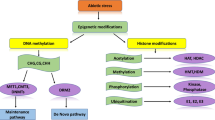
Abstract
Main conclusion
Roots play an important role in adaptive plasticity of rice under dry/direct-sown conditions. However, hypomethylation of genes in leaves (resulting in up-regulated expression) complements the adaptive plasticity of Nagina-22 under DSR conditions.
Abstract
Rice is generally cultivated by transplanting which requires plenty of water for irrigation. Such a practice makes rice cultivation a challenging task under global climate change and reducing water availability. However, dry-seeded/direct-sown rice (DSR) has emerged as a resource-saving alternative to transplanted rice (TPR). Though some of the well-adapted local cultivars are used for DSR, only limited success has been achieved in developing DSR varieties mainly because of a limited knowledge of adaptability of rice under fluctuating environmental conditions. Based on better morpho-physiological and agronomic performance of Nagina-22 (N-22) under DSR conditions, N-22 and IR-64 were grown by transplanting and direct-sowing and used for whole genome methylome analysis to unravel the epigenetic basis of adaptive plasticity of rice. Comparative methylome and transcriptome analyses indicated a large number (4078) of genes regulated through DNA methylation/demethylation in N-22 under DSR conditions. Gene × environment interactions play important roles in adaptive plasticity of rice under direct-sown conditions. While genes for pectinesterase, LRK10, C2H2 zinc-finger protein, splicing factor, transposable elements, and some of the unannotated proteins were hypermethylated, the genes for regulation of transcription, protein phosphorylation, etc. were hypomethylated in CG context in the root of N-22, which played important roles in providing adaptive plasticity to N-22 under DSR conditions. Hypomethylation leading to up-regulation of gene expression in the leaf complements the adaptive plasticity of N-22 under DSR conditions. Moreover, differential post-translational modification of proteins and chromatin assembly/disassembly through DNA methylation in CHG context modulate adaptive plasticity of N-22. These findings would help developing DSR cultivars for increased water-productivity and ecological efficiency.

















Similar content being viewed by others
Data availability
The raw data for WGBS (submission ID: SUB11780954) and transcriptome (BioProject ID PRJNA805549) have been submitted to NCBI Sequence Read Archive (SRA) database (https://www.ncbi.nlm.nih.gov/sra), which can also be made available on request.
Abbreviations
- DEG:
-
Differentially expressed gene
- DMR:
-
Differentially methylated region
- DSR:
-
Dry/direct-sown rice
- GO:
-
Gene ontology
- TE:
-
Transposable element
- TF:
-
Transcription factor
- TPR:
-
Transplanted rice
References
Anastasiadi D, Esteve-Codina A, Piferrer F (2018) Consistent inverse correlation between DNA methylation of the first intron and gene expression across tissues and species. Epigenetics Chromatin 11:37
Azmi M, Chin DV, Vongsaroj P, Johnson DE (2005) Emerging issues in weed management of direct-seeded rice in Malaysia, Vietnam, and Thailand. In: Rice is life: Scientific perspectives for the 21st century. Proc World Rice Res Conf Tsukuba, Japan, November 4–7, 2004, 196–197
Bam B, Wang H, Yang X, Zhao JF, Wang CG (2002) Aerobic rice (Han Dao): A new way of growing rice in water-short areas. Proc 12th Int Soil Conservation Organization Conf, Tsinghua University Press, Beijing, 26:31
Bewick AJ, Schmitz RJ (2017) Gene body DNA methylation in plants. Curr Opin Plant Biol 36:103–110
Bhatia H, Khemka N, Jain M, Garg R (2018) Genome-wide bisulphite-sequencing reveals organ-specific methylation patterns in chickpea. Sci Rep 8:1–12
Borgel J, Guibert S, Li Y, Chiba H, Schübeler D, Sasaki H, Forné T, Weber M (2010) Targets and dynamics of promoter DNA methylation during early mouse development. Nat Genet 42:1093–1100
Casartelli A, Riewe D, Hubberten HM, Altmann T, Hoefgen R, Heuer S (2018) Exploring traditional aus-type rice for metabolites conferring drought tolerance. Rice 11:9
Chaudhary A, Venkatramanan V, Kumar Mishra A, Sharma S (2023) Agronomic and environmental determinants of direct seeded rice in South Asia. Circ Econ Sustain 3(1):253–290
Chu S, Zhang X, Yu K, Lv L, Sun C, Liu X et al (2020) Genome-wide analysis reveals dynamic epigenomic differences in soybean response to low-phosphorus stress. Int J Mol Sci 21:6817
Dubin MJ, Zhang P, Meng D, Remigereau MS, Osborne EJ, Paolo Casale F et al (2015) DNA methylation in Arabidopsis has a genetic basis and shows evidence of local adaptation. Elife 4:e05255
Garg R, Chevala VN, Shankar R, Jain M (2015) Divergent DNA methylation patterns associated with gene expression in rice cultivars with contrasting drought and salinity stress response. Sci Rep 5:4922
Gent JI, Ellis NA, Guo L, Harkess AE, Yao Y, Zhang X et al (2013) CHH islands: de novo DNA methylation in near-gene chromatin regulation in maize. Genome Res 23:628–637
Godfrey K, Lillycrop K, Burdge G et al (2007) Epigenetic mechanisms and the mismatch concept of the developmental origins of health and disease. Pediatr Res 61:5–10
Grzybkowska D, Nowak K, Gaj MD (2020) Hypermethylation of auxin-responsive motifs in the promoters of the transcription factor genes accompanies the somatic embryogenesis induction in Arabidopsis. Int J Mol Sci 21:6849
Gutierrez-Arcelus M, Ongen H, Lappalainen T, Montgomery SB, Buil A, Yurovsky A et al (2015) Tissue-specific effects of genetic and epigenetic variation on gene regulation and splicing. PLoS Genet 11:e1004958
Hashimoto K, Otero M, Imagawa K, de Andrés MC, Coico JM, Roach HI et al (2013) Regulated transcription of human matrix metalloproteinase 13 (mmp13) and interleukin-1beta (IL1B) genes in chondrocytes depends on methylation of specific proximal promoter CpG sites. J Biol Chem 288:10061–10072
Hu G, Huang B, Wang K, Frasse P, Maza E, Djari A et al (2021) Histone posttranslational modifications rather than DNA methylation underlie gene reprogramming in pollination-dependent and pollination-independent fruit set in tomato. New Phytol 229:902–919
Huff JT, Zilberman D, Roy SW (2016) Mechanism for DNA transposons to generate introns on genomic scales. Nature 538:533–536
Jabre I, Chaudhary S, Guo W, Kalyna M, Reddy AS, Chen W et al (2021) Differential nucleosome occupancy modulates alternative splicing in Arabidopsis thaliana. New Phytol 229:1937–1945
Jagadish S, Cairns J, Lafitte R, Wheeler T, Price A, Craufurd P (2010) Genetic analysis of heat tolerance at anthesis in rice. Crop Sci 50:1633–1641
Jalota SK, Singh KB, Chahal GBS, Gupta RK, Chakraborty S, Sood A, Ray SS, Panigrahy S (2009) Integrated effect of transplanting date, cultivar and irrigation on yield, water saving and water productivity of rice (Oryza sativa L.) in Indian Punjab: Field and simulation study. Agric Water Manage 96:1096–1104
Jat RK, Meena VS, Kumar M, Jakkula VS, Reddy IR, Pandey AC (2022) Direct seeded rice: Strategies to improve crop resilience and food security under adverse climatic conditions. Land 11(3):382. https://doi.org/10.3390/land11030382
Jegu T, Latrasse D, Delarue M, Hirt H, Domenichini S, Ariel F et al (2014) The BAF60 subunit of the SWI/SNF chromatin-remodeling complex directly controls the formation of a gene loop at FLOWERING LOCUS C in Arabidopsis. Plant Cell 26:538–551
Kang JG, Park JS, Ko JH, Kim YS (2019) Regulation of gene expression by altered promoter methylation using a CRISPR/Cas9-mediated epigenetic editing system. Sci Rep 9:11960
Karlova R, Boer D, Hayes S, Testerink C (2021) Root plasticity under abiotic stress. Plant Physiol 187:1057–1070
Koevoets IT, Venema JH, Elzenga JTM, Testerink C (2016) Roots withstanding their environment: Exploiting root system architecture responses to abiotic stress to improve crop tolerance. Front Plant Sci 7:1335
Krueger F, Andrews SR (2011) Bismark: a flexible aligner and methylation caller for bisulfite-seq applications. Bioinformatics 27:1571–1572
Kumar S (2019a) Epigenetics and epigenomics for crop improvement: Current opinion. Adv Biotechnol Microbiol 14:555879. https://doi.org/10.19080/AIBM.2019.14.555879
Kumar S (2019b) Genome editing to epigenome editing: towards unraveling the enigmas in developmental biology. Trends Dev Biol 12:31–38. https://doi.org/10.31300/TDB.12.2019.31-38
Kumar S (2020) Abiotic stresses and their effects on plant growth, yield and nutritional quality of agricultural produce. Int J Food Sci Agriculture 4(4):367–378. https://doi.org/10.26855/ijfsa.2020.12.002
Kumar S (2021) Saving water for ecological integrity: agricultural perspective of per drop more crop. Int J Environ Sci Nat Res 28:1–7. https://doi.org/10.19080/IJESNR.2021.28.556248
Kumar S, Mohapatra T (2021) Dynamics of DNA methylation and its functions in plant growth and development. Front Plant Sci 12:596236
Kumar S, Singh A (2016) Epigenetic regulation of abiotic stress tolerance in plants. Adv Plants Agric Res 5:517–521. https://doi.org/10.15406/apar.2016.05.00179
Kumar S, Chinnusamy V, Mohapatra T (2018) Epigenetics of modified DNA bases: 5-methylcytosine and beyond. Front Genet 9:640
Kumar S, Kaur S, Seem K, Kumar S, Mohapatra T (2021) Understanding 3D genome organization and its effect on transcriptional gene regulation under environmental stress in plant: A chromatin perspective. Front Cell Dev Biol 9:774719
Kumar S, Agrawal A, Seem K et al (2022a) Transcriptome analysis of a near-isogenic line and its recurrent parent reveals the role of Pup1 QTL in phosphorus deficiency tolerance of rice at tillering stage. Plant Mol Biol 109:29–50
Kumar S, Kumar S, GopalaKrishnan S, Mohapatra T (2022b) Molecular basis of genetic plasticity to varying environmental conditions on growing rice by direct-sowing and transplanting with imposition of drought stress. Front Plant Sci 13:1013207
Kumar S, Seem K, Kumar S et al (2022c) RNA-seq analysis reveals the genes/pathways responsible for genetic plasticity of rice to varying environmental conditions on direct-sowing and transplanting. Sci Rep 12:2241
Kumar S, Seem K, Kumar S, Vinod KK, Chinnusamy V, Mohapatra T (2022d) Pup1 QTL regulates gene expression through epigenetic modification of DNA under phosphate starvation stress in rice. Front Plant Sci 13:871890
Kumar S, Seem K, Mohapatra T (2023) Biochemical and epigenetic modulations under drought: remembering the stress tolerance mechanism in rice. Life (basel) 13(5):1156. https://doi.org/10.3390/life13051156
Le TN, Schumann U, Smith NA et al (2014) DNA demethylases target promoter transposable elements to positively regulate stress responsive genes in Arabidopsis. Genome Biol 15:458
Lenka SK, Katiyar A, Chinnusamy V, Bansal KC (2011) Comparative analysis of drought-responsive transcriptome in Indica rice genotypes with contrasting drought tolerance. Plant Biotechnol J 9:315–327
Li X, Zhu J, Hu F, Ge S, Ye M, Xiang H et al (2012) Single-base resolution maps of cultivated and wild rice regulatory roles of DNA methylation in plant gene expression. BMC Genomics 13:300
Li Y, Kumar S, Qian W (2018) Active DNA demethylation: Mechanism and role in plant development. Plant Cell Rep 37(1):77–85. https://doi.org/10.1007/s00299-017-2215-z
Liang P, Song F, Ghosh S, Morien E, Qin M, Mahmood S et al (2011) Genome-wide survey reveals dynamic widespread tissue-specific changes in DNA methylation during development. BMC Genomics 12:231
Liu H, Hussain S, Zheng M et al (2014) Progress and constraints of dry direct-seeded rice in China. J Food Agric Environ 12:465–472
Liu H, Hussain S, Zheng M et al (2015) Dry seeded rice as an alternative to transplanted-flooded rice in Central China. Agron Sustain Develop 35:285–294
Mager S, Ludewig U (2018) Massive loss of DNA methylation in nitrogen-, but not in phosphorus-deficient Zea mays roots is poorly correlated with gene expression differences. Front Plant Sci 9:497
Martin GT, Seymour DK, Gaut BS (2021) CHH methylation islands: A nonconserved feature of grass genomes that is positively associated with transposable elements but negatively associated with gene-body methylation. Genome Biol Evol 13(8):evab144. https://doi.org/10.1093/gbe/evab144
Niyikiza D, Piya S, Routray P, Miao L, Kim W-S, Burch-Smith T et al (2020) Interactions of gene expression, alternative splicing, and DNA methylation in determining nodule identity. Plant J 103:1744–1766
Ong-Abdullah M, Ordway J, Jiang N et al (2015) Loss of Karma transposon methylation underlies the mantled somaclonal variant of oil palm. Nature 525:533–537
Rao AN, Johnson DE, Sivaprasad B, Ladha JK, Mortimer AM (2007) Weed management in direct-seeded rice. Adv Agron 93:155–255
Rogozin IB, Carmel L, Csuros M, Koonin EV (2012) Origin and evolution of spliceosomal introns. Biol Direct 7:11
Sagare DB, Abbai R, Jain J et al (2020) More and more of less and less: Is genomics-based breeding of dry direct-seeded rice (DDSR) varieties the need of hour? Plant Biotechnol J 18:2173–2186
Sandhu N, Subedi SR, Singh VK et al (2019) Deciphering the genetic basis of root morphology, nutrient uptake, yield, and yield related traits in rice under dry direct-seeded cultivation systems. Sci Rep 9:9334
Secco D, Wang C, Shou H, Schultz MD, Chiarenza S et al (2015) Stress induced gene expression drives transient DNA methylation changes at adjacent repetitive elements. Elife 4:e09343
Seem K, Kumar S (2021) Cultivation of rice: evolving towards climate-smart crops for precision in resource use efficiency. MC Agric Env Sci 1:41–49
Shrestha R, Al-Shugeairy Z, Al-Ogaidi F et al (2014) Comparing simple root phenotyping methods on a core set of rice genotypes. Plant Biol 16:632–642
Smith S, De Smet I (2012) Root system architecture: insights from Arabidopsis and cereal crops. Philos Trans R Soc B 367:1441–1452. https://doi.org/10.1098/rstb.2011.0234
Smith P, Martino D, Cai Z, Gwary D, Janzen H, Kumar P et al (2008) Greenhouse gas mitigation in agriculture. Philos Trans R Soc B 363:789–813
Sun L, Hussain S, Liu H et al (2015) Implications of low sowing rate for hybrid rice varieties under dry direct-seeded rice system in Central China. Field Crop Res 175:87–95
Sura W, Kabza M, Karlowski WM et al (2017) Dual role of the histone variant H2A.Z in transcriptional regulation of stress-response genes. Plant Cell 29:791–807
Tuong TP, Bouman BAM, Mortimer M (2005) More rice, less water-integrated approaches for increasing water productivity in irrigated rice-based systems in Asia. Plant Prod Sci 8:231–241
Vikram P, Swamy BM, Dixit S, Ahmed HU, Cruz MTS, Singh AK, Singh NK (2011) qDTY1.1 a major QTL for rice grain yield under reproductive-stage drought stress with a consistent effect in multiple elite genetic backgrounds. BMC Genet 12:89
Vining KJ, Romanel E, Jones RC, Klocko A, Alves-Ferreira M, Hefer CA et al (2015) The floral transcriptome of Eucalyptus grandis. New Phytol 206(4):1406–1422
Wada Y, Miyamoto K, Kusano T, Sano H (2004) Association between Up-regulation of stress-responsive genes and hypomethylation of genomic DNA in tobacco plants. Mol Genet Genomics 271:658–666
Wang Y, Wang X, Lee TH, Mansoor S, Paterson AH (2013) Gene body methylation shows distinct patterns associated with different gene origins and duplication modes and has a heterogeneous relationship with gene expression in Oryza sativa (rice). New Phytol 198:274–283
Wang P, Xia H, Zhang Y, Zhao S, Zhao C, Hou L et al (2015) Genome-wide high-resolution mapping of DNA methylation identifies epigenetic variation across embryo and endosperm in maize (Zea mays). BMC Genomics 16:21
Wang L, Cao S, Wang P, Lu K, Song Q, Zhao F-J, Chen ZJ (2021a) DNA hypomethylation in tetraploid rice potentiates stress-responsive gene expression for salt tolerance. Proc Natl Acad Sci USA 118:e2023981118
Wang X, Hao D, Kadarmideen HN (2021b) GeneDMRs: An R package for gene-based differentially methylated regions analysis. J Comput Biol 28:304–3016
Yong-Villalobos L, González-Morales SI, Wrobel K et al (2015) Methylome analysis reveals an important role for epigenetic changes in the regulation of the Arabidopsis response to phosphate starvation. Proc Natl Acad Sci USA 112:E7293–E7302
Yun SI, Wada Y, Maeda T, Miura K, Watanabe K (1997) Growth and yield of japonica x indica hybrid cultivars under direct seeding and upland cultivation condition. Japan J Crop Sci 66:386–393
Zhang X, Wang T (2021) Plant 3D chromatin organization: Important insights from chromosome conformation capture analyses of the last 10 years. Plant Cell Physiol 62:648–1661
Zhang M, Xu C, von Wettstein D, Liu B (2011) Tissue-specific differences in cytosine methylation and their association with differential gene expression in Sorghum bicolor. Plant Physiol 156:1955–1966
Zhang H, Lang Z, Zhu J-K (2018) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19:489–506
Zheng X, Chen L, Xia H, Wei H, Lou Q, Li M et al (2017) Transgenerational epimutations induced by multi-generation drought imposition mediate rice plant’s adaptation to drought condition. Sci Rep 7:17885
Zilberman D, Gehring M, Tran RK, Ballinger T, Henikoff S (2007) Genome-wide analysis of Arabidopsis thaliana DNA methylation uncovers an interdependence between methylation and transcription. Nat Genet 39:61–69
Funding
The research was carried out with financial support from the National Agricultural Science Fund (NASF/ABP-70161/2018–19) from the Indian Council of Agricultural Research, Govt. of India, New Delhi, India.
Author information
Authors and Affiliations
Contributions
SuK and TM conceived the experiments. GKS provided genetically pure rice cultivars and helped grow them through different methods of planting over the generations. KS, SuK and AS carried out the experiments. SaK performed bioinformatic analyses. SuK and AS prepared the initial draft. TM revised the manuscript. All the authors approved the finalized draft for publication.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests. Santosh Kumar is employed at the Decode Genomics Private Limited, New Delhi, and the work was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, S., Seem, K., Kumar, S. et al. DNA methylome analysis provides insights into gene regulatory mechanism for better performance of rice under fluctuating environmental conditions: epigenomics of adaptive plasticity. Planta 259, 4 (2024). https://doi.org/10.1007/s00425-023-04272-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04272-3