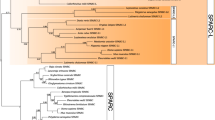
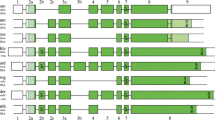
Abstract
The African bichir (Polypterus senegalus) is a living representative of Polypteriformes. P. senegalus possesses teeth composed of dentin covered by an enameloid cap and a layer of collar enamel on the tooth shaft, as in lepisosteids. A thin layer of enamel matrix can also be found covering the cap enameloid after its maturation and during the collar enamel formation. Teleosts fish do not possess enamel; teeth are protected by cap and collar enameloid, and inversely in sarcopterygians, where teeth are only covered by enamel, with the exception of the cap enameloid in teeth of larval urodeles. The presence of enameloid and enamel in the teeth of the same organism is an opportunity to solve the evolutionary history of the presence of enamel/enameloid in basal actinopterygians. In silico analyses of the jaw transcriptome of a juvenile bichir provided twenty SCPP transcripts. They included enamel, dentin, and bone-specific SCPPs known in sarcopterygians and several actinopterygian-specific SCPPs. The expression of these 20 genes was investigated by in situ hybridizations on jaw sections during tooth and dentary bone formation. A spatiotemporal expression patterns were established and compared with previous studies of SCPP gene expression during enamel/enameloid and bone formation. Similarities and differences were highlighted, and several SCPP transcripts were found specifically expressed during tooth or bone formation suggesting either conserved or new functions of these SCPPs.



Similar content being viewed by others
Data availability
DNA Sequences are accessible in genbank; Genbank accession numbers: (names given in 2016 in Genbank by Sire et al.)
AMBN: KX833883
ENAM: KX923546
AMTN: KX833904
ODAM: KX833888
ODAM-like: KX833889
SCPP-PQ1: KX833901
DSPP: KX833884
IBSP: KX833886
MEPE: KX833887
SPP1: KX833902
SCPP1: KX833890; L.o: KU189281
SCPP3: KX833891 (=SCPP3A); P.s C: KX833892 (=SCPP3C); KX833903 (=SCPP3B
SCPP5: KX833893
SCPP7: KX833896
SCPP7-Like: KX833897
SCPP9: KX833898; L.o: KU189286
SCPP5-like: KX833895
SCPP-LPQ15: KX833900
Abbreviations
- AMBN :
-
ameloblastin
- AMEL :
-
amelogenin
- AMTN :
-
amelotin
- bp :
-
base pairs
- DMP1 :
-
dentin matrix acidic phosphoprotein 1
- DSPP :
-
dentin sialophosphoprotein
- DSPPL1 :
-
dentin sialophosphoprotein-like 1
- EMP :
-
enamel matrix protein
- ENAM :
-
enamelin
- FPKM :
-
fragments per kilobase of exon per million fragments mapped
- IBSP :
-
Integrin binding sialophosphoprotein
- KLK :
-
kallikrein
- MABT :
-
maleic acid buffer tween
- MA :
-
million years
- MEPE :
-
matrix extracellular phosphoglycoprotein
- MMP20 :
-
matrix metalloproteinase 20
- NBT/BCIP :
-
nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolylphosphate
- NGSP :
-
nested gene specific primer
- No :
-
number
- NTM :
-
NaCL, TrisHCl, MgCl2
- ODAM :
-
odontogenic ameloblast-associated protein
- PBS :
-
phosphate buffered saline
- PCR :
-
polymerase chain reaction
- RACE :
-
rapid amplification of cDNA end
- RGD :
-
arg gly-asp domain
- RT-PCR :
-
reverse transcription polymerase chain reaction
- SCPP :
-
secretory calcium-binding phosphoprotein
- SCPP-LPQ :
-
secretory calcium-binding phosphoprotein-like, proline and glutamine rich
- SCPPPQ1 :
-
secretory calcium-binding phosphoprotein, proline and glutamine rich 1
- SPP1 :
-
secreted phosphoprotein 1
- SSC :
-
saline sodium citrate
- UTR :
-
untranslated region
References
Addison WN, Nakano Y, Loisel T, Crine P, McKee MD (2008) MEPE-ASARM peptides control extracellular matrix mineralization by binding to hydroxyapatite: an inhibition regulated by PHEX cleavage of ASARM. J Bone Miner Res 23:1638–1649
Aguiar VRC, Castelli EC, Single RM, Bashirova A, Ramsuran V, Kulkarni S, Augusto DG, Martin MP, Gutierrez-Arcelus M, Carrington M, Meyer D (2023) Comparison between qPCR and RNA-seq reveals challenges of quantifying HLA expression. Immunogenetics 75(3):249–262
Assaraf-Weill N, Gasse B, Al-Hashimi N, Delgado S, Sire J-Y, Davit-Béal T (2013) Conservation of amelogenin gene expression during tetrapod evolution. J Exp Zool B Mol Dev Evol 320:200–209
Assaraf-Weill N, Gasse B, Silvent J, Bardet C, Sire J-Y, Davit-Béal T (2014) Ameloblasts express type I collagen during amelogenesis. J Dent Res 93:502–507
Bartlett JD, Ganss B, Goldberg M, Moradian-Oldak J, Paine ML, Snead ML et al (2006) Protein-protein interactions of the developing enamel matrix. Curr Top Dev Biol 74:57–115
Berkovitz B, Shellis P (2018) The Teeth of Mammalian Vertebrates; Academic Press: Amsterdam, The Netherlands; Elsevier: New York, NY, USA
Bianco P, Fisher LW, Young MF, Termine JD, Robey PG (1991) Expression of bone sialoprotein (BSP) in developing human tissues. Calcif Tissue Int 49:421–426
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120
Braasch I, Gehrke AR, Smith JJ, Kawasaki K, Manousaki T, Pasquier J et al (2016) The spotted gar genome illuminates vertebrate evolution and facilitates human-teleost comparisons. Nat Genet 48:427–437
Chen S, Rani S, Wu Y, Unterbrink A, Gu TT, Gluhak-Heinrich J et al (2005) Differential regulation of dentin sialophosphoprotein expression by Runx2 during odontoblast cytodifferentiation. J Biol Chem 280:29717–29727
Chibon P, Roux J-P, Spinelli M (1971) Presence de 1’email dans les dents des Amphibiens Urodèles et Anoures. Etude autoradiographique et ultrastructurale avant et apres la metamorphose. CR Acad Sci (Paris) 272:1–3
Coenye T (2021) Do results obtained with RNA-sequencing require independent verification? Biofilm 3:100043
Delgado S, Casane D, Bonnaud L, Laurin M, Sire J-Y, Girondot M (2001) Molecular evidence for Precambrian origin of amelogenin, the major protein of vertebrate enamel. Mol Biol Evol 18:2146–2153
Delgado S, Couble M-L, Magloire H, Sire J-Y (2006) Cloning, sequencing, and expression of the amelogenin gene in two scincid lizards. J Dent Res 85:138–143
Dohi N, Murakami C, Tanabe T, Yamakoshi Y, Fukae M, Yamamoto Y et al (1998) Immunocytochemical and immunochemical study of enamelins, using antibodies against porcine 89-kDa enamelin and its N-terminal synthetic peptide, in porcine tooth germs. Cell Tissue Res 293:313–325
D’Souza SE, Ginsberg MH, Plow EF (1991) Arginyl-glycyl-aspartic acid (RGD): a cell adhesion motif. Trends Biochem Sci 16:246–250
D’Souza RN, Cavender A, Sunavala G, Alvarez J, Ohshima T, Kulkarni AB et al (1997) Gene expression patterns of murine dentin matrix protein 1 (Dmp1) and dentin sialophosphoprotein (DSPP) suggest distinct developmental functions in vivo. J Bone Miner Res 2:2040–2049
Fearnhead RW (1979) Matrix--mineral relationships in enamel tissues. J Dent Res 58:909–921
Fong CD, Cerný R, Hammarström L, Slaby I (1998) Sequential expression of an amelin gene in mesenchymal and epithelial cells during odontogenesis in rats. Eur J Oral Sci 106(Suppl 1):324–330
Fraser GJ, Cerny R, Soukup V, Bronner-Fraser M, Streelman JT (2010) The odontode explosion: the origin of tooth-like structures in vertebrates. BioEssays 32:808–817
Ganss B, Abbarin N (2014) Maturation and beyond: proteins in the developmental continuum from enamel epithelium to junctional epithelium. Front Physiol 5:371
Gasse B, Chiari Y, Silvent J, Davit-Béal T, Sire J-Y (2015) Amelotin: an enamel matrix protein that experienced distinct evolutionary histories in amphibians, sauropsids and mammals. BMC Evol Biol 15:47
Gasse B, Sire J-Y (2015) Comparative expression of the four enamel matrix protein genes, amelogenin, ameloblastin, enamelin and amelotin during amelogenesis in the lizard Anolis carolinensis. EvoDevo 6:29
George A, Sabsay B, Simonian PA, Veis A (1993) Characterization of a novel dentin matrix acidic phosphoprotein. Implications for induction of biomineralization. J Biol Chem 268:12624–12630
Germain D, Meunier F (2019) Teeth of extant polypteridae and amiidae have plicidentine organization. Acta Zool 100(2):119–125
Gibson CW, Yuan ZA, Hall B, Longenecker G, Chen E, Thyagarajan T et al (2001) Amelogenin-deficient mice display an amelogenesis imperfecta phenotype. J Biol Chem 276:31871–31875
Gordon JAR, Tye CE, Sampaio AV, Underhill TM, Hunter GK, Goldberg HA (2007) Bone sialoprotein expression enhances osteoblast differentiation and matrix mineralization in vitro. Bone 41:462–473
Gouy M, Guindon S, Gascuel O (2010) SeaView version 4: a multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol Biol Evol 27(2):221–224
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Haas BJ, Papanicolaou A, Yassour M, Grabherr M, Blood PD, Bowden J et al (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8(8):1494–1512
Hu C-C, Hart TC, Dupont BR, Chen JJ, Sun X, Qian Q et al (2000) Cloning human enamelin cDNA, chromosomal localization, and analysis of expression during tooth development. J Dent Res 79:912–919
Huysseune A, Takle H, Soenens M, Taerwe K, Witten PE (2008) Unique and shared gene expression patterns in Atlantic salmon (Salmo salar) tooth development. Dev Genes Evol 218:427–437
Irisarri I, Baurain D, Brinkmann H, Delsuc F, Sire J-Y, Kupfer A, Petersen J, Jarek M, Meyer A, Vences M, Philippe H (2017) Phylotranscriptomic consolidation of the jawed vertebrate timetree. Nat Ecol Evol 1(9):1370–1378
Iwasaki K, Bajenova E, Somogyi-Ganss E, Miller M, Nguyen V, Nourkeyhani H et al (2005) Amelotin--a novel secreted, ameloblast-specific protein. J Dent Res 84:1127–1132
Kawasaki K, Shimoda S, Fukae M (1987) Histological and biochemical observations of developing enameloid of the sea bream. Adv Dent Res 1:191–195
Kawasaki K, Weiss KM (2003) Mineralized tissue and vertebrate evolution: the secretory calcium-binding phosphoprotein gene cluster. Proc Natl Acad Sci U S A 100:4060–4065
Kawasaki K, Suzuki T, Weiss KM (2005) Phenogenetic drift in evolution: the changing genetic basis of vertebrate teeth. Proc Natl Acad Sci U S A 102:18063–18068
Kawasaki K, Weiss KM (2006) Evolutionary genetics of vertebrate tissue mineralization: the origin and evolution of the secretory calcium-binding phosphoprotein family. J Exp Zool B Mol Dev Evol 306(3):295–316
Kawasaki K, Weiss KM (2008) SCPP gene evolution and the dental mineralization continuum. J Dent Res 87:520–531
Kawasaki K (2009) The SCPP gene repertoire in bony vertebrates and graded differences in mineralized tissues. Dev Genes Evol 219:147–157
Kawasaki K (2013) Odontogenic ameloblast-associated protein (ODAM) and amelotin: major players in hypermineralization of enamel and enameloid. J Oral Biosci 55:85–90
Kawasaki K, Amemiya CT (2014) SCPP genes in the coelacanth: tissue mineralization genes shared by sarcopterygians. J Exp Zool B Mol Dev Evol 322(6):390–402
Kawashima N, Okiji T (2016) Odontoblasts: specialized hard-tissue-forming cells in the dentin-pulp complex. Congenit Anom 56:144–153
Kawasaki K, Mikami M, Nakatomi M, Braasch I, Batzel PH, Postlethwait J, Sato A, Sasagawa I, Ishiyama M (2017) SCPP genes and their relatives in gar: rapid expansion of mineralization genes in osteichthyans. J Exp Zool B Mol Dev Evol 328(7):645–665
Kawasaki K, Keating JN, Nakatomi M, Welten M, Mikami M, Sasagawa I, Puttick MN, Donoghue PCJ, Ishiyama M (2021) Coevolution of enamel, ganoin, enameloid, and their matrix SCPP genes in osteichthyans. Iscience 24(1):102023
Keating JN, Marquart CL, Marone F et al (2018) The nature of aspidin and the evolutionary origin of bone. Nat Ecol Evol 2:1501–1506
Kerr T (1958) Development and structure of some actinopterygian and urodele teeth. Proc Zool Soc Lond 133:401–422
Kumar S, Stecher G, Suleski M, Hedges B (2017) TimeTree: a resource for timelines, Timetrees, and divergence times. Mol Biol Evol 34(7):1812–1819
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory- efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25
Langmead B, Salzberg S (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359
Lee SK, Krebsbach PH, Matsuki Y, Nanci A, Yamada KM, Yamada Y (1996) Ameloblastin expression in rat incisors and human tooth germs. Int J Dev Biol 40:1141–1150
MacDougall M, Gu TT, Luan X, Simmons D, Chen J (1998) Identification of a novel isoform of mouse dentin matrix protein 1: spatial expression in mineralized tissues. J Bone Miner Res 13:422–431
MacDougall M, Simmons D, Gu TT, Forsman-Semb K, Kärrman Mårdh C, Mesbah M et al (2000) Cloning, characterization and immunolocalization of human ameloblastin. Eur J Oral Sci 108:303–310
MacDougall M, Simmons D, Gu TT, Dong J (2002) MEPE/OF45, a new dentin/bone matrix protein and candidate gene for dentin diseases mapping to chromosome 4q21. Connect Tissue Res 43:320–330
Mackie EJ (2003) Osteoblasts: novel roles in orchestration of skeletal architecture. Int J Biochem Cell Biol 35:1301–1305
McKee MD, Farach-Carson MC, Butler WT, Hauschka PV, Nanci A (1993) Ultrastructural immunolocalization of noncollagenous (osteopontin and osteocalcin) and plasma (albumin and alpha 2HS-glycoprotein) proteins in rat bone. J Bone Miner Res 8:485–496
Malaval L, Aubin JE, Vico L (2009) Role of the small integrin-binding ligand N-linked glycoprotein (SIBLING), bone sialoprotein (BSP) in bone development and remodeling. Osteoporos Int 20:1077–1080
Marks SC, Odgren PR (2002) Structure and development of the skeleton. Princ Bone Biol 1:3–15
Mitsiadis TA, Filatova A, Papaccio G, Goldberg M, About I, Papagerakis P (2014) Distribution of the amelogenin protein in developing, injured and carious human teeth. Front Physiol 5:477
Mikami M, Ineno T, Thompson AW, Braasch I, Ishiyama M, Kawasaki K (2022) Convergent losses of SCPP genes and ganoid scales among non-teleost actinopterygians. Gene 811:146091
Moffatt P, Smith CE, St-Arnaud R, Nanci A (2008) Characterization of Apin, a secreted protein highly expressed in tooth-associated epithelia. J Cell Biochem 103:941–956
Moffatt P, Wazen RM, Neves JDS, Nanci A (2014) Characterisation of secretory calcium-binding phosphoprotein-proline-glutamine-rich 1: a novel basal lamina component expressed at cell-tooth interfaces. Cell Tissue Res 358:843–855
Murdock D, Dong XP, Repetski J et al (2013) The origin of conodonts and of vertebrate mineralized skeletons. Nature 502:546–549
Nampei A, Hashimoto J, Hayashida K, Tsuboi H, Shi K, Tsuji I et al (2004) Matrix extracellular phosphoglycoprotein (MEPE) is highly expressed in osteocytes in human bone. J Bone Miner Metab 22:176–184
Nanci A, Zalzal S, Lavoie P, Kunikata M, Chen W, Krebsbach PH et al (1998) Comparative immunochemical analyses of the developmental expression and distribution of ameloblastin and amelogenin in rat incisors. J Histochem Cytochem 46:911–934
Oida S, Nagano T, Yamakoshi Y, Ando H, Yamada M, Fukae M (2002) Amelogenin gene expression in porcine odontoblasts. J Dent Res 81:103–108
Papagerakis P, MacDougall M, Hotton D, Bailleul-Forestier I, Oboeuf M, Berdal A (2003) Expression of amelogenin in odontoblasts. Bone 32:228–240
Park J-C, Park J-T, Son H-H, Kim H-J, Jeong M-J, Lee C-S et al (2007) The amyloid protein APin is highly expressed during enamel mineralization and maturation in rat incisors. Eur J Oral Sci 115:153–160
Peyer B (1968) Osteichthyes. In: Zangerl R (ed) Comparative odontology. University of Chicago Press, New-York and London, pp 80–110
Poole D (1967) Phylogeny of tooth tissues: Enameloid and enamel in recent vertebrates with a note in the history of cementum. Miles AEW, Struct Chem Organ Teeth, New York, pp 111–149
Poole D (1971) An introduction to the phylogeny of calcified tissues. In: Dahlberg AA (ed) Dental morphology and evolution. University of Chicago Press
Qu Q, Haitina T, Zhu M, Ahlberg PE (2015) New genomic and fossil data illuminate the origin of enamel. Nature 526:108–111
Rafidi K, Simkina I, Johnson E, Moore MA, Gerstenfeld LC (1994) Characterization of the chicken osteopontin-encoding gene. Gene 140:163–169
Roux JP, Chibon P (1973a) Étude ultrastructurale de l’amélogenèse chez la larve du triton Pleurodeles waltlii (Amphibien Urodèle). J Biol Buccale 1:33–44
Roux JP, Chibon P (1973b) Relationship between metamorphosis and thickness of the enamel layer on teeth in urodele amphibians. CR Hebd Séances Acad Sci 276(10):1613–1615
Sasagawa I (1997) Fine structure of the cap enameloid and of the dental epithelial cells during enameloid mineralisation and early maturation stages in the tilapia, a teleost. J Anat 190(Pt4):589–600
Sasagawa I, Ishiyama M, Yokosuka H, Mikami M, Uchida T (2009) Tooth enamel and enameloid in actinopterygian fish. Front Mater Sci 3:174
Sasagawa I, Yokosuka H, Ishiyama M, Mikami M, Shimokawa H, Uchida T (2012) Fine structural and immunohistochemical detection of collar enamel in the teeth of Polypterus senegalus, an actinopterygian fish. Cell Tissue Res 347:369–381
Sasagawa I, Ishiyama M, Yokosuka H, Mikami M (2013) Teeth and ganoid scales in Polypterus and Lepisosteus, the basic actinopterygian fish: an approach to understand the origin of the tooth enamel. J Oral Biosci 55:76–84
Shellis RP, Miles AEW (1974) Autoradiographic study of the formation of enameloid and dentine matrices in teleost fishes using tritiated amino acids. Proc R Soc Lond B Biol Sci 185(1078):51–72
Shintani S, Kobata M, Toyosawa S, Ooshima T (2006) Expression of ameloblastin during enamel formation in a crocodile. J Exp Zoolog B Mol Dev Evol 306B:126–133
Silvent J, Sire J-Y, Delgado S (2013) The dentin matrix acidic phosphoprotein 1 (DMP1) in the light of mammalian evolution. J Mol Evol 76:59–70
Sire JY, Davit-Béal T, Delgado S, Gu X (2007) The origin and evolution of enamel mineralization genes. Cells Tissues Organs 186(1):25–48
Sire JY, Donoghue PC, Vickaryous MK (2009) Origin and evolution of the integumentary skeleton in non-tetrapod vertebrates. J Anat 214(4):409–440
Sire JY, Kawasaki K (2012) Origin and evolution of bone a nd dentin and of acidic secretory calcium-binding phosphoproteins. In: Phosphorylated extracelular matrix proteins of bone and dentin. Bentham Science Publishers LtdEditors: M Goldberg, pp 3–58
Smith MM, Miles AEW (1971) The ultrastructure of odontogenesis in larval and adult urodeles; differentiation of the dental epithelial cells. Z Zellforsch Mikrosk Anat 121(4):470–498
Smith MM, Hall BK (1990) Development and evolutionary origins of vertebrate skeletogenic and odontogenic tissues. Biol Rev 65:277–373
Smith MM (1992) Microstructure and evolution of enamel amongst osteichthyan fishes and early tetrapods. In: Structure, function, and evolution of teeth. Freund Publishing House, pp 73–101
Smith MM (1995) Heterochrony in the evolution of enamel in vertebrates. In: Evolutionary change and heterochrony, pp 125–150
Smith MM, Sansom I, Smith M (1996) ‘Teeth’ before armour: The earliest vertebrate mineralized tissues. Mod Geol 20:303–319
Termine JD, Belcourt AB, Christner PJ, Conn KM, Nylen MU (1980) Properties of dissociatively extracted fetal tooth matrix proteins. I. Principal molecular species in developing bovine enamel. J Biol Chem 255:9760–9768
Trueb B, Taeschler S, Schild C, Lang NP (2007) Expression of phosphoproteins and amelotin in teeth. Int J Mol Med 19:49–54
Vandenplas S, De Clercq A, Huysseune A (2014) Tooth replacement without a dental lamina: the search for epithelial stem cells in Polypterus senegalus. J Exp Zool B Mol Dev Evol 322(5):281–93. https://doi.org/10.1002/jez.b.22577
Wilhelm BT, Landry JR (2009) RNA-Seq-quantitative measurement of expression through massively parallel RNA-sequencing. Methods. 48(3):249–257
Young G, Karatajute-Talimaa V, Smith MM (1996) A possible Late Cambrian vertebrate from Australia. Nature. 383:810–812
Acknowledgements
We thank Marie-Claire Lajarille for paraffin section cutting. We are grateful to the Roscoff Bioinformatics platform ABiMS (http://abims.sb-roscoff.fr), part of the Institut Français de Bioinformatique (ANR-11-INBS-0013) and BioGenouest network, for providing computing and storage resources….
Funding
This project was financially supported by Sorbonne Université, CNRS, and ANR-12-BSV7-020, project “Jaws”.
Author information
Authors and Affiliations
Contributions
Fernandez-Trujillo MA and Silvent J carried out the molecular biology experiments. Liu X, Corre E and Sire JY performed in silico data recovery. Fernandez-Trujillo MA, Silvent J, Sire JY conceived and designed the experiments, and wrote the manuscript. Houée G and Delgado S wrote the manuscript and critically revised the manuscript. All authors read and approved the final manuscript. Houée G and Delgado S corrected, submitted the manuscript. Thanks to François Meunier for his corrections and proofreading of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
All animal experiments conformed to the directives of the European parliament and of the council of 22 September 2010 on the protection of animals used for scientific purposes (Directive 2010/63/EU) and the French Rural Code (Article R214-87 to R214-137, Decree no. 2013-118 of 1st February 2013). Certificate of authorization for vertebrate animal experiment was obtained under the no. 75-600.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Matthias Hammerschmidt
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary file 1
Supplementary material S1. Forward and reverse primer sequences used for normal PCR in order to build probes for in situ hybridization (ISH) and to check the 20 cDNA sequences identified in the jaw transcriptome of Polypterus senegalus. RACE PCR was used to complete the ENAM and DSPP sequences at the 3' and 5' extremities. The expected length (base pairs) of the probe is indicated between brackets. Two values represent the length of variants 1 and 2, respectively. GenBank accession numbers are indicated. (DOCX 15 kb)
Supplementary file 2
Supplementary material S2. The 20 SCPPs identified in the jaw transcriptome of Polypterus senegalus (P.s). The amino acid sequences of 18 of them are aligned with the homologous sequence (the larger sequence when two variants have been revealed, i.e. in ENAM, SCPP5 and SCPP9) of the spotted gar Lepisosteus oculatus (L.o). The two other bichir SCPPs, SCPP5-like (not found in the submitted spotted gar sequences) and SCPP-LPQ15 (only a short, partial sequence in spotted gar) are presented alone. Signal peptides are underlined and SxE and RGD motifs are highlighted in grey. (.): identical residue to P.s amino acid; (-) gap inserted for alignment purpose; (?): unknown residue. GenBank accession numbers for each sequence is indicated. (DOCX 26 kb)

Supplementary file 3
Supplementary material S3. A) In situ hybridization showing no expression of SCPPs during tooth development. B) Expression of SCPP5 variant 1 and SCPP9 variant 2 transcripts. (PNG 818 kb). Scale bars = 50 μm.
Supplementary file 4
Supplementary material S4. Adapted from Mikami et al. (2022). SCPP genes identified in the Senegal bichir genome from Mikami et al. (2022). The 20 genes identified in jaw transcriptome are Surrounded in grey. Scpps coding for P/Q rich proteins are in red, Scpps coding for acidic proteins are in blue. (DOCX 22 kb)
Supplementary file 5
Supplementary material S5 (DOCX 14 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Delgado, S., Fernandez-Trujillo, M., Houée, G. et al. Expression of 20 SCPP genes during tooth and bone mineralization in Senegal bichir. Dev Genes Evol 233, 91–106 (2023). https://doi.org/10.1007/s00427-023-00706-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-023-00706-w