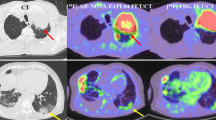
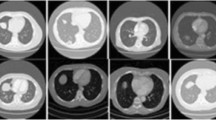
Abstract
Purpose
To investigate the incremental value of enhanced CT-based radiomics in discriminating between pulmonary tuberculosis (PTB) and pulmonary adenocarcinoma (PAC) presenting as solid nodules or masses and to develop an optimal radiomics model.
Methods
A total of 128 lesions (from 123 patients) from three hospitals were retrospectively analyzed and were randomly divided into training and test datasets at a ratio of 7:3. Independent predictors in subjective image features were used to develop the subjective image model (SIM). The plain CT-based and enhanced CT-based radiomics features were screened by the correlation coefficient method, univariate analysis, and the least absolute shrinkage and selection operator, then used to build the plain CT radiomics model (PRM) and enhanced CT radiomics model (ERM), respectively. Finally, the combined model (CM) combining PRM and ERM was established. In addition, the performance of three radiologists and one respiratory physician was evaluated. The areas under the receiver operating characteristic curve (AUCs) were used to assess the performance of each model.
Results
The differential diagnostic capability of the ERM (training: AUC = 0.933; test: AUC = 0.881) was better than that of the PRM (training: AUC = 0.861; test: AUC = 0.756) and the SIM (training: AUC = 0.760; test: AUC = 0.611). The CM was optimal (training: AUC = 0.948; test: AUC = 0.917) and outperformed the respiratory physician and most radiologists.
Conclusions
The ERM was more helpful than the PRM for identifying PTB and PAC that present as solid nodules or masses, and the CM was the best.







Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- PTB:
-
Pulmonary tuberculosis
- PAC:
-
Pulmonary adenocarcinoma
- VOIs:
-
Volumes of interest
- GLCM:
-
Gray-level co-occurrence matrix
- GLDM:
-
Gray-level dependence matrix
- GLRLM:
-
Gray-level run-length matrix
- GLSZM:
-
Gray-level size zone matrix
- NGTDM:
-
Neighboring gray-tone difference matrix
- ICC:
-
Inter- and intra-class correlation coefficients
- LASSO:
-
Least absolute shrinkage and selection operator
- SIM:
-
Subjective image model
- PRM:
-
Plain CT radiomics model
- ERM:
-
Enhanced CT radiomics model
- CM:
-
Combined model
- LR:
-
Logistic regression
- RF:
-
Random forest
- SVM:
-
Support vector machine
- DT:
-
Decision tree
- KNN:
-
K-nearest neighbor
- ROC:
-
Receiver operating characteristic
- AUC:
-
Area under the receiver operator characteristic curve
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- TRIPOD:
-
Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis
References
Ai B, Zhang L, Huang D, Chen J, Liu Z, Hu X, Zhou S, Hu Y, Zhao J, Yang F (2020) Efficacy and safety of bevacizumab in advanced lung adenocarcinoma patients with stable disease after two cycles of first-line chemotherapy: a multicenter prospective cohort study. Thorac Cancer 11:3641–3644. https://doi.org/10.1111/1759-7714.13687
Beig N, Khorrami M, Alilou M, Prasanna P, Braman N, Orooji M, Rakshit S, Bera K, Rajiah P, Ginsberg J, Donatelli C, Thawani R, Yang M, Jacono F, Tiwari P, Velcheti V, Gilkeson R, Linden P, Madabhushi A (2019) Perinodular and intranodular radiomic features on lung CT images distinguish adenocarcinomas from granulomas. Radiology 290:783–792. https://doi.org/10.1148/radiol.2018180910
Burrill J, Williams CJ, Bain G, Conder G, Hine AL, Misra RR (2007) Tuberculosis: a radiologic review. Radiographics 27:1255–1273. https://doi.org/10.1148/rg.275065176
Cardinale L, Ardissone F, Novello S, Busso M, Solitro F, Longo M, Sardo D, Giors M, Fava C (2009) The pulmonary nodule: clinical and radiological characteristics affecting a diagnosis of malignancy. Radiol Med 114:871–889. https://doi.org/10.1007/s11547-009-0399-1
Chen X, Feng B, Chen Y, Liu K, Li K, Duan X, Hao Y, Cui E, Liu Z, Zhang C, Long W, Liu X (2020a) A CT-based radiomics nomogram for prediction of lung adenocarcinomas and granulomatous lesions in patient with solitary sub-centimeter solid nodules. Cancer Imaging 20:45. https://doi.org/10.1186/s40644-020-00320-3
Chen X, Yang Z, Yang J, Liao Y, Pang P, Fan W, Chen X (2020b) Radiomics analysis of contrast-enhanced CT predicts lymphovascular invasion and disease outcome in gastric cancer: a preliminary study. Cancer Imaging 20:24. https://doi.org/10.1186/s40644-020-00302-5
Chen W, Li M, Mao D, Ge X, Wang J, Tan M, Ma W, Huang X, Lu J, Li C, Hua Y, Wu H (2021) Radiomics signature on CECT as a predictive factor for invasiveness of lung adenocarcinoma manifesting as subcentimeter ground glass nodules. Sci Rep 11:3633. https://doi.org/10.1038/s41598-021-83167-3
Choe J, Lee SM, Do K-H, Kim S, Choi S, Lee J-G, Seo JB (2020) Outcome prediction in resectable lung adenocarcinoma patients: value of CT radiomics. Eur Radiol 30:4952–4963. https://doi.org/10.1007/s00330-020-06872-z
Collins GS, Reitsma JB, Altman DG, Moons KGM (2015) Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. J Clin Epidemiol 682:112–121. https://doi.org/10.1016/j.jclinepi.2014.11.010
Cui E-N, Yu T, Shang S-J, Wang X-Y, Jin Y-L, Dong Y, Zhao H, Luo Y-H, Jiang X-R (2020) Radiomics model for distinguishing tuberculosis and lung cancer on computed tomography scans. World J Clin Cases 8:5203–5212. https://doi.org/10.12998/wjcc.v8.i21.5203
Fang R, Yang Y, Han H, Fu X, Dong L, Xie B, Lu W, Ma C, Cui F, Hu J, Wang J (2018) Analysis of risk factors for stage I lung adenocarcinoma using low-dose high-resolution computed tomography. Oncol Lett 16:2483–2489. https://doi.org/10.3892/ol.2018.8921
Feng B, Chen X, Chen Y, Liu K, Li K, Liu X, Yao N, Li Z, Li R, Zhang C, Ji J, Long W (2020a) Radiomics nomogram for preoperative differentiation of lung tuberculoma from adenocarcinoma in solitary pulmonary solid nodule. Eur J Radiol 128:109022. https://doi.org/10.1016/j.ejrad.2020.109022
Feng B, Chen X, Chen Y, Lu S, Liu K, Li K, Liu Z, Hao Y, Li Z, Zhu Z, Yao N, Liang G, Zhang J, Long W, Liu X (2020b) Solitary solid pulmonary nodules: a CT-based deep learning nomogram helps differentiate tuberculosis granulomas from lung adenocarcinomas. Eur Radiol 30:6497–6507. https://doi.org/10.1007/s00330-020-07024-z
Gao C, Xiang P, Ye J, Pang P, Wang S, Xu M (2019) Can texture features improve the differentiation of infiltrative lung adenocarcinoma appearing as ground glass nodules in contrast-enhanced CT? Eur J Radiol 117:126–131. https://doi.org/10.1016/j.ejrad.2019.06.010
Gao J, Han F, Wang X, Duan S, Zhang J (2021) Multi-phase CT-based radiomics nomogram for discrimination between pancreatic serous cystic neoplasm from mucinous cystic neoplasm. Front Oncol 11:699812. https://doi.org/10.3389/fonc.2021.699812
Geneva: World Health Organization (2020) Global tuberculosis report 2020. Licence: CC BY-NC-SA 3.0 IGO. https://www.who.int/tb/publications/global_report/en/. Accessed 15 Oct 2020
Guo Y, Song Q, Jiang M, Guo Y, Xu P, Zhang Y, Fu CC, Fang Q, Zeng M, Yao X (2021) Histological subtypes classification of lung cancers on CT images using 3D deep learning and radiomics. Acad Radiol 28:e258–e266. https://doi.org/10.1016/j.acra.2020.06.010
He L, Huang Y, Ma Z, Liang C, Liang C, Liu Z (2016) Effects of contrast-enhancement, reconstruction slice thickness and convolution kernel on the diagnostic performance of radiomics signature in solitary pulmonary nodule. Sci Rep 6:34921. https://doi.org/10.1038/srep34921
Horvat N, Veeraraghavan H, Khan M, Blazic I, Zheng J, Capanu M, Sala E, Garcia-Aguilar J, Gollub MJ, Petkovska I (2018) MR imaging of rectal cancer: radiomics analysis to assess treatment response after neoadjuvant therapy. Radiology 287:833–843. https://doi.org/10.1148/radiol.2018172300
Hu Y, Zhao X, Zhang J, Han J, Dai M (2021) Value of 18F-FDG PET/CT radiomic features to distinguish solitary lung adenocarcinoma from tuberculosis. Eur J Nucl Med Mol Imaging 48:231–240. https://doi.org/10.1007/s00259-020-04924-6
Khorrami M, Bera K, Thawani R, Rajiah P, Gupta A, Fu P, Linden P, Pennell N, Jacono F, Gilkeson RC, Velcheti V, Madabhushi A (2021) Distinguishing granulomas from adenocarcinomas by integrating stable and discriminating radiomic features on non-contrast computed tomography scans. Eur J Cancer 148:146–158. https://doi.org/10.1016/j.ejca.2021.02.008
Kono R, Fujimoto K, Terasaki H, Muller NL, Kato S, Sadohara J, Hayabuchi N, Takamori S (2007) Dynamic MRI of solitary pulmonary nodules: comparison of enhancement patterns of malignant and benign small peripheral lung lesions. AJR Am J Roentgenol 188:26–36. https://doi.org/10.2214/AJR.05.1446
Lambin P, Leijenaar RTH, Deist TM, Peerlings J, de Jong EEC, van Timmeren J, Sanduleanu S, Larue R, Even AJG, Jochems A, van Wijk Y, Woodruff H, van Soest J, Lustberg T, Roelofs E, van Elmpt W, Dekker A, Mottaghy FM, Wildberger JE, Walsh S (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762. https://doi.org/10.1038/nrclinonc.2017.141
Lee HS, Oh JY, Lee JH, Yoo CG, Lee CT, Kim YW, Han SK, Shim YS, Yim JJ (2004) Response of pulmonary tuberculomas to anti-tuberculous treatment. Eur Respir J 23:452–455. https://doi.org/10.1183/09031936.04.00087304
Leung AN (1999) Pulmonary tuberculosis: the essentials. Radiology 210:307–322. https://doi.org/10.1148/radiology.210.2.r99ja34307
Lin X, Jiao H, Pang Z, Chen H, Wu W, Wang X, Xiong L, Chen B, Huang Y, Li S, Li L (2021) Lung cancer and granuloma identification using a deep learning model to extract 3-dimensional radiomics features in CT imaging. Clin Lung Cancer 22:e756–e766. https://doi.org/10.1016/j.cllc.2021.02.004
Mazzone PJ, Lam L (2022) Evaluating the patient with a pulmonary nodule: a review. JAMA 327:264–273. https://doi.org/10.1001/jama.2021.24287
Nachiappan AC, Rahbar K, Shi X, Guy ES, Mortani Barbosa EJ, Shroff GS, Ocazionez D, Schlesinger AE, Katz SI, Hammer MM (2017) Pulmonary tuberculosis: role of radiology in diagnosis and management. Radiographics 37:52–72. https://doi.org/10.1148/rg.2017160032
National Lung Screening Trial Research T, Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, Gareen IF, Gatsonis C, Marcus PM, Sicks JD (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 365:395–409. https://doi.org/10.1056/NEJMoa1102873
Shen L, Fu H, Tao G, Liu X, Yuan Z, Ye X (2021) Pre-immunotherapy contrast-enhanced CT texture-based classification: a useful approach to non-small cell lung cancer immunotherapy efficacy prediction. Front Oncol 11:591106. https://doi.org/10.3389/fonc.2021.591106
Shi L, Zhao J, Peng X, Wang Y, Liu L, Sheng M (2021) CT-based radiomics for differentiating invasive adenocarcinomas from indolent lung adenocarcinomas appearing as ground-glass nodules: a systematic review. Eur J Radiol 144:109956. https://doi.org/10.1016/j.ejrad.2021.109956
Skoura E, Zumla A, Bomanji J (2015) Imaging in tuberculosis. Int J Infect Dis 32:87–93. https://doi.org/10.1016/j.ijid.2014.12.007
Snoeckx A, Reyntiens P, Desbuquoit D, Spinhoven MJ, Van Schil PE, van Meerbeeck JP, Parizel PM (2018) Evaluation of the solitary pulmonary nodule: size matters, but do not ignore the power of morphology. Insights Imaging 9:73–86. https://doi.org/10.1007/s13244-017-0581-2
Son JY, Lee HY, Kim J-H, Han J, Jeong JY, Lee KS, Kwon OJ, Shim YM (2016) Quantitative CT analysis of pulmonary ground-glass opacity nodules for distinguishing invasive adenocarcinoma from non-invasive or minimally invasive adenocarcinoma: the added value of using iodine mapping. Eur Radiol 26:43–54. https://doi.org/10.1007/s00330-015-3816-y
Sun K, Chen S, Zhao J, Wang B, Yang Y, Wang Y, Wu C, Sun X (2021) Convolutional neural network-based diagnostic model for a solid, indeterminate solitary pulmonary nodule or mass on computed tomography. Front Oncol 11:792062. https://doi.org/10.3389/fonc.2021.792062
Tamponi M, Crivelli P, Montella R, Sanna F, Gabriele D, Poggiu A, Sanna E, Marini P, Meloni GB, Sverzellati N (2021) Exploring the variability of radiomic features of lung cancer lesions on unenhanced and contrast-enhanced chest CT imaging. Phys Med 82:321–331. https://doi.org/10.1016/j.ejmp.2021.02.014
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, Beer DG, Powell CA, Riely GJ, Van Schil PE, Garg K, Austin JH, Asamura H, Rusch VW, Hirsch FR, Scagliotti G, Mitsudomi T, Huber RM, Ishikawa Y, Jett J et al (2011) International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285. https://doi.org/10.1097/JTO.0b013e318206a221
Tsutani Y, Miyata Y, Nakayama H, Okumura S, Adachi S, Yoshimura M, Okada M (2013) Oncologic outcomes of segmentectomy compared with lobectomy for clinical stage IA lung adenocarcinoma: propensity score-matched analysis in a multicenter study. J Thorac Cardiovasc Surg 146:358–364. https://doi.org/10.1016/j.jtcvs.2013.02.008
Wang J, Tang S, Mao Y, Wu J, Xu S, Yue Q, Chen J, He J, Yin Y (2022) Radiomics analysis of contrast-enhanced CT for staging liver fibrosis: an update for image biomarker. Hepatol Int. https://doi.org/10.1007/s12072-022-10326-7
Wu FZ, Huang YL, Wu CC, Tang EK, Chen CS, Mar GY, Yen Y, Wu MT (2016) Assessment of selection criteria for low-dose lung screening CT among Asian ethnic groups in Taiwan: from mass screening to specific risk-based screening for non-smoker lung cancer. Clin Lung Cancer 17:e45–e56. https://doi.org/10.1016/j.cllc.2016.03.004
Wu W, Pierce LA, Zhang Y, Pipavath SNJ, Randolph TW, Lastwika KJ, Lampe PD, Houghton AM, Liu H, Xia L, Kinahan PE (2019) Comparison of prediction models with radiological semantic features and radiomics in lung cancer diagnosis of the pulmonary nodules: a case-control study. Eur Radiol 29:6100–6108. https://doi.org/10.1007/s00330-019-06213-9
Wu Y-J, Wu F-Z, Yang S-C, Tang E-K, Liang C-H (2022) Radiomics in early lung cancer diagnosis: from diagnosis to clinical decision support and education. Diagnostics 12:1064. https://doi.org/10.3390/diagnostics12051064
Xia C, Liu M, Li X, Zhang H, Li X, Wu D, Ren D, Hua Y, Dong M, Liu H, Chen J (2021) Prediction model for lung cancer in high-risk nodules being considered for resection: development and validation in a Chinese population. Front Oncol 11:700179. https://doi.org/10.3389/fonc.2021.700179
Yang X, He J, Wang J, Li W, Liu C, Gao D, Guan Y (2018) CT-based radiomics signature for differentiating solitary granulomatous nodules from solid lung adenocarcinoma. Lung Cancer 125:109–114. https://doi.org/10.1016/j.lungcan.2018.09.013
Yang X, Liu M, Ren Y, Chen H, Yu P, Wang S, Zhang R, Dai H, Wang C (2022) Using contrast-enhanced CT and non-contrast-enhanced CT to predict EGFR mutation status in NSCLC patients-a radiomics nomogram analysis. Eur Radiol 32:2693–2703. https://doi.org/10.1007/s00330-021-08366-y
Zheng Z, Pan Y, Guo F, Wei H, Wu S, Pan T, Li J (2011) Multimodality FDG PET/CT appearance of pulmonary tuberculoma mimicking lung cancer and pathologic correlation in a tuberculosis-endemic country. South Med J 104:440–445. https://doi.org/10.1097/SMJ.0b013e318218700a
Zheng Y-M, Xu W-J, Hao D-P, Liu X-J, Gao C-P, Tang G-Z, Li J, Wang H-X, Dong C (2021) A CT-based radiomics nomogram for differentiation of lympho-associated benign and malignant lesions of the parotid gland. Eur Radiol 31:2886–2895. https://doi.org/10.1007/s00330-020-07421-4
Zhuo Y, Zhan Y, Zhang Z, Shan F, Shen J, Wang D, Yu M (2021) Clinical and CT radiomics nomogram for preoperative differentiation of pulmonary adenocarcinoma from tuberculoma in solitary solid nodule. Front Oncol 11:701598. https://doi.org/10.3389/fonc.2021.701598
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
WZ and ZX: Conceptualization, Methodology, Investigation, Data Curation, Writing—Original Draft. YJ: Methodology, Investigation. KW, XL, and DQ: Resources, Data Curation. MZ: Software, Visualization, Formal analysis. AL: Resources. ZL: Conceptualization, Investigation, Writing—Review and Editing, Supervision, Project administration. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Approval was obtained from the Ethics Committee of the First Affiliated Hospital of Dalian Medical University, the Ethics Committee of the Second Hospital of Dalian Medical University, and Dalian Public Health Clinical Center Ethics Committee. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Consent to participate
Since this study was a retrospective study, the Ethics Committee of the First Affiliated Hospital of Dalian Medical University, the Ethics Committee of the Second Hospital of Dalian Medical University, and the Dalian Public Health Clinical Center Ethics Committee waived the need to obtain informed consent from the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, W., Xiong, Z., Jiang, Y. et al. Radiomics based on enhanced CT for differentiating between pulmonary tuberculosis and pulmonary adenocarcinoma presenting as solid nodules or masses. J Cancer Res Clin Oncol 149, 3395–3408 (2023). https://doi.org/10.1007/s00432-022-04256-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04256-y