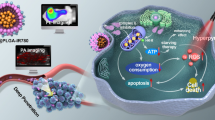
Abstract
Background
Bladder cancer has a high rate of recurrence and drug resistance due to the lack of effective therapies. IR-780 iodide, a near-infrared (NIR) mitochondria-targeting fluorescent agent, has been demonstrated to achieve higher selectivity than other drugs in different tumor types and exhibited tumor-killing effects in some cancers. However, this therapeutic strategy is rarely studied in bladder cancer.
Material and methods
The accumulation of IR-780 in bladder cancer was measured by NIR imaging. Human bladder cell lines (T24, 5637, and TCCSUP) were treated with IR-780 or combined IR-780 and hyperbaric oxygen (HBO). Cell viability, cell apoptosis, cellular ATP production, mitochondrial reactive oxygen species (ROS), and plasma membrane potential were detected. Mitochondrial complex I protein NDUFS1 was measured by western blot. To confirm the anti-tumor efficacy of IR-780 + HBO, mouse bladder cell line (MB49) tumor-bearing mice were established and tumor size and weight were recorded. Besides, cell apoptosis and tumor size were assessed in drug-resistant bladder cancer cells (T24/DDP) and xenografts to evaluate the effect of IR-780 + HBO on drug-resistant bladder cancer.
Results
IR-780 selectively accumulated in bladder cancer (bladder cancer cells, transplanted tumors, and bladder cancer tissue from patients) and could induce cancer cell apoptosis by targeting the mitochondrial complex I protein NDUFS1. The combination with HBO could significantly enhance the anti-tumor effect of IR-780 in vitro by promoting cancer cell uptake and inducing excessive mitochondrial ROS production, while suppressing tumor growth and recurrence in animal models without causing apparent toxicity. Moreover, this combination antitumor strategy was also demonstrated in drug-resistant bladder cancer cells (T24/DDP) and xenografts.
Conclusion
We identified for the first time a combination of IR-780 and HBO (IR-780 + HBO), which exhibits mitochondria-targeting and therapeutic capabilities, as a novel treatment paradigm for bladder cancer.







Similar content being viewed by others
Availability of data and materials
The datasets used during the current study are available from the corresponding author on reasonable request.
References
Alexander R, Vultur I, Bogeski H, Wang K, Zimmermann D, Speicher C, Körbel M, Laschke P, Gimotty S, Philipp E, Krause S, Pätzold J, Villanueva C, Krepler M, Fukunaga-Kalabis M, Hoth Boris C, Bastian T, Vogt M, Herlyn (2013) Overcoming Intrinsic Multidrug Resistance in Melanoma by Blocking the Mitochondrial Respiratory Chain of Slow-Cycling JARID1Bhigh Cells. Cancer Cell 23(6):811–825
Alfred Witjes J, Lebret T, Comperat EM, Cowan NC, De Santis M, Bruins HM, Hernandez V, Espinos EL, Dunn J, Rouanne M et al (2017) Updated 2016 EAU guidelines on muscle-invasive and metastatic bladder cancer. Eur Urol 71:462–475
Berkenblit A, Eder JP Jr, Ryan DP, Seiden MV, Tatsuta N, Sherman ML, Dahl TA, Dezube BJ, Supko JG (2007) Phase I clinical trial of STA-4783 in combination with paclitaxel in patients with refractory solid tumors. Clin Cancer Res 13:584–590
Cao H, Zou L, He B, Zeng L, Huang Y, Yu H, Zhang P, Yin Q, Zhang Z, Li Y (2017) Albumin biomimetic nanocorona improves tumor targeting and penetration for synergistic therapy of metastatic breast cancer. Adv Funct Mater 27(11):1605679
Cierlitza M, Chauvistre H, Bogeski I, Zhang X, Hauschild A, Herlyn M, Schadendorf D, Vogt T, Roesch A (2015) Mitochondrial oxidative stress as a novel therapeutic target to overcome intrinsic drug resistance in melanoma cell subpopulations. Exp Dermatol 24:155–157
Cui Q, Wang J-Q, Assaraf YG, Ren L, Gupta P, Wei L, Ashby CR, Yang D-H, Chen Z-S (2018) Modulating ROS to overcome multidrug resistance in cancer. Drug Resist Updates 41:1–25
Davis S, Weiss M, Wong J, Lampidis T, Chen L (1985) Mitochondrial and plasma membrane potentials cause unusual accumulation and retention of rhodamine 123 by human breast adenocarcinoma-derived MCF-7 cells. J Biol Chem 260:13844–13850
Deribe YL, Sun Y, Terranova C, Khan F, Martinez-Ledesma J, Gay J, Gao G, Mullinax RA, Khor T, Feng N et al (2018) Author correction: mutations in the SWI/SNF complex induce a targetable dependence on oxidative phosphorylation in lung cancer. Nat Med 24:1627
Desai N (2012) Challenges in development of nanoparticle-based therapeutics. AAPS J 14:282–295
Dewhirst M, Mowery Y, Mitchell J, Cherukuri M, Secomb T (2019) Rationale for hypoxia assessment and amelioration for precision therapy and immunotherapy studies. J Clin Investig 129:489–491
Erlong Z, Shenglin L, Xu T, Chunmeng, and Shi, (2014) Mechanistic study of IR-780 dye as a potential tumor targeting and drug delivery agent - ScienceDirect. Biomaterials 35:771–778
Fang D, Maldonado E (2018) VDAC regulation: a mitochondrial target to stop cell proliferation. Adv Cancer Res 138:41–69
Hao W, Chang CP, Tsao CC, Xu J (2010) Oligomycin-induced bioenergetic adaptation in cancer cells with heterogeneous bioenergetic organization. J Biol Chem 285:12647–12654
Hoskin P, Sibtain A, Daley F, Wilson G (2003) GLUT1 and CAIX as intrinsic markers of hypoxia in bladder cancer: relationship with vascularity and proliferation as predictors of outcome of ARCON. Br J Cancer 89:1290–1297
Housman G, Byler S, Heerboth S, Lapinska K, Longacre M, Snyder N, Sarkar S (2014) Drug resistance in cancer: an overview. Cancers 6:1769–1792
Idris NM, Jayakumar MK, Bansal A, Zhang Y (2015) Upconversion nanoparticles as versatile light nanotransducers for photoactivation applications. Chem Soc Rev 44:1449–1478
Jennifer R., Molina Y, Sun M, Protopopova S, Gera M, Bandi C, Bristow T, McAfoos P, Morlacchi J, Ackroyd AA., Agip G, Al-Atrash J, Asara J, Bardenhagen CC., Carrillo C, Carroll E, Chang S, Ciurea JB., Cross B, Czako A, Deem N, Daver JF, de Groot JW, Dong N Feng G, Gao J, Gay Mary Geck, Do Jr, Greer Vi, Giuliani J, Han L Han VK., Henry J, Hirst S, Huang Y, Jiang Z, Kang T, Khor S, Konoplev YH, Lin G, Liu A, Lodi Timothy, Lofton Helen, Ma Mikhila, Mahendra Polina, Matre Robert, Mullinax Michael, Peoples Alessia, Petrocchi Jaime, Rodriguez-Canale Riccardo, Serreli Thomas, Shi Melinda, Smith Yoko, Tabe Jay, Theroff Stefano, Tiziani Quanyun, Xu Qi, Zhang Florian, Muller Ronald A., DePinho Carlo, Toniatti Giulio F, Draetta TP, Heffernan M, Konopleva P, Jones M. E, Di Francesco JR., Marszalek (2018) An inhibitor of oxidative phosphorylation exploits cancer vulnerability. Nat Med 24(7):1036–S1046
Jiang Q, Zhang C, Wang H, Peng T, Zhang L, Wang Y, Han W, Shi C (2019) Mitochondria-targeting immunogenic cell death inducer improves the adoptive T-cell therapy against solid tumor. Front Oncol 9:1196
Jose C, Bellance N, Rossignol R (2011) Choosing between glycolysis and oxidative phosphorylation: a tumor’s dilemma? Biochim Biophys Acta 1807:552–561
Klyubin I, Kirpichnikova K, Ischenko A, Zhakhov A, Gamaley I (2000) The role of reactive oxygen species in membrane potential changes in macrophages and astrocytes. Membr Cell Biol 13:557–566
Kuang Y, Zhang K, Cao Y, Chen X, Wang K, Liu M, Pei R (2017) Hydrophobic IR-780 dye encapsulated in cRGD-conjugated solid lipid nanoparticles for NIR imaging-guided photothermal therapy. ACS Appl Mater Interfaces 9:12217–12226
Lucky SS, Muhammad Idris N, Li Z, Huang K, Soo KC, Zhang Y (2015) Titania coated upconversion nanoparticles for near-infrared light triggered photodynamic therapy. ACS Nano 9:191–205
Luo S, Zhang E, Su Y, Cheng T, Shi C (2011) A review of NIR dyes in cancer targeting and imaging. Biomaterials 32:7127–7138
Luo S, Tan X, Fang S, Wang Y, Liu T, Wang X, Yuan Y, Sun H, Qi Q, Shi C (2016) Mitochondria-targeted small-molecule fluorophores for dual modal cancer phototherapy. Adv Func Mater 26:2826–2835
Molina JR, Sun Y, Protopopova M, Gera S, Bandi M, Bristow C, McAfoos T, Morlacchi P, Ackroyd J et al (2018) An inhibitor of oxidative phosphorylation exploits cancer vulnerability. Nat Med 24:1036–1046
Nolfi-Donegan D, Braganza A, Shiva S (2020) Mitochondrial electron transport chain: oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol 37:101674
Oresta B, Pozzi C, Braga D, Hurle R, Lazzeri M, Colombo P, Frego N, Erreni M, Faccani C, Elefante G (2021) Mitochondrial metabolic reprogramming controls the induction of immunogenic cell death and efficacy of chemotherapy in bladder cancer. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aba6110
Ploeg M, Aben KKH, Kiemeney LA (2009) The present and future burden of urinary bladder cancer in the world. World J Urol 27:289–293
Porporato PE, Filigheddu N, Pedro JMB-S, Kroemer G, Galluzzi L (2017) Mitochondrial metabolism and cancer. Cell Res 28:265–280
Richters A, Aben K, Kiemeney L (2020) The global burden of urinary bladder cancer: an update. World J Urol 38:1895–1904
Roesch A, Vultur A, Bogeski I, Wang H, Zimmermann KM, Speicher D, Korbel C, Laschke MW, Gimotty PA, Philipp SE et al (2013) Overcoming intrinsic multidrug resistance in melanoma by blocking the mitochondrial respiratory chain of slow-cycling JARID1B(high) cells. Cancer Cell 23:811–825
Sabharwal SS, Schumacker PT (2014) Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles’ heel? Nat Rev Cancer 14:709–721
Sahu D, Lotan Y, Wittmann B, Neri B, Hansel DE (2017) Metabolomics analysis reveals distinct profiles of nonmuscle-invasive and muscle-invasive bladder cancer. Cancer Med 6(9):2106–2120. https://doi.org/10.1002/cam4.1109
Santos J, Blackman RK, Cheung-Ong K, Gebbia M, Proia DA, He S, Kepros J, Jonneaux A, Marchetti P, Kluza J et al (2012) Mitochondrial electron transport is the cellular target of the oncology drug elesclomol. PLoS ONE 7(1):e29798
Selvendiran K, Kuppusamy ML, Ahmed S, Bratasz A, Meenakshisundaram G, Rivera BK, Khan M, Kuppusamy P (2010) Oxygenation inhibits ovarian tumor growth by downregulating STAT3 and cyclin-D1 expressions. Cancer Biol Ther 10:386–390
Stępień K, Ostrowski R, Matyja E (2016) Hyperbaric oxygen as an adjunctive therapy in treatment of malignancies, including brain tumours. Med Oncol (northwood, London, England) 33:101
Tan X, Luo S, Long L, Wang Y, Wang D, Fang S, Ouyang Q, Su Y, Cheng T, Shi C (2017) Structure-guided design and synthesis of a mitochondria-targeting near-infrared fluorophore with multimodal therapeutic activities. Adv Mater 29(43):1704196. https://doi.org/10.1002/adma.201704196
Teo MY, Rosenberg JE (2018) Nivolumab for the treatment of urothelial cancers. Expert Rev Anticancer Ther 14737140(14732018):11432357
Teoh J, Kamat A, Black P, Grivas P, Shariat S, Babjuk M (2022) Recurrence mechanisms of non-muscle-invasive bladder cancer - a clinical perspective. Nat Rev Urol 19:280–294
Uthaman S, Mathew AP, Park HJ, Lee BI, Kim HS, Huh KM, Park IK (2018) IR 780-loaded hyaluronic acid micelles for enhanced tumor-targeted photothermal therapy. Carbohydr Polym 181:1–9
Wang Y, Liu T, Zhang E, Luo S, Tan X, Shi C (2014) Preferential accumulation of the near infrared heptamethine dye IR-780 in the mitochondria of drug-resistant lung cancer cells. Biomaterials 35:4116–4124
Wang Y, Liao X, Sun J, Yi B, Luo S, Liu T, Tan X, Liu D, Chen Z, Wang X et al (2018a) Characterization of HIF-1α/glycolysis hyperactive cell population via small-molecule-based imaging of mitochondrial transporter activity. Adv Sci 5(3):1700392
Wang Y, Luo S, Zhang C, Liao X, Liu T, Jiang Z, Liu D, Tan X, Long L, Wang Y et al (2018b) An NIR-fluorophore-based therapeutic endoplasmic reticulum stress inducer. Adv Mater. https://doi.org/10.1002/adma.201800475
Weinberg SE, Chandel NS (2015) Targeting mitochondria metabolism for cancer therapy. Nat Chem Biol 11:9–15
Wen S, Zhu D, Huang P (2013) Targeting cancer cell mitochondria as a therapeutic approach. Future Med Chem 5:53–67
Woolbright Benjamin L, Ayres M et al (2018) Metabolic changes in bladder cancer. Urol Orol 36(7):327
Wu L, Zhao J, Cao K, Liu X, Cai H, Wang J, Li W, Chen Z (2018) Oxidative phosphorylation activation is an important characteristic of DOX resistance in hepatocellular carcinoma cells. Cell Commun Signal. https://doi.org/10.1186/s12964-018-0217-2
Wx Z, Jd R, E, W. (2016) Mitochondria and cancer. Mol Cell 61:667–676
Xiao T, Fan JK, Huang HL, Gu JF, Li LY, Liu XY (2010) VEGI-armed oncolytic adenovirus inhibits tumor neovascularization and directly induces mitochondria-mediated cancer cell apoptosis. Cell Res 20:367–378
Xiao L, Xu C, Lin P, Mu L, Yang X (2022) Novel dihydroartemisinin derivative Mito-DHA induces apoptosis associated with mitochondrial pathway in bladder cancer cells. BMC Pharmacol Toxicol 23:10
Xu Y, Lei Z, Zhu J, Wan L (2022) Mivebresib synergized with PZ703b, a novel Bcl-xl PROTAC degrader, induces apoptosis in bladder cancer cells via the mitochondrial pathway. Biochem Biophys Res Commun 623:120–126
Yan F, Duan W, Li Y, Wu H, Zhou Y, Pan M, Liu H, Liu X, Zheng H (2016) NIR-laser-controlled drug release from DOX/IR-780-loaded temperature-sensitive-liposomes for chemo-photothermal synergistic tumor therapy. Theranostics 6:2337–2351
Yufeng Shi S, Lim K, Liang Q, Iyer SV, Wang HY, Wang Z, Xie X, Sun D et al (2019) Gboxin is an oxidative phosphorylation inhibitor that targets glioblastoma. Nature 567:341–346
Zhang C, Liu T, Su Y, Luo S, Zhu Y, Tan X, Fan S, Zhang L, Zhou Y, Cheng T et al (2010) A near-infrared fluorescent heptamethine indocyanine dye with preferential tumor accumulation for in vivo imaging. Biomaterials 31:6612–6617
Zhang E, Luo S, Tan X, Shi C (2014) Mechanistic study of IR-780 dye as a potential tumor targeting and drug delivery agent. Biomaterials 35:771–778
Zhao T, Wu W, Sui L, Huang Q, Nan Y, Liu J, Ai K (2022) Reactive oxygen species-based nanomaterials for the treatment of myocardial ischemia reperfusion injuries. Bioact Mater 7:47–72
Zhu Y, Zhao T, Liu M, Wang S, Liu S, Yang Y, Yang Y, Nan Y, Huang Q, Ai K (2022) Rheumatoid arthritis microenvironment insights into treatment effect of nanomaterials. Nano Today 42:101358. https://doi.org/10.1016/j.nantod.2021.101358
Zielonka J, Joseph J, Sikora A, Hardy M, Ouari O, Vasquez-Vivar J, Cheng G, Lopez M, Kalyanaraman B (2017) Mitochondria-targeted triphenylphosphonium-based compounds: syntheses, mechanisms of action, and therapeutic and diagnostic applications. Chem Rev 117:10043–10120
Acknowledgements
We thank the neurosurgery department of Southwest Hospital (Chongqing, China) for providing a temperature-adjustable hyperbaric chamber. The manuscript has been presented as “pre-print” with the following link: https://www.researchsquare.com/article/rs-111008/v1.
Funding
This work was supported by National Natural Science Foundation of Chongqing Province (No.cstc 2018jcyjAX0034), National Key Research and Development Program (2016YFC1000805, to C. Shi), National Natural Science Foundation of Chongqing Province (No.cstc 2021 jcyj-bsh0238 to X. Yue), Key research incubation in the Third Affiliated Hospital of Chongqing Medical University (KY08026, to Y. Zhi), Science and technology project of Yubei District, Chongqing (2021-NS-38, to Y. Zhi).
Author information
Authors and Affiliations
Contributions
LWB & SCM conceived and designed the experiments, supervised the experiments and revised the manuscript. SCX performed the experiments, analyzed the data and drafted the manuscript. YXF and WJW took part in the animal experiments, drafted the manuscript and analysis of optical properties. DLY and LJJ participated in Flow Cytometer and western blot analysis. FQ and ZY contributed to collecting human tissues. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
The collection of human tumor samples and related procedures were approved by the Ethical Committee of the Third Affiliated Hospital, Chongqing Medical University. The animal experiments in our study were conducted in accordance with the Helsinki Declaration and approved by the Laboratory Animal Welfare and Ethics Committee of Third Military Medical University.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, C., Yue, X., Dai, L. et al. Hyperbaric oxygen enhanced the chemotherapy of mitochondrial targeting molecule IR-780 in bladder cancer. J Cancer Res Clin Oncol 149, 683–699 (2023). https://doi.org/10.1007/s00432-022-04385-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04385-4