Abstract
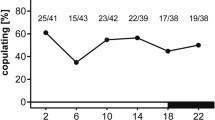
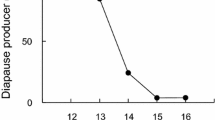
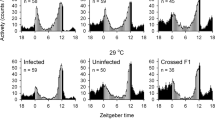
The short day lengths of late summer in moderate regions are used to induce diapause in various insects. Many studies have shown the maternal effect of photoperiod on diapause induction of Trichogramma wasps, but there is no study to show the relationship between photoperiodic regimes and clock genes in these useful biological control agents. Here, we investigated the role of photoperiods on diapause, fecundity, and clock gene expression (clk, cyc, cry2, per, and timeout) in asexual and sexual Trichogramma brassicae as a model insect to find any differences between two strains. Asexual strain was infected by Wolbachia, an endosymbiont bacterium. The diapause percentage was significantly higher under short days (8 h in sexual and 12 h in the asexual T. brassicae), although the diapause percentage of the sexual strain was significantly higher than the asexual one in all the photoperiods. The ANOVA revealed no significant changes between different photoperiods in the clock gene expression in the sexual strain but significant photoperiodic changes in clk, cyc, and timeout in the asexual strain. Our results showed that the mRNA levels of clock genes of asexual T. brassicae were significantly lower than those of sexual strain. The fecundity was significantly higher in the asexual strain. These results suggest that Wolbachia infection makes disturbance on the clock gene expression which consequently reduces the percentage of diapause but increases the fecundity in asexual T. brassicae.





Similar content being viewed by others
References
Babendreier D, Kuske S, Bigler F (2003) Overwintering of the egg parasitoid Trichogramma brassicae in Northern Switzerland. BioControl 48(3):261–273. https://doi.org/10.1023/A:1023661420247
Bertossa RC, van de Zande L, Beukeboom LW, Beersma DGM (2014) Phylogeny and oscillating expression of period and cryptochrome in short and long photoperiods suggest a conserved function in Nasonia vitripennis. Chronobiol Int 31(6):749–760. https://doi.org/10.3109/07420528.2014.880451
Bian G, Xu Y, Lu P, Xie Y, Xi Z (2010) The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog 6(4):e1000833. https://doi.org/10.1371/journal.ppat.1000833
Boivin G (1994) Overwintering strategies of egg parasitoids. In: Wajnberg E, Hassan SA (eds) Biological control with egg parasitoids. CAB International, Wallingford, pp 219–244
Bonnemaison L (1972) Diapause et superparasitisme chez Trichogramma evanescens Westw. (Hymenoptera, Trichogrammatidae). Bull Soc Entomol Fr 77:122–132
Bordenstein SR, Werren JH (2000) Do Wolbachia influence fecundity in Nasonia vitripennis? Heredity 84(1):54–62. https://doi.org/10.1046/j.1365-2540.2000.00637.x
Danilevski AS (1965) Photoperiodism and seasonal development of insects. Oliver & Boyd, London
Dedeine F, Vavre F, Fleury F, Loppin B, Hochberg ME, Treau MB (2001) Removing symbiotic Wolbachia bacteria specifically inhibits oogenesis in a parasitic wasp. Proc Natl Acad Sci U S A 98(11):6247–6252. https://doi.org/10.1073/pnas.101304298
Denlinger DL (2002) Regulation of diapause. Annu Rev Entomol 47(1):93–122. https://doi.org/10.1146/annurev.ento.47.091201.145137
Girin C, Bouletreau M (1995) Microorganism-associated variation in host infestation efficiency in a parasitoid wasp Trichogramma bourarachae (Hymenoptera: Trichogrammatidae). Experientia 52:398–401
Goto SG, Denlinger DL (2002) Short-day and long-day expression patterns of genes involved in the flesh fly clock mechanism: period, timeless, cycle and cryptochrome. J Insect Physiol 48(8):803–816. https://doi.org/10.1016/S0022-1910(02)00108-7
Gu HF, Xiao JH, Niu LM, Wang B, Ma GC, Dunn DW, Huang DW (2014) Adaptive evolution of the circadian gene timeout in insects. Sci Rep 4:4212. https://doi.org/10.1038/srep04212
Hardin PE (2005) The circadian timekeeping system of Drosophila. Curr Biol 15:714–722
Hohmann CL, Luck RF, Stouthamer R (2001) Effect of Wolbachia on the survival and reproduction of Trichogramma kaykai Pinto & Stouthamer (Hymenoptera: Trichogrammatidae). Neotrop Entomol 30(4):607–612. https://doi.org/10.1590/S1519-566X2001000400015
Huigens ME, Hohmann CL, Luck RF, Gort G, Stouthamer R (2004) Reduced competitive ability due to Wolbachia infection in the parasitoid wasp Trichogramma kaykai. Entomol Exp Appl 110(2):115–123. https://doi.org/10.1111/j.0013-8703.2004.00126.x
Hurst GDD, Johnson AP, Schulenburg JHGVD, Fuyama Y (2000) Male-killing Wolbachia in Drosophila: a temperature-sensitive trait with a threshold bacterial density. Genetics 156(2):699–709
Hussain M, Frentiu FE, Moreira LA, O’Neill SL, Asgari S (2011) Wolbachia uses host microRNAs to manipulate host gene expression and facilitate colonization of the dengue vector Aedes aegypti. Proc Natl Acad Sci U S A 108(22):9250–9255. https://doi.org/10.1073/pnas.1105469108
Ikeno T, Tanaka SI, Numata H, Goto SG (2010) Photoperiodic diapause under the control of circadian clock genes in an insect. BMC Biol 8(1):116. https://doi.org/10.1186/1741-7007-8-116
Ikeno T, Numata H, Goto SG (2011) Circadian clock genes period and cycle regulate photoperiodic diapause in the bean bug Riptortus pedestris males. J Insect Physiol 57(7):935–938. https://doi.org/10.1016/j.jinsphys.2011.04.006
Ivanov MF, Reznik SY (2008) Photoperiodic regulation of the diapause of the progeny in Trichogramma embryophagum Htg. (Hymenoptera, Trichogrammatidae): dynamics of sensitivity to photoperiod at the immature stages of maternal females. Entomol Rev 88(3):261–268. https://doi.org/10.1134/S0013873808030019
Kambris Z, Blagborough AM, Pinto SB, Blagrove MSC, Godfray HCJ, Sinden RE, Sinkins SP (2010) Wolbachia stimulates immune gene expression and inhibits Plasmodium development in Anopheles gambiae. PLoS Pathog 6(10):e1001143. https://doi.org/10.1371/journal.ppat.1001143
Kishani Farahani H, Ashouri A, Goldansaz SH, Farrokhi S, Ainouche A, van Baaren J (2015) Does Wolbachia infection affect decision-making in a parasitic wasp? Entomol Exp Appl 155(2):102–116. https://doi.org/10.1111/eea.12293
Kremer N, Charif D, Henri H, Gavory F, Wincker P, Mavingui P, Vavre F (2012) Influence of Wolbachia on host gene expression in an obligatory symbiosis. BMC Microbiol 12(1):S7. https://doi.org/10.1186/1471-2180-12-S1-S7
Majercak J, Sidote D, Hardin PE, Edery I (1999) How a circadian clock adapts to seasonal decreases in temperature and day length. Neuron 24(1):219–230. https://doi.org/10.1016/S0896-6273(00)80834-X
Meuti ME, Denlinger DL (2013) Evolutionary links between circadian clocks and photoperiodic diapause in insects. Integr Comp Biol 53(1):131–143. https://doi.org/10.1093/icb/ict023
Meyer P, Saez L, Young MW (2006) PER-TIM interactions in living Drosophila cells: an interval timer for the circadian clock. Science 311(5758):226–229. https://doi.org/10.1126/science.1118126
Moreira LA, Iturbe-Ormaetxe I, Jeffery JA, Lu G, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M, Hugo LE, Johnson KN, Kay BH, McGraw EA, van den Hurk AF, Ryan PA, O'Neill SL (2009) A Wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and Plasmodium. Cell 139(7):1268–1278. https://doi.org/10.1016/j.cell.2009.11.042
Moriyama Y, Sakamoto T, Karpova SG, Matsumoto A, Noji S, Tomioka K (2008) RNA interference of the clock gene period disrupts circadian rhythms in the cricket Gryllus bimaculatus. J Biol Rhythm 23(4):308–318. https://doi.org/10.1177/0748730408320486
O’Neill SL, Karr T (1990) Bidirectional incompatibility between conspecific populations of Drosophila simulans. Nature 348(6297):178–180. https://doi.org/10.1038/348178a0
Pinto JD, Stouthamer R (1994) Systematics of Trichogrammatidae with emphasis on Trichogramma. In: Wajnberg E, Hassan SA (eds) Biological control with egg parasitoids. CAB International, Wallingford, pp 1–36
Pintureau B, Grenier S, Heddi A, Charles H (2002) Biodiversity of Wolbachia and of their effects in Trichogramma (Hymenoptera: Trichogrammatidae). Ann Soc Entomol Fr 38(4):333–338. https://doi.org/10.1080/00379271.2002.10697347
Pintureau B, Pizzol J, Bolland P (2003) Effects of endosymbiotic Wolbachia on the diapause in Trichogramma hosts and effects of the diapause on Wolbachia. Entomol Exp Appl 106(3):193–200. https://doi.org/10.1046/j.1570-7458.2003.00024.x
Pizzol J, Pintureau B (2008) Effect of photoperiod experienced by parents on diapause induction in Trichogramma cacoeciae. Entomol Exp Appl 127(1):72–77. https://doi.org/10.1111/j.1570-7458.2008.00671.x
Poorjavad N (2011) Morphological, molecular and reproductive compatibility studies on the systematic of the genus Trichogramma Westwood (Hymenoptera: Trichogrammatidae) in Tehran and Mazandran Province (Iran). PhD dissertation, University of Tehran, Iran
Reznik SY (2011) Ecological and evolutionary aspects of photo-thermal regulation of diapause in Trichogramma. J Evol Biochem Physiol 47(6):512–523. https://doi.org/10.1134/S0022093011060020
Reznik SY, Kats TS (2004) Exogenous and endogenous factors inducing diapause in Trichogramma principium Sug. & Sor. (Hymenoptera, Trichogrammatidae). Entomol Rev 84:963–970
Reznik SY, Voinovich ND, Vaghina NP (2011) Maternal influence on diapause induction in Trichogramma (Hymenoptera, Trichogrammatidae): the dynamics of photosensitivity. J Appl Entomol 135(6):438–445. https://doi.org/10.1111/j.1439-0418.2010.01563.x
Reznik SY, Vaghina NP, Vasiljev AL (2013) Photo-thermal regulation of diapause in Trichogramma piceum Djur. (Hymenoptera, Trichogrammatidae). Entomol Rev 93(1):9–13. https://doi.org/10.1134/S0013873813010028
Rousset F, Bouchon D, Pintureau B, Juchault P, Solignac M (1992) Wolbachia endosymbionts responsible for various alterations of sexuality in arthropods. Proc R Soc Lond B Biol Sci 250(1328):91–98. https://doi.org/10.1098/rspb.1992.0135
Sakamoto T, Uryu O, Tomioka K (2009) The clock gene period plays an essential role in photoperiodic control of nymphal development in the cricket Modicogryllus siamensis. J Biol Rhythm 24(5):379–390. https://doi.org/10.1177/0748730409341523
Sandrelli F, Costa R, Kyriacou CP, Rosato E (2008) Comparative analysis of circadian clock genes in insects. Insect Mol Biol 17(5):447–463. https://doi.org/10.1111/j.1365-2583.2008.00832.x
Saunders DS, Steel CGH, Vafopoulou X, Lewis RD (2002) Insect clocks. Elsevier, Amsterdam
Stolk C, Stouthamer R (1996) Influence of a cytoplasmic incompatibility-inducing Wolbachia on the fitness of the parasitoid wasp Nasonia vitripennis. Proc Sect Exp Appl Entomol Neth Entomol Soc (NEV) 7:33–37
Stouthamer R, Werren JR (1993) Microbes associated with parthenogenesis in wasps of the genus Trichogramma. J Invertebr Pathol 61(1):6–9. https://doi.org/10.1006/jipa.1993.1002
Stouthamer R, Hu J, van Kan FJPM, Platner GR, Pinto JD (1999) The utility of internally transcribed spacer 2 DNA sequences of the nuclear ribosomal gene for distinguishing sibling species of Trichogramma. BioControl 43(4):421–440. https://doi.org/10.1023/A:1009937108715
Tagami Y, Miura K, Stouthamer R (2001) How does infection with parthenogenesis-inducing Wolbachia reduce the fitness of Trichogramma? J Invertebr Pathol 78(4):267–271. https://doi.org/10.1006/jipa.2002.5080
Turley AP (2013) Does Wolbachia modify dengue mosquito biology: investigating effects of Wolbachia infections on Aedes aegypti behaviour, physiology and gene expression. PhD dissertation, School of Biological Sciences, The University of Queensland
Voinovich ND, Umarova TY, Kats TS, Reznik SY (2003) Variation of the photoperiodic reaction in Trichogramma embryophagum (Hymenoptera, Trichogrammatidae). Entomol Obozr 82(2):264–269
Werren JH (1997) Biology of Wolbachia. Annu Rev Entomol 42(1):587–609. https://doi.org/10.1146/annurev.ento.42.1.587
Werren JH (2003) Invasion of the gender benders: by manipulating sex and reproduction in their hosts, many parasites improve their own odds of survival and may shape the evolution of sex itself. J Nat Hist 112(1):58
Yuan Q, Metterville D, Briscoe AD, Reppert SM (2007) Insect cryptochromes: gene duplication and loss define diverse ways to construct insect circadian clocks. Mol Biol Evol 24(4):948–955. https://doi.org/10.1093/molbev/msm011
Yue S, Zuo-Rui S, Zhe W, Hong Yue L (2009) Triple infection of Wolbachia in Trichogramma ostriniae (Hymenoptera: Trichogrammatidae). Acta Entomol Sin 52(4):445–452
Zaslavski VA, Umarova TY (1990) Environmental and endogenous control of diapause in Trichogramma species. Entomophaga 35(1):23–29. https://doi.org/10.1007/BF02374297
Acknowledgements
The authors appreciate Dr. Nafiseh Poorjavad (Isfahan University of Technology, Isfahan, Iran) for the molecular and morphological identification of asexual and sexual T. brassicae and Atsushi Tokuoka and Yuuki Kutaragi (Okayama University, Okayama, Japan) for their kind assistance in the experimental part of this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Claude Desplan
Rights and permissions
About this article
Cite this article
Rahimi-Kaldeh, S., Ashouri, A., Bandani, A. et al. The effect of Wolbachia on diapause, fecundity, and clock gene expression in Trichogramma brassicae (Hymenoptera: Trichogrammatidae). Dev Genes Evol 227, 401–410 (2017). https://doi.org/10.1007/s00427-017-0597-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-017-0597-0