Abstract
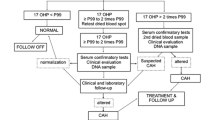
The aim of this study was to assess the effectivity of the identification of patients with congenital adrenal hyperplasia (CAH) in Hungary in the absence of systematic neonatal screening and to estimate the incidence. Dried blood-spot samples of patients clinically suspected at any age to have CAH were collected between 1978 and 1998 throughout the whole country. 17-Hydroxyprogesterone (17-OHP) was measured by radioimmunoassay. Age-specific cut-offs were used. The effectivity of the system was retrospectively assessed. Additional cases were sought to assess the overall incidence of CAH in Hungary. Among the 1,837 patients investigated, 185 cases of CAH were identified. The overall effectivity was 94.7%. The sensitivity and the specificity were 98.9% and 94.2%, respectively. Salt-wasting (SW) boys were, on average, diagnosed 2 weeks later than SW girls, while both boys and girls with the simple virilising (SV) form were diagnosed at similar ages (2 versus 2.5 years). An additional 19 cases were diagnosed during the study period using other methods (plasma and urinary steroid profiles without blood-spot 17-OHP measurements). The incidence of classical CAH in Hungary was 1:14,300 (CI 95% between 1:12,450 and 1:16,795). Presuming that the incidence of CAH is the same among boys and girls, one can calculate that the diagnosis was missed in 24 boys (2 SW, 22 SV). Conclusion: it is possible to identify the vast majority of classical cases of congenital adrenal hyperplasia without a neonatal mass screening programme. However, a significant number of boys with the simple virilising form missed whereas both salt-wasting boys and girls are diagnosed safely.

Similar content being viewed by others
Abbreviations
- CAH :
-
congenital adrenal hyperplasia
- 11β-OHD :
-
11β-hydroxylase deficiency
- 21-OHD :
-
21-hydroxylase deficiency
- 17-OHP :
-
17-hydroxyprogesterone
- NCAH :
-
non-classical congenital adrenal hyperplasia
- RIA :
-
radioimmunoassay
- SV :
-
simple virilising
- SW :
-
salt-wasting
References
Balsamo A, Cacciari E, Piazzi S, Cassio A, Bozza D, Pirazzoli P, Zappulla F (1996) Congenital adrenal hyperplasia: neonatal mass screening compared with clinical diagnosis only in the Emilia-Romagna region of Italy, 1980–1995. Pediatrics 98: 362–367
Brosnan CA, Brosnan P, Therrell BL, Slater CH, Swint JM, Annegers JF, Riley WJ (1998) A comparative cost analysis of newborn screening for classical congenital adrenal hyperplasia in Texas. Public Health Reports 113: 170–178
Brosnan PG, Brosnan CA, Kemp SF, Domek DB, Jelley DH, Blackett PR, Riley WJ (1999) Effect of newborn screening for congenital adrenal hyperplasia. Arch Dis Pediatr Adolesc Med 153: 1272–1278
Donaldson MD, Thomas PH, Love JG, Murray GD, McNich AW, Savage DC (1994) Presentation, acute illness, and learning difficulties in salt wasting 21-hydroxylase deficiency. Arch Dis Child 70: 214–218
Ferenczi A, Garami M, Kiss E, Pék M, Sasvári-Székely M, Barta C, Staub M, Sólyom J, Fekete G (1999) Screening for mutations of 21-hydroxylase gene in Hungarian patients with congenital adrenal hyperplasia. J Clin Endocrinol Metab 84: 2369–2372
Hargitai G, Sólyom J, Battelino T, Lebl J, Pribilincová Z, Hauspie R, Kovács J, Waldhauser F, Frisch H and the MEWPE-CAH study group (2001) Growth patterns and final height in congenital adrenal hyperplasia due to classical 21-hydroxylase deficiency. Horm Res 55: 161–171
Homoki J, Sólyom J, Teller WM (1988) Detection of late onset steroid 21-hydroxylase deficiency by capillary gas chromatographic profiling of urinary steroids in children and adolescents. Eur J Pediatr 147: 257–262
Homoki J, Sólyom J, Wachter U, Teller WM (1992) Urinary excretion of 17-hydroxypregnanolones in patients with different forms of congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency. Eur J Pediatr 151: 24–28
Honour JW, Torresani T (2001) Evaluation of neonatal screening for congenital adrenal hyperplasia. Horm Res 55: 206–211
Joint ESPE/LWPES CAH working group (2002) Consensus statement on 21-hydroxylase deficiency from the European Society for Paediatric Endocrinology and the Lawson Wilkins Pediatric Endocrine Society. Horm Res 58: 188–195
Kovács J, Votava F, Heinze G, Sólyom J, Lebl J, Pribilincová Z, Frisch H, Battelino T, Waldhauser F (2001) Lessons from 30 years of clinical diagnosis and treatment of congenital adrenal hyperplasia in five Middle European countries. J Clin Endocrinol Metab 86: 2958–2964
Kwon C, Farrell PM (2000) The magnitude of false-positive newborn screening test results. Arch Pediatr Adolesc Med 154: 714–718
Pang S, Shook MK (1997) Current status of neonatal screening for congenital adrenal hyperplasia. Curr Opin Pediatr 9: 419–423
Pang S, Hotchkiss J, Drash AL, Levine LS, New MI (1977) Microfilter paper method for 17-hydroxyprogesterone radioimmunoassay, its application for rapid screening for congenital adrenal hyperplasia. J Clin Endocrinol Metab 45: 1003–1008
Sólyom J (1981) Blood-spot 17α-hydroxyprogesterone radioimmunoassay in the follow-up of congenital adrenal hyperplasia. Clin Endocrinol 14: 547–553
Sólyom J, Hughes IA (1989) Value of selective screening for congenital adrenal hyperplasia in Hungary. Arch Dis Child 64: 338–342
Sólyom J, Hammond GL, Vihko R (1979) A method for identification and follow-up of patients with a steroid-21-hydroxylase deficiency. Clin Chim Acta 92: 117–124
Sólyom J, Rácz K, Péter F, Homoki J, Sippel WG, Peter M (2001) Clinical, hormonal and molecular genetic characterization of Hungarian patients with 11β-hydroxylase deficiency. J Endocrine Genet 2: 37–44
Therrell BL (2001) Newborn screening for congenital adrenal hyperplasia. Endocrinol Metab Clin North Am 30: 15–30
Therrell BL, Berenbaum SA, Manter-Kapanke V, Simmank J, Korman K, Prentice L, Gonzalez J, Gunn S (1998) Results of screening 1.9 million Texas newborns for 21-hydroxylase-deficient congenital adrenal hyperplasia. Pediatrics 101: 583–590
Thilén A, Nordenstrom A, Hagenfeldt L, von Dobeln U, Guthenberg C, Larsson A (1998) Benefits of neonatal screening for congenital adrenal hyperplasia (21-hydroxylase deficiency) in Sweden. Pediatrics 101: 694
Van der Kamp HJ, Noordam K, Elvers B, Van Baarle M, Otten BJ, Verkerk PH (2001) Newborn screening for congenital adrenal hyperplasia in the Netherlands. Pediatrics 108: 1320–1324
Virdi NK, Rayner PHW, Rudd BT, Green A (1987) Should we screen for congenital adrenal hyperplasia? A review of 117 cases. Arch Dis Child 62: 659–662
Votava F, Török D, Kovács J, Möslinger D, Sólyom J, Pribilincova Z, Battelino T, Lebl J, Frisch H, Waldhauser F for the MEWPE-CAH (2002) Estimation of the false negative rate in the newborn screening of congenital adrenal hyperplasia. Horm Res 58[Suppl 2]: 98
White PC, Speiser PW (2000) Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Endocr Rev 21: 245–291
Acknowledgements
This work was supported by ETT grant (296/2000) and OTKA grants (T-037559 and T-025178).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Török, D., Eckhardt, G. & Sólyom, J. Twenty years experience in rapid identification of congenital adrenal hyperplasia in Hungary. Eur J Pediatr 162, 844–849 (2003). https://doi.org/10.1007/s00431-003-1311-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-003-1311-y