Abstract
Purpose
Dissociated response (DR, reduction at baseline or increase < 20% in target lesions compared with nadir in the presence of new lesions) was observed in 20–34% of patients treated with immune checkpoint inhibitors (ICIs). DRs were defined as progression disease (PD) per response evaluation criteria in solid tumors (RECIST v1.1), while evaluation criteria related to immunotherapy incorporated the new lesions into the total tumor burden or conducted further evaluation after 4–8 weeks rather than declaring PD immediately. The main objective of this study is to compare survival between people who continuing initial ICIs treatment and those who switched to other anticancer therapy at the time of DR.
Patients and methods
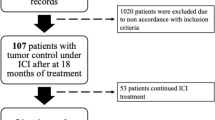
235 patients with advanced lung cancer (LC) treated with ICIs were evaluated. Propensity score matching (PSM) was used to minimize potential confounding factors. Post-DR OS, target lesion changes were evaluated.
Results
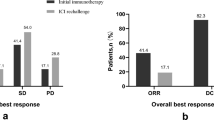
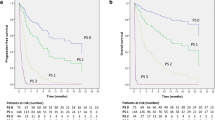
52 patients had been estimated as DRs. After PSM, the continuing ICIs treatment Post-DR cohort still had a significantly longer median post-DR OS than discontinuing ICIs treatment Post-DR cohort, 10.63 months (95% CI 6.27–NA) versus 4.33 months (95% CI 1.77–NA), respectively (p = 0.016).
Conclusion
Within the limitations of this single-center retrospective analysis, clinically stable patients who were judged by clinicians to be eligible for continuing ICIs treatment post-DR derived apparent OS benefit than discontinuing counterpart.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Austin PC (2009) Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med 28:3083–3107. https://doi.org/10.1002/sim.3697
Balar AV, Weber JS (2017) PD-1 and PD-L1 antibodies in cancer: current status and future directions. Cancer Immunol Immunother: CII 66:551–564. https://doi.org/10.1007/s00262-017-1954-6
Beaver JA et al (2018) Patients with melanoma treated with an anti-PD-1 antibody beyond RECIST progression: a US food and drug administration pooled analysis. Lancet Oncol 19:229–239. https://doi.org/10.1016/s1470-2045(17)30846-x
Bohnsack O, Hoos A, Ludajic K (1070padaptation) 1070padaptation of the immune related response criteria: irrecist. Ann Oncol 25:iv369–iv369. https://doi.org/10.1093/annonc/mdu342.23
Borghaei H et al (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373:1627–1639. https://doi.org/10.1056/NEJMoa1507643
Chatterjee M et al (2016) Systematic evaluation of pembrolizumab dosing in patients with advanced non-small-cell lung cancer. Ann Oncol: Off J Eur Soc Med Oncol 27:1291–1298. https://doi.org/10.1093/annonc/mdw174
Chmielowski B (2018) How should we assess benefit in patients receiving checkpoint inhibitor therapy? J Clin Oncol: Off J Am Soc Clin Oncol 36:835–836. https://doi.org/10.1200/jco.2017.76.9885
Escudier B et al (2017) Treatment beyond progression in patients with advanced renal cell carcinoma treated with nivolumab in checkMate 025. Eur Urol 72:368–376. https://doi.org/10.1016/j.eururo.2017.03.037
Ferté C, Marabelle A (2017) iRECIST: a clarification of tumour response assessment in the immunotherapy era. Eur J Cancer 77:165–167. https://doi.org/10.1016/j.ejca.2017.02.015(Oxford, England: 1990)
Gandara DR et al (2018) Atezolizumab treatment beyond progression in advanced NSCLC: results from the randomized phase III OAK study. J Thorac Oncol 13:1906–1918. https://doi.org/10.1016/j.jtho.2018.08.2027
Garon EB et al (2015) Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 372:2018–2028. https://doi.org/10.1056/NEJMoa1501824
George S et al (2016) Safety and efficacy of nivolumab in patients with metastatic renal cell carcinoma treated beyond progression: a subgroup analysis of a randomized clinical trial. JAMA Oncol 2:1179–1186. https://doi.org/10.1001/jamaoncol.2016.0775
Gettinger S et al (2016) Nivolumab monotherapy for first-line treatment of advanced non-small-cell lung cancer. J Clin Oncol: Off J Am Soc Clin Oncol 34:2980–2987. https://doi.org/10.1200/jco.2016.66.9929
Herbst RS et al (2016) Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet (London, England) 387:1540–1550. https://doi.org/10.1016/s0140-6736(15)01281-7
Hodi FS et al (2014) Patterns of response in patients with advanced melanoma treated with Pembrolizumab (MK-3475) and evaluation of immune-related response criteria (irRC). J Immunother Cancer 2:P103–P103. https://doi.org/10.1186/2051-1426-2-S3-P103
Hodi FS et al (2016) Evaluation of immune-related response criteria and RECIST v1.1 in patients with advanced melanoma treated with pembrolizumab. J Clin Oncol: Off J Am Soc Clin Oncol 34:1510–1517. https://doi.org/10.1200/jco.2015.64.0391
Hodi FS et al (2018) Immune-modified response evaluation criteria in solid tumors (imRECIST): refining guidelines to assess the clinical benefit of cancer immunotherapy. J Clin Oncol: Off J Am Soc Clin Oncol 36:850–858. https://doi.org/10.1200/jco.2017.75.1644
Hoos A, Wolchok JD, Humphrey RW, Hodi FS (2015) CCR 20th anniversary commentary: immune-related response criteria-capturing clinical activity in immuno-oncology. Clin Cancer Res: Off J Am Assoc Cancer Res 21:4989–4991. https://doi.org/10.1158/1078-0432.Ccr-14-3128
Horn L et al (2018) First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N Engl J Med 379:2220–2229. https://doi.org/10.1056/NEJMoa1809064
Kanda S et al (2016) Safety and efficacy of nivolumab and standard chemotherapy drug combination in patients with advanced non-small-cell lung cancer: a four arms phase Ib study. Ann Oncol: Off J Eur Soc Med Oncol 27:2242–2250. https://doi.org/10.1093/annonc/mdw416
Kazandjian D, Keegan P, Suzman DL, Pazdur R, Blumenthal GM (2017) Characterization of outcomes in patients with metastatic non-small cell lung cancer treated with programmed cell death protein 1 inhibitors past RECIST version 1.1-defined disease progression in clinical trials. Semin oncology 44:3–7. https://doi.org/10.1053/j.seminoncol.2017.01.001
Liang H, Xu Y, Chen M, Zhong W, Wang M, Zhao J (2020) Patterns of response in metastatic NSCLC during PD-1 or PD-L1 inhibitor therapy: comparison of the RECIST 11 and iRECIST criteria. Thorac Cancer. https://doi.org/10.1111/1759-7714.13367
Long GV et al (2017) Nivolumab for patients with advanced melanoma treated beyond progression: analysis of 2 phase 3 clinical trials. JAMA Oncol 3:1511–1519. https://doi.org/10.1001/jamaoncol.2017.1588
Necchi A et al (2017) Atezolizumab in platinum-treated locally advanced or metastatic urothelial carcinoma: post-progression outcomes from the phase II IMvigor210 study. Ann Oncol: Off J Eur Soc Med Oncol 28:3044–3050. https://doi.org/10.1093/annonc/mdx518
Nishino M (2016) Immune-related response evaluations during immune-checkpoint inhibitor therapy: establishing a “common language” for the new arena of cancer treatment. J Immunother Cancer 4:30. https://doi.org/10.1186/s40425-016-0134-0
Nishino M, Giobbie-Hurder A, Gargano M, Suda M, Ramaiya NH, Hodi FS (2013) Developing a common language for tumor response to immunotherapy: immune-related response criteria using unidimensional measurements. Clin Cancer Res: Off J Am Assoc Cancer Res 19:3936–3943. https://doi.org/10.1158/1078-0432.Ccr-13-0895
Nishino M, Gargano M, Suda M, Ramaiya NH, Hodi FS (2014) Optimizing immune-related tumor response assessment: does reducing the number of lesions impact response assessment in melanoma patients treated with ipilimumab? J Immunother Cancer 2:17. https://doi.org/10.1186/2051-1426-2-17
Nishino M et al (2017) Immune-related tumor response dynamics in melanoma patients treated with pembrolizumab: identifying markers for clinical outcome and treatment decisions. Clin Cancer Res: Off J Am Assoc Cancer Res 23:4671–4679. https://doi.org/10.1158/1078-0432.Ccr-17-0114
Queirolo P, Spagnolo F (2017) Atypical responses in patients with advanced melanoma, lung cancer, renal-cell carcinoma and other solid tumors treated with anti-PD-1 drugs: a systematic review. Cancer Treat Rev 59:71–78. https://doi.org/10.1016/j.ctrv.2017.07.002
Ricciuti B et al (2019) Safety and efficacy of nivolumab in patients with advanced non-small-cell lung cancer treated beyond progression. Clin Lung Cancer 20:178–185.e172. https://doi.org/10.1016/j.cllc.2019.02.001
Rizvi NA et al (2015) Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2, single-arm trial. Lancet Oncol 16:257–265. https://doi.org/10.1016/s1470-2045(15)70054-9
Seymour L et al (2017) iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol 18:e143–e152. https://doi.org/10.1016/s1470-2045(17)30074-8
Tazdait M et al (2018) Patterns of responses in metastatic NSCLC during PD-1 or PDL-1 inhibitor therapy: comparison of RECIST 1.1, irRECIST and iRECIST criteria. Eur J Cancer 88:38–47. https://doi.org/10.1016/j.ejca.2017.10.017
Wolchok JD et al (2009) Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res: Off J Am Assoc Cancer Res 15:7412–7420. https://doi.org/10.1158/1078-0432.Ccr-09-1624
Acknowledgements
The authors would like to thank all the clinical staff in the Department of thoracic oncology, West China Hospital, Sichuan University, who provided great help to this study.
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by HZ, YS, WX, JH, LZ, JS, HW, YW and JZ. The first draft of the manuscript was written by HZ, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of Sichuan University approved this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, H., Sun, Y., Xiu, W. et al. Overall survival benefit of continuing immune checkpoint inhibitors treatment post dissociated response in patients with advanced lung cancer. J Cancer Res Clin Oncol 146, 2979–2988 (2020). https://doi.org/10.1007/s00432-020-03282-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03282-y