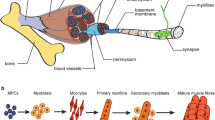
Abstract
It is now over 30 years since Bodo Christ first demonstrated that the musculature of the limb originated from the somites and overturned the then prevailing view that limb muscle develops from a local source. Subsequently, using electron microscopy and histological procedures, Bodo Christ identified that cells of the somites undergo an epithelial to mesenchymal transition which enabled them to move from their paraxial point of origin to distal locations. These studies defined this translocation as one of the major mechanisms allowing myogenic cells to translocate around the body. The other means used to translocate muscle involves the movement of cells as a sheet. The deployment of one of these two mechanisms has been postulated to be involved in the formation of all the hypaxial musculature of the vertebrate body. In this paper we describe the formation of muscles both in the head and in the body, which use a translocatory mechanism during their development. We highlight recent data showing that muscle translocation is a far more complex process than first thought but which in itself can be used as a valuable tool to address questions regarding tissue patterning and development.

Similar content being viewed by others
References
Borue X, Noden DM (2004) Normal and aberrant craniofacial myogenesis by grafted trunk somitic and segmental plate mesoderm. Development 131:3967–3980
Brand-Saberi B, Christ B (1999) Genetic and epigenetic control of muscle development in vertebrates. Cell Tissue Res 296:199–212
Brand-Saberi B, Muller TS, Wilting J, Christ B, Birchmeier C (1996) Scatter factor/hepatocyte growth factor (SF/HGF) induces emigration of myogenic cells at interlimb level in vivo. Dev Biol 179:303–308
Burke AC, Nowicki JL (2003) A new view of patterning domains in the vertebrate mesoderm. Dev Cell 4:159–165
Chevallier A (1975) Role of the somitic mesoderm in the development of the rib cage of bird embryos. I. Origin of the sternal component and conditions for the development of the ribs (author’s transl). J Embryol Exp Morphol 33:291–311
Chevallier A (1979) Role of the somitic mesoderm in the development of the thorax in bird embryos. II. Origin of thoracic and appendicular musculature. J Embryol Exp Morphol 49:73–88
Chevallier A, Kieny M, Mauger A (1977) Limb-somite relationship: origin of the limb musculature. J Embryol Exp Morphol 41:245–258
Chevallier A, Kieny M, Mauger A (1978) Limb-somite relationship: effect of removal of somitic mesoderm on the wing musculature. J Embryol Exp Morphol 43:263–278
Christ B, Ordahl CP (1995) Early stages of chick somite development. Anat Embryol (Berl) 191:381–396
Christ B, Jacob HJ, Jacob M (1977) Experimental analysis of the origin of the wing musculature in avian embryos. Anat Embryol (Berl) 150:171–186
Christ B, Jacob M, Jacob HJ (1983) On the origin and development of the ventrolateral abdominal muscles in the avian embryo. An experimental and ultrastructural study. Anat Embryol (Berl) 166:87–101
Cossu G, Kelly R, Tajbakhsh S, Di Donna S, Vivarelli E, Buckingham M (1996) Activation of different myogenic pathways: myf-5 is induced by the neural tube and MyoD by the dorsal ectoderm in mouse paraxial mesoderm. Development 122:429–437
Couly GF, Coltey PM, Le Douarin NM (1993) The triple origin of skull in higher vertebrates: a study in quail-chick chimeras. Development 117:409–429
Dietrich S (1999) Regulation of hypaxial muscle development. Cell Tissue Res 296:175–182
Dietrich S, Schubert FR, Healy C, Sharpe PT, Lumsden A (1998) Specification of the hypaxial musculature. Development 125:2235–2249
Evans DJ (2003) Contribution of somitic cells to the avian ribs. Dev Biol 256:114–126
Evans DJ, Noden DM (2006) Spatial relations between avian craniofacial neural crest and paraxial mesoderm cells. Dev Dyn 235:1310–1325
Galis F (2001) Evolutionary history of vertebrate appendicular muscle. Bioessays 23:383–387
Hayashi K, Ozawa E (1995) Myogenic cell migration from somites is induced by tissue contact with medial region of the presumptive limb mesoderm in chick embryos. Development 121:661–669
Heymann S, Koudrova M, Arnold H, Koster M, Braun T (1996) Regulation and function of SF/HGF during migration of limb muscle precursor cells in chicken. Dev Biol 180:566–578
Huang R, Zhi Q, Izpisua-Belmonte JC, Christ B, Patel K (1999) Origin and development of the avian tongue muscles. Anat Embryol (Berl) 200:137–152
Jacob M, Christ B, Jacob HJ (1979) The migration of myogenic cells from the somites into the leg region of avian embryos. An ultrastructural study. Anat Embryol (Berl) 157:291–309
Jones CL (1979) The morphogenesis of the thigh of the mouse with special reference to tetrapod muscle homologies. J Morphol 162:275–309
Kardon G (1998) Muscle and tendon morphogenesis in the avian hind limb. Development 125:4019–4032
Kardon G, Campbell JK, Tabin CJ (2002) Local extrinsic signals determine muscle and endothelial cell fate and patterning in the vertebrate limb. Dev Cell 3:533–545
Kardon G, Harfe BD, Tabin CJ (2003) A Tcf4-positive mesodermal population provides a prepattern for vertebrate limb muscle patterning. Dev Cell 5:937–944
Kontges G, Lumsden A (1996) Rhombencephalic neural crest segmentation is preserved throughout craniofacial ontogeny. Development 122:3229–3242
Mackenzie S, Walsh FS, Graham A (1998) Migration of hypoglossal myoblast precursors. Dev Dyn 213:349–358
Matsuoka T, Ahlberg PE, Kessaris N, Iannarelli P, Dennehy U, Richardson WD, McMahon AP, Koentges G (2005) Neural crest origins of the neck and shoulder. Nature 436:347–355
Mootoosamy RC, Dietrich S (2002) Distinct regulatory cascades for head and trunk myogenesis. Development 129:573–583
Myokai F, Washio N, Asahara Y, Yamaai T, Tanda N, Ishikawa T, Aoki S, Kurihara H, Murayama Y, Saito T (1995) Expression of the hepatocyte growth factor gene during chick limb development. Dev Dyn 202:80–90
Noden DM (1983) The embryonic origins of avian cephalic and cervical muscles and associated connective tissues. Am J Anat 168:257–276
Noden DM (1986) Patterning of avian craniofacial muscles. Dev Biol 116:347–356
Noden DM, Trainor PA (2005) Relations and interactions between cranial mesoderm and neural crest populations. J Anat 207:575–601
Noden DM, Marcucio R, Borycki AG, Emerson CP Jr (1999) Differentiation of avian craniofacial muscles: I. Patterns of early regulatory gene expression and myosin heavy chain synthesis. Dev Dyn 216:96–112
Olivera-Martinez I, Coltey M, Dhouailly D, Pourquie O (2000) Mediolateral somitic origin of ribs and dermis determined by quail-chick chimeras. Development 127:4611–4617
Ordahl CP, Le Douarin NM (1992) Two myogenic lineages within the developing somite. Development 114:339–353
Ordahl CP, Williams BA, Denetclaw W (2000) Determination and morphogenesis in myogenic progenitor cells: an experimental embryological approach. Curr Top Dev Biol 48:319–367
Parkyn G, Mootoosamy RC, Cheng L, Thorpe C, Dietrich S (2002) Hypaxial muscle development. Results Probl Cell Differ 38:127–141
Pourquie O, Coltey M, Breant C, Le Douarin NM (1995) Control of somite patterning by signals from the lateral plate. Proc Natl Acad Sci USA 92:3219–3223
Prunotto C, Crepaldi T, Forni PE, Ieraci A, Kelly RG, Tajbakhsh S, Buckingham M, Ponzetto C (2004) Analysis of Mlc-lacZ Met mutants highlights the essential function of Met for migratory precursors of hypaxial muscles and reveals a role for Met in the development of hyoid arch-derived facial muscles. Dev Dyn 231:582–591
Rees E, Young RD, Evans DJ (2003) Spatial and temporal contribution of somitic myoblasts to avian hind limb muscles. Dev Biol 253:264–278
Seno T (1961) An experimental study on the formation of the body wall in the chick. Acta Anat (Basel) 45:60–82
Trainor PA, Tam PP (1995) Cranial paraxial mesoderm and neural crest cells of the mouse embryo: co-distribution in the craniofacial mesenchyme but distinct segregation in branchial arches. Development 121:2569–2582
Tremblay P, Dietrich S, Mericskay M, Schubert FR, Li Z, Paulin D (1998) A crucial role for Pax3 in the development of the hypaxial musculature and the long-range migration of muscle precursors. Dev Biol 203:49–61
Valasek P, Evans DJ, Maina F, Grim M, Patel K (2005) A dual fate of the hindlimb muscle mass: cloacal/perineal musculature develops from leg muscle cells. Development 132:447–458
Vasyutina E, Stebler J, Brand-Saberi B, Schulz S, Raz E, Birchmeier C (2005) CXCR4 and Gab1 cooperate to control the development of migrating muscle progenitor cells. Genes Dev 19:2187–2198
Wahl CM, Noden DM, Baker R (1994) Developmental relations between sixth nerve motor neurons and their targets in the chick embryo. Dev Dyn 201:191–202
Wilson-Rawls J, Hurt CR, Parsons SM, Rawls A (1999) Differential regulation of epaxial and hypaxial muscle development by paraxis. Development 126:5217–5229
Acknowledgments
We would like to thank Anthony Otto for critical review of the manuscript. We are indebted to the Wellcome Trust (grant 077750) for providing funds enabling this work. We apologise to those authors whose work has not been cited in full. This is due to constraints in manuscript length.
Author information
Authors and Affiliations
Corresponding author
Additional information
Darrell J.R. Evans, Petr Valasek, Corina Schmidt have contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Evans, D.J.R., Valasek, P., Schmidt, C. et al. Skeletal muscle translocation in vertebrates. Brain Struct Funct 211 (Suppl 1), 43–50 (2006). https://doi.org/10.1007/s00429-006-0121-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-006-0121-1