Abstract
Purpose
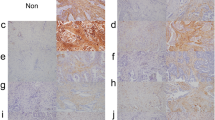
Podoplanin-positive cancer-associated fibroblasts (CAFs) play an essential role in tumor progression. However, it is still unclear whether specific genomic alterations of cancer cells are required to recruit podoplanin-positive CAFs. The aim of this study was to investigate the relationship between the mutation status of lung adenocarcinoma cells and the presence of podoplanin-positive CAFs.
Methods
Ninety-seven lung adenocarcinomas for which whole exome sequencing data were available were enrolled. First, we analyzed the clinicopathological features of the cases, and then, evaluated the relationship between genetic features of cancer cells (major driver mutations and the number of single nucleotide variants, SNVs) and the presence of podoplanin-positive CAFs.
Results
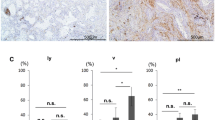
The presence of podoplanin-positive CAFs was associated with smoking history, solid predominant subtype, and lymph node metastasis. We could not find any significant correlations between major genetic mutations (EGFR, KRAS, TP53, MET, ERBB2, BRAF, and PIC3CA) in cancer cells and the presence of podoplanin-positive CAFs. However, cases with podoplanin-positive CAFs had a significantly higher number of SNVs in cancer cells than the podoplanin-negative CAFs cases (median 84 vs 37, respectively; p = 0.001). This was also detected in a non-smoker subgroup (p = 0.037). Multivariate analyses revealed that the number of SNVs in cancer cells was the only statistically significant independent predictor for the presence of podoplanin-positive CAFs (p = 0.044).
Conclusions
In lung adenocarcinoma, the presence of podoplanin-positive CAFs was associated with higher numbers of SNVs in cancer cells, suggesting a relationship between accumulations of SNVs in cancer cells and the generation of a tumor-promoting microenvironment.




Similar content being viewed by others
References
Alexandrov LB, Ju YS, Haase K et al (2016) Mutational signatures associated with tobacco smoking in human cancer. Science 354:618–622. https://doi.org/10.1126/science.aag0299
Bavarva JH, Tae H, McIver L, Garner HR (2014) Nicotine and oxidative stress induced exomic variations are concordant and overrepresented in cancer-associated genes. Oncotarget 5:4788–4798. https://doi.org/10.18632/oncotarget.2033
Bristow RG, Hill RP (2008) Hypoxia and metabolism. Hypoxia, DNA repair and genetic instability. Nat Rev Cancer 8:180–192. https://doi.org/10.1038/nrc2344
Collins MA, Bednar F, Zhang Y et al (2012) Oncogenic Kras is required for both the initiation and maintenance of pancreatic cancer in mice. J Clin Invest 122:639–653. https://doi.org/10.1172/JCI59227
Dimanche-Boitrel MT, Vakaet L, Pujuguet P et al (1994) In vivo and in vitro invasiveness of a rat colon-cancer cell line maintaining E-cadherin expression: an enhancing role of tumor-associated myofibroblasts. Int J Cancer 56:512–521
Gou L-YY, Niu F-YY, Wu Y-LL, Zhong W-ZZ (2015) Differences in driver genes between smoking-related and non-smoking-related lung cancer in the Chinese population. Cancer 121 (Suppl 17):3069–3079. https://doi.org/10.1002/cncr.29531
Hoshino A, Ishii G, Ito T et al (2011) Podoplanin-positive fibroblasts enhance lung adenocarcinoma tumor formation: podoplanin in fibroblast functions for tumor progression. Cancer Res 71:4769–4779. https://doi.org/10.1158/0008-5472.CAN-10-3228
Inoue H, Tsuchiya H, Miyazaki Y et al (2014) Podoplanin expressing cancer-associated fibroblasts in oral cancer. Tumour Biol 35:11345–11352. https://doi.org/10.1007/s13277-014-2450-7
Ito M, Ishii G, Nagai K et al (2012) Prognostic impact of cancer-associated stromal cells in patients with stage I lung adenocarcinoma. Chest 142:151–158. https://doi.org/10.1378/chest.11-2458
Kalluri R, Zeisberg M (2006) Fibroblasts in cancer. Nat Rev Cancer 6:392–401. https://doi.org/10.1038/nrc1877
Kanda Y (2013) Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/10.1038/bmt.2012.244
Kuchnio A, Moens S, Bruning U et al (2015) The cancer cell oxygen sensor PHD2 promotes metastasis via activation of cancer-associated fibroblasts. Cell Rep 12:992–1005. https://doi.org/10.1016/j.celrep.2015.07.010
Luoto KR, Kumareswaran R, Bristow RG (2013) Tumor hypoxia as a driving force in genetic instability. Genome Integr 4:5. https://doi.org/10.1186/2041-9414-4-5
Marsh T, Pietras K, McAllister SS (2013) Fibroblasts as architects of cancer pathogenesis. Biochim Biophys Acta 1832:1070–1078. https://doi.org/10.1016/j.bbadis.2012.10.013
Neri S, Ishii G, Hashimoto H et al (2015) Podoplanin-expressing cancer-associated fibroblasts lead and enhance the local invasion of cancer cells in lung adenocarcinoma. Int J Cancer 137:784–796. https://doi.org/10.1002/ijc.29464
Ono S, Ishii G, Nagai K et al (2013) Podoplanin-positive cancer-associated fibroblasts could have prognostic value independent of cancer cell phenotype in stage I lung squamous cell carcinoma: usefulness of combining analysis of both cancer cell phenotype and cancer-associated fibroblast phenotype. Chest 143:963–970. https://doi.org/10.1378/chest.12-0913
Orimo A, Gupta P, Sgroi D et al (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348. https://doi.org/10.1016/j.cell.2005.02.034
Pfeifer GP, Denissenko MF, Olivier M et al (2002) Tobacco smoke carcinogens, DNA damage and p53 mutations in smoking-associated cancers. Oncogene 21:7435–7451. https://doi.org/10.1038/sj.onc.1205803
Povey JE, Darakhshan F, Robertson K et al (2007) DNA repair gene polymorphisms and genetic predisposition to cutaneous melanoma. Carcinogenesis 28:1087–1093. https://doi.org/10.1093/carcin/bgl257
Rodríguez-Jiménez FJ, Moreno-Manzano V, Lucas-Dominguez R, Sánchez-Puelles J-MM (2008) Hypoxia causes downregulation of mismatch repair system and genomic instability in stem cells. Stem Cells 26:2052–2062. https://doi.org/10.1634/stemcells.2007-1016
Schoppmann SF, Berghoff A, Dinhof C et al (2012) Podoplanin-expressing cancer-associated fibroblasts are associated with poor prognosis in invasive breast cancer. Breast Cancer Res Treat 134:237–244. https://doi.org/10.1007/s10549-012-1984-x
Schoppmann SF, Jesch B, Riegler MF et al (2013) Podoplanin expressing cancer associated fibroblasts are associated with unfavourable prognosis in adenocarcinoma of the esophagus. Clin Exp Metastasis 30:441–446. https://doi.org/10.1007/s10585-012-9549-2
Shan T, Chen S, Chen X et al (2017) Cancer-associated fibroblasts enhance pancreatic cancer cell invasion by remodeling the metabolic conversion mechanism. Oncol Rep 37:1971–1979. https://doi.org/10.3892/or.2017.5479
Suzuki A, Mimaki S, Yamane Y et al (2013) Identification and characterization of cancer mutations in Japanese lung adenocarcinoma without sequencing of normal tissue counterparts. PLoS One 8:e73484. https://doi.org/10.1371/journal.pone.0073484
Tape CJ, Ling S, Dimitriadi M et al (2016) Oncogenic KRAS regulates tumor cell signaling via stromal reciprocation. Cell 165:910–920. https://doi.org/10.1016/j.cell.2016.03.029
Yuan J, Glazer PM (1998) Mutagenesis induced by the tumor microenvironment. Mutat Res 400:439–446
Zhang J, Cao J, Ma S et al (2014) Tumor hypoxia enhances non-small cell lung cancer metastasis by selectively promoting macrophage M2 polarization through the activation of ERK signaling. Oncotarget 5:9664–9677. https://doi.org/10.18632/oncotarget.1856
Funding
This study was supported in part by the Advanced Research for Medical Products Mining Programme of the National Institute of Biomedical Innovation (NIBIO), National Cancer Center Research and Development Fund (25-A-6 and 28-A-9).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
. The association between podoplanin expression in CAFs and number of single nucleotide variants (SNVs) in cancer cells in smoking cases. A, Current smoking cases (n = 30). B, Former smoking cases (n = 24). Supplementary Figure 2. The association between the grade of podoplanin-positive CAFs and number of single nucleotide variants (SNVs) in cancer cells. The grade of expression of podoplanin in the CAFs was defined as follows: 0 was defined as when less than 9% of spindle cells within the tumor stroma were stained for podoplanin. Grade1 was defined as when 10 to 49 % of spindle cells within the tumor stroma were stained for podoplanin. Grade2 was defined as when 50% or more of spindle cells within the tumor stroma were stained for podoplanin. (PPTX 174 KB)
Rights and permissions
About this article
Cite this article
Nakasone, S., Mimaki, S., Ichikawa, T. et al. Podoplanin-positive cancer-associated fibroblast recruitment within cancer stroma is associated with a higher number of single nucleotide variants in cancer cells in lung adenocarcinoma. J Cancer Res Clin Oncol 144, 893–900 (2018). https://doi.org/10.1007/s00432-018-2619-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2619-3