Abstract
Purpose
TMEM176B was recently described as a negative modulator of Nlrp3 inflammasome activation in mice. In the mouse model, the inhibition of TMEM176B leads to an increased anti-tumoral activity which is dependent on Nlrp3. Since we have recently shown that single nucleotide variants (SNPs) in inflammasome genes, including NLRP3, significantly affect colorectal cancer (CRC) prognosis, we proposed to investigate here the association between genetic variants in TMEM176B and CRC prognosis.
Methods
Considering that, up to now, no genetic study analyzing this gene in humans exists, we selected possible functional SNPs and genotyped them in a cohort of CRC patients submitted to surgery and followed up for more than 10 years. Genotype-guided assays were realized to evaluate the effect of the variant on NLRP3 inflammasome activation. Gene expression from The Cancer Genome Atlas (TCGA) cohort was analyzed to valid possible prognostic and predictive features.
Results
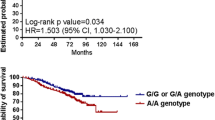
We identified the Ala134Thr variant (rs2072443) in TMEM176B as a protective factor for CRC prognosis. This SNP is associated with decreased gene expression and with an increased activation of NLRP3 inflammasome, at least in monocytes and dendritic cells. Furthermore, low TMEM176B expression is associated with higher overall survival.
Conclusion
Altogether, these findings supported the role of TMEM176B in NLRP3 inflammasome biology and for the first time demonstrated the genetic association between rs2072443 and CRC in humans.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ali MF, Dasari H, Van Keulen VP, Carmona EM (2017) Canonical stimulation of the NLRP3 inflammasome by fungal antigens links innate and adaptive B-lymphocyte responses by modulating IL-1β and IgM production. Front Immunol 8:1504. https://doi.org/10.3389/fimmu.2017.01504
Allen IC, TeKippe EM, Woodford RM, Uronis JM, Holl EK, Rogers AB, Herfarth HH, Jobin C, Ting JP (2010) The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. J Exp Med 207(5):1045–1056. https://doi.org/10.1084/jem.20100050
Anderson NM, Simon MC (2020) The tumor microenvironment. Curr Biol 30(16):R921–R925. https://doi.org/10.1016/j.cub.2020.06.081
Arbore G, West EE, Spolski R, Robertson AAB, Klos A, Rheinheimer C, Dutow P, Woodruff TM, Yu ZX, O’Neill LA, Coll RC, Sher A, Leonard WJ, Köhl J, Monk P, Cooper MA, Arno M, Afzali B, Lachmann HJ, Cope AP, Mayer-Barber KD, Kemper C (2016) T helper 1 immunity requires complement-driven NLRP3 inflammasome activity in CD4+ T cells. Science 352(6292):aad1210. https://doi.org/10.1126/science.aad1210
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21(2):263–265. https://doi.org/10.1093/bioinformatics/bth457
BRASIL (2014) Ministério da Saúde. Gabinete do Ministro de Saúde. Secretaria de Atenção à Saúde. Portaria GM/MS nº 1.271, de 06 de junho de 2014. http://bvsms.saude.gov.br/bvs/saudelegis/gm/2014/prt1271_06_06_2014.html .
Cambui RAG, do Espírito Santo GF, Fernandes FP, Leal VNC, Galera BB, Fávaro EGP, Rizzo LA, Elias RM, Pontillo A (2020) Double-edged sword of inflammasome genetics in colorectal cancer prognosis. Clin Immunol 213:108373. https://doi.org/10.1016/j.clim.2020.108373
Christgen S, Kanneganti T-D (2020) Inflammasomes and the fine line between defense and disease. Curr Opin Immunol 62:39–44. https://doi.org/10.1016/j.coi.2019.11.007
Condamine T, Le Texier L, Howie D, Lavault A, Hill M, Halary F, Cobbold S, Waldmann H, Cuturi MC, Chiffoleau E (2010) Tmem176B and Tmem176A are associated with the immature state of dendritic cells. J Leukoc Biol 88(3):507–515. https://doi.org/10.1189/jlb.1109738
De Mattia E, Bignucolo A, Toffoli G, Cecchin E (2020) Genetic markers of the host to predict the efficacy of colorectal cancer targeted therapy. Curr Med Chem 27(25):4249–4273. https://doi.org/10.2174/0929867326666190712151417
Dmitrieva-Posocco O, Dzutsev A, Posocco DF, Hou V, Yuan W, Thovarai V, Mufazalov IA, Gunzer M, Shilovskiy IP, Khaitov MR, Trinchieri G, Waisman A, Grivennikov SI (2019) Cell-type-specific responses to interleukin-1 control microbial invasion and tumor-elicited inflammation in colorectal cancer. Immunity 50(1):166-180.e7. https://doi.org/10.1016/j.immuni.2018.11.015
Dos Reis EC, Leal VNC, Soares JLDS, Fernandes FP, Souza de Lima D, de Alencar BC, Pontillo A (2019) Flagellin/NLRC4 pathway rescues NLRP3-Inflammasome defect in dendritic cells from HIV-infected patients: perspective for new adjuvant in immunocompromised individuals. Front Immunol 2019(10):1291. https://doi.org/10.3389/fimmu.2019.01291
Dupaul-Chicoine J, Arabzadeh A, Dagenais M, Douglas T, Champagne C, Morizot A, Rodrigue-Gervais IG, Breton V, Colpitts SL, Beauchemin N, Saleh M (2015) The Nlrp3 inflammasome suppresses colorectal cancer metastatic growth in the liver by promoting natural killer cell tumoricidal activity. Immunity 43(4):751–763. https://doi.org/10.1016/j.immuni.2015.08.013
Fernandes FP, Leal VNC, Souza de Lima D, Reis EC, Pontillo A (2020) Inflammasome genetics and complex diseases: a comprehensive review. Eur J Hum Genet 28(10):1307–1321. https://doi.org/10.1038/s41431-020-0631-y
Gattorno M, Tassi S, Carta S, Delfino L, Ferlito F, Pelagatti MA, D'Osualdo A, Buoncompagni A, Alpigiani MG, Alessio M, Martini A, Rubartelli A (2007) Pattern of interleukin-1beta secretion in response to lipopolysaccharide and ATP before and after interleukin-1 blockade in patients with CIAS1 mutations. Arthritis Rheum 56(9):3138–3148. https://doi.org/10.1002/art.22842
Huber S, Gagliani N, Zenewicz LA, Huber FJ, Bosurgi L, Hu B, Hedl M, Zhang W, O’Connor W Jr, Murphy AJ, Valenzuela DM, Yancopoulos GD, Booth CJ, Cho JH, Ouyang W, Abraham C, Flavell RA (2012) IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature 491(7423):259–263. https://doi.org/10.1038/nature11535
Karki R, Man SM, Kanneganti TD (2017) Inflammasomes and cancer. Cancer Immunol Res 5(2):94–99. https://doi.org/10.1158/2326-6066
Kolb R, Liu GH, Janowski AM, Sutterwala FS, Zhang W (2014) Inflammasomes in cancer: a double-edged sword. Protein Cell 5(1):12–20. https://doi.org/10.1007/s13238-013-0001-4
Levandowski CB, Mailloux CM, Ferrara TM, Gowan K, Ben S, Jin Y, McFann KK, Holland PJ, Fain PR, Dinarello CA, Spritz RA (2013) NLRP1 haplotypes associated with vitiligo and autoimmunity increase interleukin-1β processing via the NLRP1 inflammasome. Proc Natl Acad Sci USA 110(8):2952–2956. https://doi.org/10.1073/pnas.1222808110
Mantovani A, Dinarello CA, Molgora M, Garlanda C (2019) Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity 50(4):778–795
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215. https://doi.org/10.1093/nar/16.3.1215
Nickles D, Chen HP, Li MM, Khankhanian P, Madireddy L, Caillier SJ, Santaniello A, Cree BA, Pelletier D, Hauser SL, Oksenberg JR, Baranzini SE (2013) Blood RNA profiling in a large cohort of multiple sclerosis patients and healthy controls. Hum Mol Genet 22(20):4194–4205. https://doi.org/10.1093/hmg/ddt267
Rathinam VAK, Chan FK (2018) Inflammasome, inflammation, and tissue homeostasis. Trends Mol Med 24(3):304–318. https://doi.org/10.1016/j.molmed.2018.01.004
Segovia M, Russo S, Jeldres M, Mahmoud YD, Perez V, Duhalde M, Charnet P, Rousset M, Victoria S, Veigas F, Louvet C, Vanhove B, Floto RA, Anegon I, Cuturi MC, Girotti MR, Rabinovich GA, Hill M (2019) Targeting TMEM176B enhances antitumor immunity and augments the efficacy of immune checkpoint blockers by unleashing inflammasome activation. Cancer Cell 35(5):767-781.e6. https://doi.org/10.1016/j.ccell.2019.04.003
Souza de Lima D, Bomfim CCB, Leal VNC, Reis EC, Soares JLS, Fernandes FP, Amaral EP, Loures FV, Ogusku MM, Lima MRD, Sadahiro A, Pontillo A (2020) Combining host genetics and functional analysis to depict inflammasome contribution in tuberculosis susceptibility and outcome in endemic areas. Front Immunol 11:550624. https://doi.org/10.3389/fimmu.2020.550624
Tulotta C, Lefley DV, Moore CK et al (2021) IL-1B drives opposing responses in primary tumours and bone metastases; harnessing combination therapies to improve outcome in breast cancer. Npj Breast Cancer 7:95. https://doi.org/10.1038/s41523-021-00305-w
Ungerbäck J, Belenki D, Jawad ul-Hassan A, Fredrikson M, Fransén K, Elander N, Verma D, Söderkvist P (2012) Genetic variation and alterations of genes involved in NFκB/TNFAIP3- and NLRP3-inflammasome signaling affect susceptibility and outcome of colorectal cancer. Carcinogenesis 33(11):2126–2134. https://doi.org/10.1093/carcin/bgs256
Verma D, Bivik C, Farahani E, Synnerstad I, Fredrikson M, Enerbäck C, Rosdahl I, Söderkvist P (2012) Inflammasome polymorphisms confer susceptibility to sporadic malignant melanoma. Pigment Cell Melanoma Res 25(4):506–513. https://doi.org/10.1111/j.1755-148X.2012.01008.x
Wang H, Wang Y, Du Q, Lu P, Fan H, Lu J, Hu R (2016) Inflammasome-independent NLRP3 is required for epithelial-mesenchymal transition in colon cancer cells. Exp Cell Res 342(2):184–192. https://doi.org/10.1016/j.yexcr.2016.03.009
Wang B, Li H, Wang X, Zhu X (2020) The association of aberrant expression of NLRP3 and p-S6K1 in colorectal cancer. Pathol Res Pract 216(1):152737. https://doi.org/10.1016/j.prp.2019.152737
Zaki MH, Boyd KL, Vogel P, Kastan MB, Lamkanfi M, Kanneganti TD (2010) The NLRP3 inflammasome protects against loss of epithelial integrity and mortality during experimental colitis. Immunity 32(3):379–391. https://doi.org/10.1016/j.immuni.2010.03.003
Zheng D, Liwinski T, Elinav E (2020) Inflammasome activation and regulation: toward a better understanding of complex mechanisms. Cell Discov 9(6):36. https://doi.org/10.1038/s41421-020-0167-x
Acknowledgements
The authors would like to acknowledge to all the volunteers and the professional team from “Instituto de Tumores e Cuidados Paliativos'' of the “Hospital Geral”, “Clínica de Tratamento Multidisciplinar do Câncer” (ONCOMED) and “Oswaldo Cruz” Hospital for the samples and clinical data. Authors also would like to thank Prof. Marcelo Hill from the “Universidad de la Republica” and Institute Pasteur, Montevideo, Uruguay for helpful discussion, and the Laboratory of Genetics and Molecular Biology, Institute of Medicine, Federal University of Mato Grosso (Cuiaba, Brazil) headed by Dr. Bianca Borsatto Galera for the assistance with the samples handling and storage.
Funding
This study was supported by “Fundação de Suporte a Pesquisa do Estado de Sao Paulo” (FAPESP; grant number 19/06363-4). A.P. is a recipient of a Fellowship from “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq; grant number 302206/2019-1). R.A.G.C. is a recipient of a PhD Fellowship from “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (CAPES; grant number 88887.469122/2019-00). The other authors have no competing interests to declare that are relevant to the content of this article.
Author information
Authors and Affiliations
Contributions
RAGC and AP designed the study, analyzed the data, wrote the manuscript and did the association study and genotype-guided assays. RAGC, VNCL and FPF performed the genotyping assay. VNCL, ECR and DSL realized the cell culture assays. GFES and RME recruited the CRC patients and collected the clinical data.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no conflict of interest.
Ethical approval
All the experiments involving human participants were approved by the Ethics Committee in Research in Human Beings (CEPSH) of the Institute of Biomedical Science/University of Sao Paulo.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cambui, R.A.G., Fernandes, F.P., Leal, V.N.C. et al. The Ala134Thr variant in TMEM176B exerts a beneficial role in colorectal cancer prognosis by increasing NLRP3 inflammasome activation. J Cancer Res Clin Oncol 149, 3729–3738 (2023). https://doi.org/10.1007/s00432-022-04284-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04284-8