Abstract
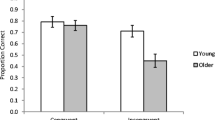
The human angular gyrus (AG) is implicated in recollection, or the ability to retrieve detailed memory content from a specific episode. A separate line of research examining the neural bases of more general mnemonic representations, extracted over multiple episodes, also highlights the AG as a core region of interest. To reconcile these separate views of AG function, the present fMRI experiment used a Remember-Know paradigm with famous (prior knowledge) and non-famous (no prior knowledge) faces to test whether AG activity could be modulated by both task-specific recollection and general prior knowledge within the same individuals. Increased BOLD activity in the left AG was observed during both recollection in the absence of prior knowledge (recollected > non-recollected or correctly rejected non-famous faces) and when prior knowledge was accessed in the absence of experiment-specific recollection (famous > non-famous correct rejections). This pattern was most prominent for the left AG as compared to the broader inferior parietal lobe. Recollection-related responses in the left AG increased with encoding duration and prior knowledge, despite prior knowledge being incidental to the recognition decision. Overall, the left AG appears sensitive to both task-specific recollection and the incidental access of general prior knowledge, thus broadening our notions of the kinds of mnemonic representations that drive activity in this region.





Similar content being viewed by others
Data availability
All data are available from the corresponding authors upon reasonable request. The data are not publicly available at this time as they contain information that could compromise research participant privacy and signed consent.
References
Akrami A, Kopec CD, Diamond ME, Brody CD (2018) Posterior parietal cortex represents sensory history and mediates its effects on behaviour. Nature 554:368–372. https://doi.org/10.1038/nature25510
Amer T, Giovanello KS, Nichol DR, Hasher L, Grady CL (2019) Neural correlates of enhanced memory for meaningful associations with age. Cereb Cortex 29:4568–4579
Andrews-Hanna JR, Reidler JS, Huang C, Buckner RL (2010) Evidence for the default network’s role in spontaneous cognition. J Neurophysiol 104:322–335. https://doi.org/10.1152/jn.00830.2009
Andrews-Hanna JR, Smallwood J, Spreng RN (2014) The default network and self-generated thought: component processes, dynamic control, and clinical relevance. Ann N Y Acad Sci 1316:29–52
Axelrod V, Rees G, Bar M (2017) The default network and the combination of cognitive processes that mediate self-generated thought. Nat Hum Behav 1(12):896–910
Bellana B, Liu Z, Anderson JA, Moscovitch M, Grady CL (2016) Laterality effects in functional connectivity of the angular gyrus during rest and episodic retrieval. Neuropsychologia 80:24–34. https://doi.org/10.1016/j.neuropsychologia.2015.11.004
Bellana B, Liu Z-X, Diamond NB, Grady CL, Moscovitch M (2017) Similarities and differences in the default mode network across rest, retrieval, and future imagining. Hum Brain Mapp 38:1155–1171. https://doi.org/10.1002/hbm.23445
Bellana B, Mansour R, Ladyka-Wojcik N, Grady CL, Moscovitch M (2021) The influence of prior knowledge on the formation of detailed and durable memories. J Mem Lang 121:104264. https://doi.org/10.1016/j.jml.2021.104264
Ben-Zvi S, Soroker N, Levy DA (2015) Parietal lesion effects on cued recall following pair associate learning. Neuropsychologia 73:176–194
Ben-Zvi Feldman S, Soroker N, Levy DA (2021) Lesion-behaviour mapping reveals multifactorial neurocognitive processes in recognition memory for unfamiliar faces. Neuropsychologia 163:108078. https://doi.org/10.1016/j.neuropsychologia.2021.108078
Berryhill ME (2012) Insights from neuropsychology: pinpointing the role of the posterior parietal cortex in episodic and working memory. Front Integr Neurosci 6:1–12. https://doi.org/10.3389/fnint.2012.00031/abstract
Binder JR, Desai RH (2011) The neurobiology of semantic memory. Trends Cogn Sci 15:527–536
Binder J, Frost J, Hammeke T, Bellgowan PSF, Rao SM, Cox RW (1999) Conceptual processing during the conscious resting state: a functional MRI study. J Cogn Neurosci 11:80–93. https://doi.org/10.1162/089892999563265
Binder JR, Desai RH, Graves WW, Conant LL (2009) Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex 19:2767–2796. https://doi.org/10.1016/j.tics.2011.10.001
Bonner MF, Peelle JE, Cook PA, Grossman M (2013) Heteromodal conceptual processing in the angular gyrus. Neuroimage 71:175–186
Bonnici HM, Richter FR, Yazar Y, Simons JS (2016) Multimodal feature integration in the angular gyrus during episodic and semantic retrieval. J Neurosci 36:5462–5471. https://doi.org/10.1523/JNEUROSCI.4310-15.2016
Brown TI, Rissman J, Chow TE, Uncapher MR, Wagner AD (2018) Differential medial temporal lobe and parietal cortical contributions to real-world autobiographical episodic and autobiographical semantic memory. Sci Rep 8:1–14
Bruce V, Young A (1986) Understanding face recognition. Br J Psychol 77:305–327
Cabeza R, Ciaramelli E, Moscovitch M (2012a) Cognitive contributions of the ventral parietal cortex: an integrative theoretical account. Trends Cogn Sci 16:338–352
Cabeza R, Ciaramelli E, Moscovitch M (2012b) Response to Nelson et al.: ventral parietal subdivisions are not incompatible with an overarching function. Trends Cogn Sci 16:400–401. https://doi.org/10.1016/j.tics.2012.06.015
Campbell K, Grigg O, Saverino C, Churchill N, Grady C (2013) Age differences in the intrinsic functional connectivity of default network subsystems. Front Hum Neurosci 5:73
Capotosto P, Baldassarre A, Sestieri C, Spadone S, Romani GL, Corbetta M (2016) Task and regions specific top-down modulation of alpha rhythms in parietal cortex. Cereb Cortex 2:1–8. https://doi.org/10.1093/cercor/bhw278
Caspers S, Geyer S, Schleicher A, Mohlberg H, Amunts K, Zilles K (2006) The human inferior parietal cortex: cytoarchitectonic parcellation and interindividual variability. Neuroimage 33:430–448
Caspers S, Schleicher A, Bacha-Trams M, Palomero-Gallagher N, Amunts K, Zilles K (2013) Organization of the human inferior parietal lobule based on receptor architectonics. Cereb Cortex 23:615–628. https://doi.org/10.1093/cercor/bhs048
Chen J, Honey CJ, Simony E, Arcaro MJ, Norman K (2016) Accessing real-life episodic information from minutes versus hours earlier modulates hippocampal and high-order cortical dynamics. Cereb Cortex 26:3428–3441. https://doi.org/10.1093/cercor/bhv155
Christoff K, Gordon AM, Smallwood J, Smith R, Schooler JW (2009) Experience sampling during fMRI reveals default network and executive system contributions to mind wandering. Proc Natl Acad Sci U S A 106:8719–8724
Ciaramelli E, Grady CL, Moscovitch M (2008) Top-down and bottom-up attention to memory: a hypothesis (AtoM) on the role of the posterior parietal cortex in memory retrieval. Neuropsychologia 46:1828–1851
Ciaramelli E, Grady C, Levine B, Ween J, Moscovitch M (2010) Top-down and bottom-up attention to memory are dissociated in posterior parietal cortex: Neuroimaging and neuropsychological evidence. J Neurosci 30:4943–4956
Ciaramelli E, Faggi G, Scarpazza C, Mattioli F, Spaniol J, Ghetti S, Moscovitch M (2017) Subjective recollection independent from multifeatural context retrieval following damage to the posterior parietal cortex. Cortex 2:1–12
Ciaramelli E, Burianová H, Vallesi A, Cabeza R, Moscovitch M (2020) Functional interplay between posterior parietal cortex and hippocampus during detection of memory targets and non-targets. Front Neurosci 14:563768
Cox RW (1996) AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res 29:162–173
Davidson PSR, Anaki D, Ciaramelli E, Cohn M, Alice SN, Murphy KJ, Troyer AK, Moscovitch M, Levine B (2008) Does lateral parietal cortex support episodic memory? Evidence from focal lesion patients. Neuropsychologia 46:1743–1755
Denkova E, Botzung A, Manning L (2006) Neural correlates of remembering/knowing famous people: an event-related fMRI study. Neuropsychologia 44:2783–2791
Duke D, Martin CB, Bowles B, Mcrae K, Köhler S (2017) Perirhinal cortex tracks degree of recent as well as cumulative lifetime experience with object concepts. Cortex 89:61–70. https://doi.org/10.1016/j.cortex.2017.01.015
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25:1325–1335
Ellamil M, Fox KCR, Dixon ML, Pritchard S, Todd RM, Thompson E, Christoff K (2016) Dynamics of neural recruitment surrounding the spontaneous arising of thoughts in experienced mindfulness practitioners. Neuroimage 136:186–196. https://doi.org/10.1016/j.neuroimage.2016.04.034
Fairhall SL, Caramazza A (2013) Brain regions that represent amodal conceptual knowledge. J Neurosci 33:10552–10558. https://doi.org/10.1523/JNEUROSCI.0051-13.2013
Fernández G, Morris RGM (2018) Memory, novelty and prior knowledge. Trends Neurosci 41:654–659. https://doi.org/10.1016/j.tins.2018.08.006
Fernandino L, Binder JR, Desai RH, Pendl SL, Humphries CJ, Gross WL, Conant LL, Seidenberg MS (2015) Concept representation reflects multimodal abstraction: a framework for embodied semantics. Cereb Cortex 2:1–17. https://doi.org/10.1093/cercor/bhv020
Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van EDC, Raichle ME (2005) The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci 102:9673–9678
Fox KCR, Spreng RN, Ellamil M, Andrews-Hanna JR, Christoff K (2015) The wandering brain: meta-analysis of functional neuroimaging studies of mind-wandering and related spontaneous thought processes. Neuroimage 111:611–621. https://doi.org/10.1016/j.neuroimage.2015.02.039
Frithsen A, Miller MB (2014) The posterior parietal cortex: comparing remember/know and source memory tests of recollection and familiarity. Neuropsychologia 2:1–14
Geschwind N (1972) Language and the brain. Sci Am 226:76–83
Gilboa A, Marlatte H (2017) Neurobiology of schemas and schema-mediated memory. Trends Cogn Sci 21:618–631
Gilmore AW, Nelson SM, McDermott KB (2015) A parietal memory network revealed by multiple MRI methods. Trends Cogn Sci 19:1–10
Gorgolewski KJ, Lurie D, Urchs S, Kipping JA, Craddock RC, Milham MP, Margulies DS, Smallwood J (2014) A correspondence between individual differences in the brain’s intrinsic functional architecture and the content and form of self-generated thoughts. PLoS One 9(5):e97176
Graves WW, Boukrina O, Mattheiss SR, Alexander EJ, Baillet S (2017) Reversing the standard neural signature of the word-nonword distinction. J Cogn Neurosci 29:79–94. https://doi.org/10.1162/jocn_a_01022
Guerin SA, Miller MB (2009) Lateralization of the parietal old/new effect: an event-related fMRI study comparing recognition memory for words and faces. Neuroimage 44:232–242. https://doi.org/10.1016/j.neuroimage.2008.08.035
Guerin SA, Miller MB (2011) Parietal cortex tracks the amount of information retrieved even when it is not the basis of a memory decision. Neuroimage 55:801–807. https://doi.org/10.1016/j.neuroimage.2010.11.066
Hasson U, Chen J, Honey CJ (2015) Hierarchical process memory: memory as an integral component of information processing. Trends Cogn Sci 19:304–313
Hower KH, Wixted J, Berryhill ME, Olson IR (2014) Impaired perception of mnemonic oldness, but not mnemonic newness, after parietal lobe damage. Neuropsychologia 56:409–417
Huijbers W, Pennartz CM, Cabeza R, Daselaar SM (2009) When learning and remembering compete: a functional MRI study. PLoS Biol 7:e11
Huijbers W, Vannini P, Sperling RA, Cabeza R, Daselaar SM (2012) Explaining the encoding/retrieval flip: Memory-related deactivations and activations in the posteromedial cortex. Neuropsychologia 50:3764–3774. https://doi.org/10.1016/j.neuropsychologia.2012.08.021
Humphreys GF, Lambon Ralph MA (2014) Fusion and fission of cognitive functions in the human parietal cortex. Cereb Cortex 25:3547–3560
Humphreys GF, Ralph MAL (2017) Mapping domain-selective and counterpointed domain-general higher cognitive functions in the lateral parietal cortex: evidence from fMRI comparisons of difficulty-varying semantic versus visuo-spatial tasks, and functional connectivity analyses. Cereb Cortex 27:4199–4212
Humphreys GF, Lambon Ralph MA, Simons JS (2021) A unifying account of angular gyrus contributions to episodic and semantic cognition. Trends Neurosci 44:452–463. https://doi.org/10.1016/j.tins.2021.01.006
Humphreys GF, Jung J, Lambon Ralph MA (2022) The convergence and divergence of episodic and semantic functions across lateral parietal cortex. Cereb Cortex. https://doi.org/10.1093/cercor/bhac044
Hutchinson JB, Uncapher MR, Weiner KS, Bressler DW, Silver M, Preston AR, Wagner AD (2014) Functional heterogeneity in posterior parietal cortex across attention and episodic memory retrieval. Cereb Cortex 24:49–66
Irish M, Piguet O (2013) The pivotal role of semantic memory in remembering the past and imagining the future. Front Behav Neurosci 7:1–11. https://doi.org/10.3389/fnbeh.2013.00027/abstract
Irish M, Vatansever D (2020) Rethinking the episodic-semantic distinction from a gradient perspective. Curr Opin Behav Sci 32:43–49. https://doi.org/10.1016/j.cobeha.2020.01.016
Jonker TR, Dimsdale-Zucker H, Ritchey M, Clarke A, Ranganath C (2018) Neural reactivation in parietal cortex enhances memory for episodically linked information. Proc Natl Acad Sci 115(43):11084–11089
Kaefer K, Stella F, McNaughton BL, Battaglia FP (2022) Replay, the default mode network and the cascaded memory systems model. Nat Rev Neurosci 23:628–640
Kim H (2016) Default network activation during episodic and semantic memory retrieval: a selective meta-analytic comparison. Neuropsychologia 80:35–46
Knecht S, Deppe M, Dräger B, Bobe L, Lohmann H, Ringelstein EB, Henningsen H (2000) Language lateralization in healthy right-handers. Brain 123:74–81
Konishi M, McLaren DG, Engen H, Smallwood J (2015) Shaped by the past: the default mode network supports cognition that is independent of immediate perceptual input. PLoS ONE 10:e0132209. https://doi.org/10.1371/journal.pone.0132209
Korkki SM, Richter FR, Gellersen HM, Simons JS (2022) Reduced memory precision in older age is associated with functional and structural differences in the angular gyrus. bioRxiv:2022.05.26.493542
Kuhl BA, Chun MM (2014) Successful remembering elicits event-specific activity patterns in lateral parietal cortex. J Neurosci 34:8051–8060. https://doi.org/10.1523/JNEUROSCI.4328-13.2014
Lee H, Kuhl BA (2016) Reconstructing perceived and retrieved faces from activity patterns in lateral parietal cortex. J Neurosci 36:6069–6082. https://doi.org/10.1523/JNEUROSCI.4286-15.2016
Lee H, Samide R, Richter FR, Kuhl BA (2018) Decomposing parietal memory reactivation to predict consequences of remembering. Cereb Cortex 2:1–14
Leiker EK, Johnson JD (2014) Neural reinstatement and the amount of information recollected. Brain Res 1582:125–138
Levy DA (2012) Towards an understanding of parietal mnemonic processes: some conceptual guideposts. Front Integr Neurosci 6:41
Liu Z-X, Grady C, Moscovitch M (2016) Effects of prior-knowledge on brain activation and connectivity during associative memory encoding. Cereb Cortex 2:47
Margulies DS, Ghosh SS, Goulas A, Falkiewicz M, Huntenburg JM, Langs G, Bezgin G, Eickhoff SB, Castellanos FX, Petrides M, Jefferies E, Smallwood J (2016) Situating the default-mode network along a principal gradient of macroscale cortical organization. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.1608282113
Mason MF, Norton MI, Van HJD, Wegner DM, Grafton ST, Macrae CN (2007) Wandering minds: stimulus-independent thought. Science 315:393–395
McClelland JL, McNaughton BL, O’Reilly RC (1995) Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychol Rev 102:419–457
Mesulam M-M (1998) From sensation to cognition. Brain 121:1013–1052
Murphy C, Jefferies E, Rueschemeyer S-A, Sormaz M, Wang H, Margulies DS, Smallwood J (2018) Distant from input: evidence of regions within the default mode network supporting perceptually-decoupled and conceptually-guided cognition. Neuroimage 171:393–401
Nelson SM, Cohen AL, Power JD, Wig GS, Miezin FM, Wheeler ME, Velanova K, Donaldson DI, Phillips JS, Schlaggar BL, Petersen SE (2010) A parcellation scheme for human left lateral parietal cortex. Neuron 67:156–170. https://doi.org/10.1016/j.neuron.2010.05.025
Nelson SM, Arnold KM, Gilmore AW, McDermott KB (2013) Neural signatures of test-potentiated learning in parietal cortex. J Neurosci 33:11754–11762
Nilakantan AS, Bridge DJ, Gagnon EP, Vanhaerents SA, Voss JL (2017) Stimulation of the posterior cortical-hippocampal network enhances precision of memory. Curr Biol. https://doi.org/10.1016/j.cub.2016.12.042
Oligschläger S, Huntenburg JM, Golchert J, Lauckner ME, Bonnen T, Margulies DS (2017) Gradients of connectivity distance are anchored in primary cortex. Brain Struct Funct 222:2173–2182. https://doi.org/10.1007/s00429-016-1333-7
Price CJ (2010) The anatomy of language: a review of 100 fMRI studies published in 2009. Ann N Y Acad Sci 1191:62–88
Price AR, Bonner MF, Grossman M (2015a) Semantic memory: cognitive and neuroanatomical perspectives. Elsevier Inc, Amsterdam. https://doi.org/10.1016/B978-0-12-397025-1.00280-3
Price AR, Bonner MF, Peelle JE, Grossman M (2015b) Converging evidence for the neuroanatomic basis of combinatorial semantics in the angular gyrus. J Neurosci 35:3276–3284
Price AR, Peelle JE, Bonner MF, Grossman M, Hamilton RH (2016) Causal evidence for a mechanism of semantic integration in the angular gyrus as revealed by high-definition transcranial direct current stimulation. J Neurosci 36:3829–3838. https://doi.org/10.1523/JNEUROSCI.3120-15.2016
Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL (2001) A default mode of brain function. Proc Natl Acad Sci U S A 98:676–682
Ramanan S, Bellana B (2019) A domain-general role for the angular gyrus in retrieving internal representations of the external world. J Neurosci 39:2978–2980. https://doi.org/10.1523/JNEUROSCI.3231-18.2019
Ramanan S, Piguet O, Irish M (2017) Rethinking the role of the angular gyrus in remembering the past and imagining the future: the contextual integration model. Neurosci. https://doi.org/10.1177/1073858417735514
Ramon M, Gobbini MI (2017) Familiarity matters: a review on prioritized processing of personally familiar faces. Vis Cogn. https://doi.org/10.1080/13506285.2017.1405134
Renoult L, Rugg MD (2020) An historical perspective on Endel Tulving’s episodic-semantic distinction. Neuropsychologia 139:2
Renoult L, Davidson P, Palombo DJ, Moscovitch M, Levine B (2012) Personal semantics: at the crossroads of semantic and episodic memory. Trends Cogn Sci 16:550–558
Renoult L, Davidson PSR, Schmitz E, Park L, Campbell K, Moscovitch M, Levine B (2015) Autobiographically significant concepts: more episodic than semantic in nature? An electrophysiological investigation of overlapping types of memory. J Cogn Neurosci 27:57–72. https://doi.org/10.1162/jocn_a_00689
Renoult L, Irish M, Moscovitch M, Rugg MD (2019) From knowing to remembering: the semantic-episodic distinction. Trends Cogn Sci 23:1041–1057. https://doi.org/10.1016/j.tics.2019.09.008
Richter FR, Cooper RA, Bays PM, Simons JS (2016) Distinct neural mechanisms underlie the success, precision, and vividness of episodic memory. Elife 5:1–18. https://doi.org/10.7554/eLife.18260
Rissman J, Chow TE, Reggente N, Wagner AD (2016) Decoding fMRI signatures of real-world autobiographical memory retrieval. J Cogn Neurosci. https://doi.org/10.1162/jocn_a_00409
Robin J, Garzon L, Moscovitch M (2019) Spontaneous memory retrieval varies based on familiarity with a spatial context. Cognition 190:81–92
Rugg MD, King DR (2018) Ventral lateral parietal cortex and episodic memory retrieval. Cortex 107:238–250
Rugg MD, Vilberg KL (2013) Brain networks underlying episodic memory retrieval. Curr Opin Neurobiol 23:255–260
Seghier ML (2013) The angular gyrus: Multiple functions and multiple subdivisions. Neurosci 19:43–61
Sestieri C, Shulman GL, Corbetta M (2017) The contribution of the human posterior parietal cortex to episodic memory. Nat Rev Neurosci 18:183–192. https://doi.org/10.1038/nrn.2017.6
Shimamura AP (2011) Episodic retrieval and the cortical binding of relational activity. Cogn Affect Behav Neurosci 11:277–291
Shimamura AP (2014) Remembering the past: neural substrates underlying episodic encoding and retrieval. Curr Dir Psychol Sci 23:257–263. https://doi.org/10.1177/0963721414536181
Simons JS, Peers PV, Mazuz YS, Berryhill ME, Olson IR (2010) Dissociation between memory accuracy and memory confidence following bilateral parietal lesions. Cereb Cortex 20:479–485. https://doi.org/10.1093/cercor/bhp116
Simons JS, Ritchey M, Fernyhough C (2022) Brain mechanisms underlying the subjective experience of remembering. Annu Rev Psychol 73:2. https://doi.org/10.1146/annurev-psych-030221-025439
Skipper-Kallal LM, Mirman D, Olson IR (2015) Converging evidence from fMRI and aphasia that the left temporoparietal cortex has an essential role in representing abstract semantic knowledge. Cortex 69:104–120. https://doi.org/10.1016/j.cortex.2015.04.021
Smallwood J, Schooler JW (2015) The science of mind wandering: empirically navigating the stream of consciousness. Ann Rev Psychol 66:487–518
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TEJ, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, De Stefano N, Brady JM, Matthews PM (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23:S208–S219
Spreng RN (2012) The fallacy of a “task-negative” network. Front Psychol 3:1–5
Spreng RN, DuPre E, Selarka D, Garcia J, Gojkovic S, Mildner J, Luh W-M, Turner GR (2014) Goal-congruent default network activity facilitates cognitive control. J Neurosci 34:14108–14114
Squire LR (1986) Mechanisms of memory. Science 232:1612–1619
Stawarczyk D, Majerus S, Maquet P, D’Argembeau A (2011) Neural correlates of ongoing conscious experience: both task-unrelatedness and stimulus-independence are related to default network activity. PLoS ONE 6:2
St-Laurent M, Abdi H, Buchsbaum BR (2015) Distributed patterns of reactivation predict vividness of recollection. J Cogn Neurosci 27:2000–2018
Thakral PP, Wang TH, Rugg MD (2015) Cortical reinstatement and the confidence and accuracy of source memory. Neuroimage 109:118–129. https://doi.org/10.1016/j.neuroimage.2015.01.003
Thakral PP, Madore KP, Schacter DL (2017a) A role for the left angular gyrus in episodic simulation and memory. J Neurosci 37:1319–1417. https://doi.org/10.1523/JNEUROSCI.1319-17.2017
Thakral PP, Wang TH, Rugg MD (2017b) Decoding the content of recollection within the core recollection network and beyond. Cortex 91:101–113. https://doi.org/10.1016/j.cortex.2016.12.011
Tibon R, Fuhrmann D, Levy DA, Simons JS, Henson R (2019) Multimodal integration and vividness in the angular gyrus during episodic encoding and retrieval. J Neurosci 39:2102–2118. https://doi.org/10.1523/JNEUROSCI.2102-18.2018
Trinkler I, King JA, Doeller CF, Rugg MD, Burgess N (2009) Neural bases of autobiographical support for episodic recollection of faces. Hippocampus 19:718–730
Tulving E (1972) Episodic and semantic memory. Organ Mem 1:381–403
Tulving E (1985) Memory and consciousness. Can Psychol Can 26:1–12. https://doi.org/10.1037/h0080017
Uddin LQ, Supekar K, Amin H, Rykhlevskaia E, Nguyen DA, Greicius MD, Menon V (2010) Dissociable connectivity within human angular gyrus and intraparietal sulcus: evidence from functional and structural connectivity. Cereb Cortex 20(11):2636–2646
Van Calster L, D’Argembeau A, Salmon E, Peters F, Majerus S (2016) Fluctuations of attentional networks and default mode network during the resting state reflect variations in cognitive states: evidence from a novel resting-state experience sampling method. J Cogn Neurosci 26:1–19. https://doi.org/10.1162/jocn_a_01025
van der Linden M, Berkers RMWJ, Morris RGM, Fernández G (2017) Angular gyrus involvement at encoding and retrieval is associated with durable but less specific memories. J Neurosci 2:3603–3616. https://doi.org/10.1523/JNEUROSCI.3603-16.2017
Vatansever D, Smallwood J, Jefferies E (2021) Varying demands for cognitive control reveals shared neural processes supporting semantic and episodic memory retrieval. Nat Commun 12:2
Vilberg KL, Rugg MD (2008) Memory retrieval and the parietal cortex: a review of evidence from a dual-process perspective. Neuropsychologia 46:1787–1799. https://doi.org/10.1016/j.neuropsychologia.2008.01.004
Vilberg KL, Rugg MD (2009a) Left parietal cortex is modulated by amount of recollected verbal information. NeuroReport 20:1295–1299. https://doi.org/10.1097/WNR.0b013e3283306798
Vilberg KL, Rugg MD (2009b) Functional significance of retrieval-related activity in lateral parietal cortex: evidence from fMRI and ERPs. Hum Brain Mapp 30:1490–1501. https://doi.org/10.1002/hbm.20618
Wagner AD, Shannon BJ, Kahn I, Buckner RL (2005) Parietal lobe contributions to episodic memory retrieval. Trends Cogn Sci 9:445–453
Wagner IC, Van BM, Kroes MCWW, Gutteling TP, Van Der LM, Morris RG, Fernández G, van Buuren M, Kroes MCWW, Gutteling TP, van der Linden M, Morris RG, Fernández G (2015) Schematic memory components converge within angular gyrus during retrieval. Elife 4:1–28. https://doi.org/10.7554/eLife.09668
Wang JX, Rogers LM, Gross EZ, Ryals AJ, Dokucu ME, Brandstatt KL, Hermiller MS, Voss JL (2014) Targeted enhancement of cortical-hippocampal brain networks and associative memory. Science 345:1054–1057. https://doi.org/10.1126/science.1252900
Willenbockel V, Sadr J, Fiset D, Horne GO, Gosselin F, Tanaka JW (2010) Controlling low-level image properties: the SHINE toolbox. Behav Res Methods 42:671–684
Winocur G, Moscovitch M, Bontempi B (2010) Memory formation and long-term retention in humans and animals: convergence towards a transformation account of hippocampal-neocortical interactions. Neuropsychologia 48:2339–2356
Xia M, Wang J, He Y (2013) BrainNet viewer: a network visualization tool for human brain connectomics. PLoS ONE 8:2
Yarkoni T, Poldrack RA, Nichols TE, Van Essen DC, Wager TD (2011) Large-scale automated synthesis of human functional neuroimaging data. Nat Methods 8(8):665–670
Yazar Y, Bergström ZM, Simons JS (2017) Reduced multimodal integration of memory features following continuous theta burst stimulation of angular gyrus. Brain Stimul 2:1–16
Yonelinas AP (2002) The nature of recollection and familiarity: a review of 30 years of research. J Mem Lang 46:441–517
Acknowledgements
The authors would like to thank Rania Mansour for her help with stimulus collection, Ali Golestani and Priya Abraham for their assistance at the Toronto Neuroimaging (ToNI) centre, and Marilyne Ziegler for her assistance with Eprime. The authors would also like to thank Bradley Buchsbaum, Michael D. Rugg, Zhong-Xu Liu, Nick Diamond, Vincent Man, Jessica Robin, Tarek Amer and Iva Brunec for helpful discussions about these data.
Funding
This work was supported by a Natural Sciences and Engineering Research Council (NSERC) grant (No. A8347 to M.M.), a Canadian Institute for Health Research (CIHR) Foundation Grant (No. MOP-143311 to C.L.G.), and scholarships awarded from NSERC and the Ontario Graduate Scholarship program (to B.B.).
Author information
Authors and Affiliations
Contributions
CRediT authorship contribution statement: BB: conceptualization; investigation; formal analysis; writing—original draft; writing—review and editing. NL-W: investigation; writing—review and editing. SL: investigation; writing—review and editing. MM: conceptualization; writing—review and editing; funding acquisition. CLG: conceptualization; writing—review and editing; funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bellana, B., Ladyka-Wojcik, N., Lahan, S. et al. Recollection and prior knowledge recruit the left angular gyrus during recognition. Brain Struct Funct 228, 197–217 (2023). https://doi.org/10.1007/s00429-022-02597-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-022-02597-5