). https://doi.org/10.1007/s00436-015-4889-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4889-x